-
化学通讯在昆虫的整个生命周期中起着至关重要的作用,是它们可以精准定位栖境、食物和配偶的基础。嗅觉是昆虫最重要的化学通讯途径。昆虫的嗅觉系统主要包括两部分:位于体壁表面特化的外周嗅觉系统和位于大脑的中枢神经系统[1],而最重要的外周嗅觉器官为位于头部前侧的触角。触角体壁表面覆盖有大量由触角表皮特化形成的毛状突起,称为触角感受器,它们是嗅觉识别的最小形态学单元[2]。化学感受器内神经元上表达有多种化学受体蛋白,如气味分子受体(odorant receptors, Ors)、味觉受体(gustatory receptors, Grs)、离子型受体(ionotropic receptors, IRs)等[3],它们负责识别和编码环境中的化学信息。通过免疫组织化学(immunohistochemical,IHC)、原位杂交(in situ hybridization,ISH)和单感器记录(single sensillum recording,SSR)等技术,解析了大量的昆虫化学受体基因在感受器内的细胞定位和化学编码特征,并依此逐步揭示了一些典型的嗅觉行为机制[4-11]。上述这些工作均需依托于对触角感受器特征的准确掌握,可以说研究昆虫触角感受器的种类、形态结构和分布特征是探索昆虫嗅觉通讯和行为机制的必要前提之一。
依托扫描和透射电子显微镜技术,大量昆虫触角感受器的基本形态和类型被描述和鉴定。总体上,依据形态分类,昆虫触角感受器主要包括刺形感器、毛形感器、锥形感器、腔锥形感器、板形感器等10余类,它们大多行使着化学编码功能[12]。例如,多数毛形感器和锥形感器是嗅觉感器, 毛形感器主要识别性外激素, 锥形感器主要识别普通气味[13]。除了化学编码功能外,触角感受器还同时具有机械感知、温湿度感知等功能[12, 14]。
白蜡吉丁肿腿蜂(Sclerodermus pupariae)是多种林木蛀干害虫的重要天敌,已经广泛应用于白蜡窄吉丁(Agrilus planipennis)、苹小吉丁(A. mali)、栗山天牛(Massicus raddei)、光肩星天牛(Anoplophora glabripennis)等蛀干害虫的生物防治[15]。该寄生蜂可寄生寄主种类的多样性,暗示着其可识别寄主和栖境所传递出的丰富的化学信号。然而,白蜡吉丁肿腿蜂的化学通讯机制一直处于空白。作者在该寄生蜂触角转录组中鉴定到了97个气味受体基因,并拟对其在触角感受器内的表达和定位开展研究,进一步研究相关基因的嗅觉编码功能,这些工作需要以准确掌握触角感受器类型和形态特征为基础。几种近缘种,如管氏肿腿蜂(S. guani)、川硬皮肿腿蜂(S. sichuanensis)、松褐天牛肿腿蜂(S. alternatusi)的触角感受器超微结构已有前期研究[16-18]。虽然这3种肿腿蜂亲缘关系很近,寄主范围也大致相同[19],但它们的触角感受器类型和分布特征却差异很大。例如,前期研究认为松褐天牛肿腿蜂触角上无刺形感器,而该类感受器在管氏肿腿蜂触角上却有分布,且存在2个亚型[17-18]。在管氏肿腿蜂和松褐天牛肿腿蜂触角上均发现2种栓锥形感器(St1和St2)。管氏肿腿蜂中,St1仅存在于雌蜂触角,St2在雌雄蜂触角上均有分布[17]。然而,对松褐天牛肿腿蜂,St1同样被发现特异性分布于雌蜂触角,但St2仅存在于雄蜂触角[18]。另外,川硬皮肿腿蜂的触角分节和亚节数与管氏肿腿蜂和松褐天牛肿腿蜂完全不同[16]。因此,几个近缘种的研究结果不足以被完全用于代表白蜡吉丁肿腿蜂触角及其感受器的结构。为了更准确地掌握白蜡吉丁肿腿蜂触角形态特征,有必要对其超微结构进行重新研究。本研究利用扫描电子显微镜观察了白蜡吉丁肿腿蜂雌性和雄性成虫触角,并对感受器的类型、形态、数量和分布规律做了进一步的描述和统计,以期能更准确地掌握该寄生蜂外周嗅觉器官的特征,这将为后续研究各类感受器的化学识别功能和该寄生蜂的嗅觉行为机制提供基础资料。
-
白蜡吉丁肿腿蜂建群种自然寄生于危害绒毛白蜡(Fraxinus velutina Torr.)的白蜡窄吉丁(Agrilus planipennis)幼虫,采集地为天津官港森林公园(38°56′ N,117°29′ E)。建群蜂带回实验室后,以麻竖毛天牛(Thyestilla gebleri)老熟幼虫作寄主,在温度25 ºC,相对湿度RH=60%,光周期L/D=8 h/16 h条件下连续繁殖45代,选择5日龄活跃成虫作为供试虫源。
-
随机选择雌雄成虫各10头,在体视显微镜下,用显微镊于每头成虫取1根触角并置于1.5 mL离心管中(左侧5根,右侧5根),离心管内装有1 mL的1 × PBS缓冲液(Invitrogen)。超声清洗存有触角的离心管30 s,重复3次,每次更换新的缓冲液。清洗完成后,将触角取出分别置于60%、70%、80%、90%和100%的乙醇中逐级脱水,每次脱水10 min。再将脱水后的触角置于CO2临界点干燥仪(Leica EM CPD300)内干燥3 h。干燥完成后,将触角粘合在导电胶上,并随即在真空镀膜仪(Leica EM ACE600)内喷金。以上各步,雌雄蜂的触角样品均分开取样和制备。
-
完成样品制备的触角在扫描电子显微镜(Hitachi SU8010)下观察和拍摄,加速电压为5 kV。对所有触角样本(♀=10,♂=10)逐一拍摄,拍摄要点包括触角整体和各节形态、所有感受器的基本形态和表面特征。在Adobe Photoshop 2020下检视所有照片,对触角感受器进行分类,标准参考Schneider[14]、Zacharuk[20]和Bleeker et al.[21]。以每根触角为一个重复,利用Adobe Photoshop 2020标尺工具测量触角各节长度和宽度。测量雌蜂和雄蜂触角各类感器的长度,随机抽样,样本量为15~40。以每根触角为一个重复,统计各类感受器在雌雄蜂触角各节上的分布情况和数量。
-
以触角各节的长度,加和得到每根触角总长度。比较雌蜂和雄蜂触角总长度的差异,比较雌蜂和雄蜂触角各节长度和宽度的差异。某种感受器在每根触角上的总数,由其在触角各节上统计到的数量加和得到。对雌蜂和雄蜂共有的感受器,比较其长度和在每根触角上数量的差异。雌蜂或雄蜂特有的感受器,仅统计其长度和数量的均值。以上差异性检验用独立样本t检验完成,数据统计用SPSS 20完成,柱形图用GraphPad Prism 7绘制。
-
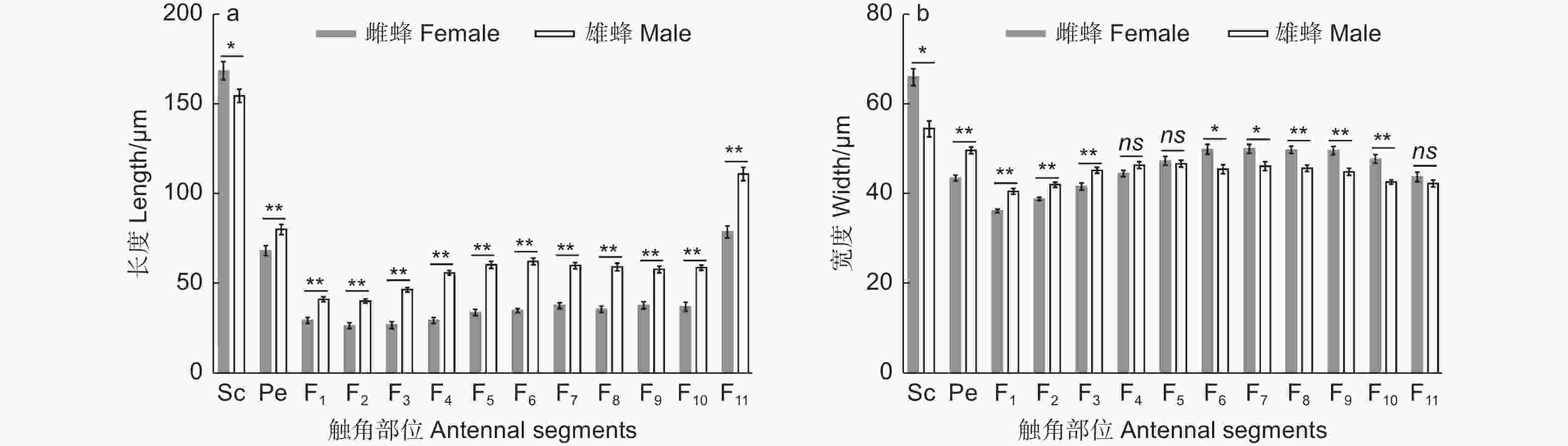
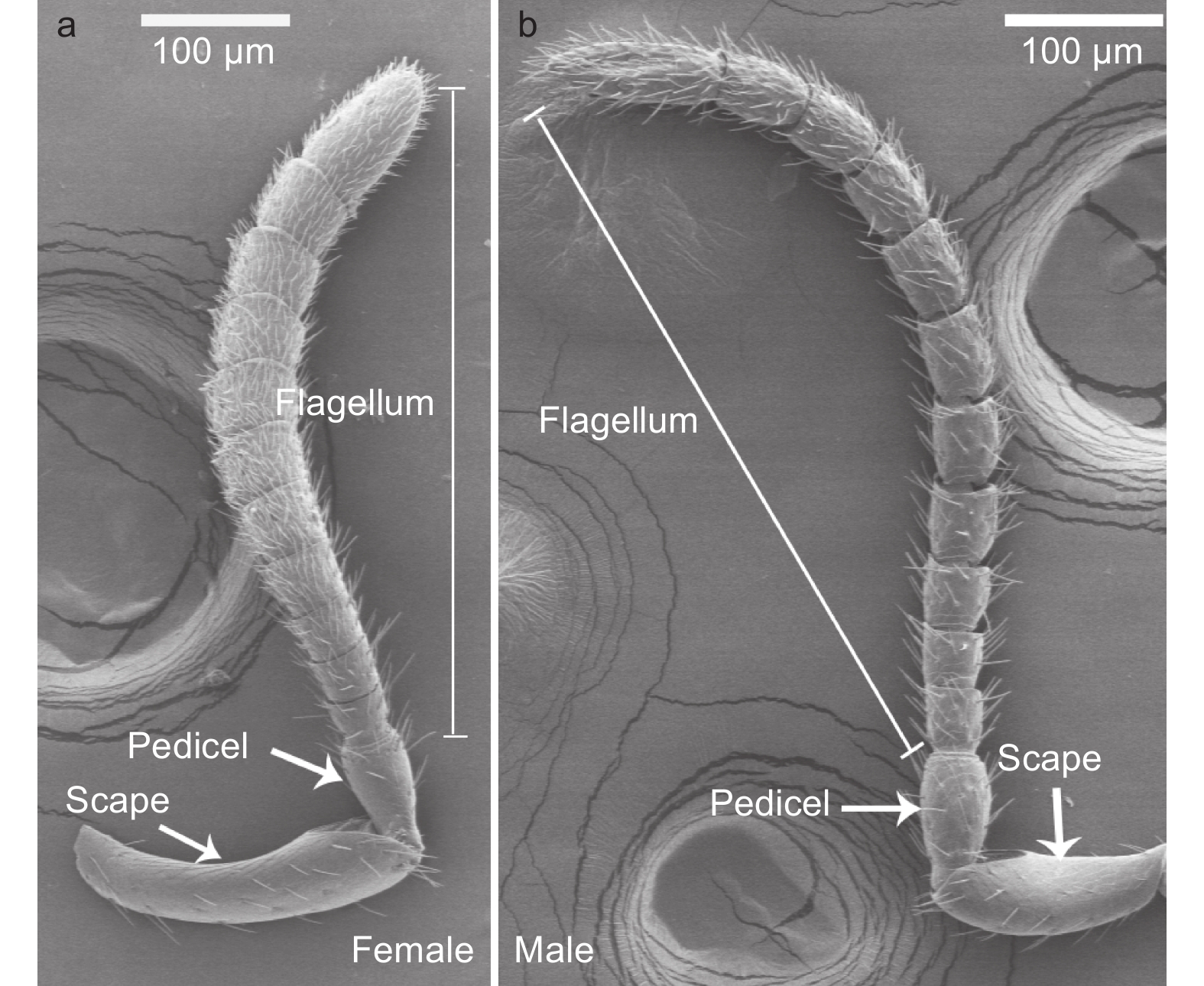
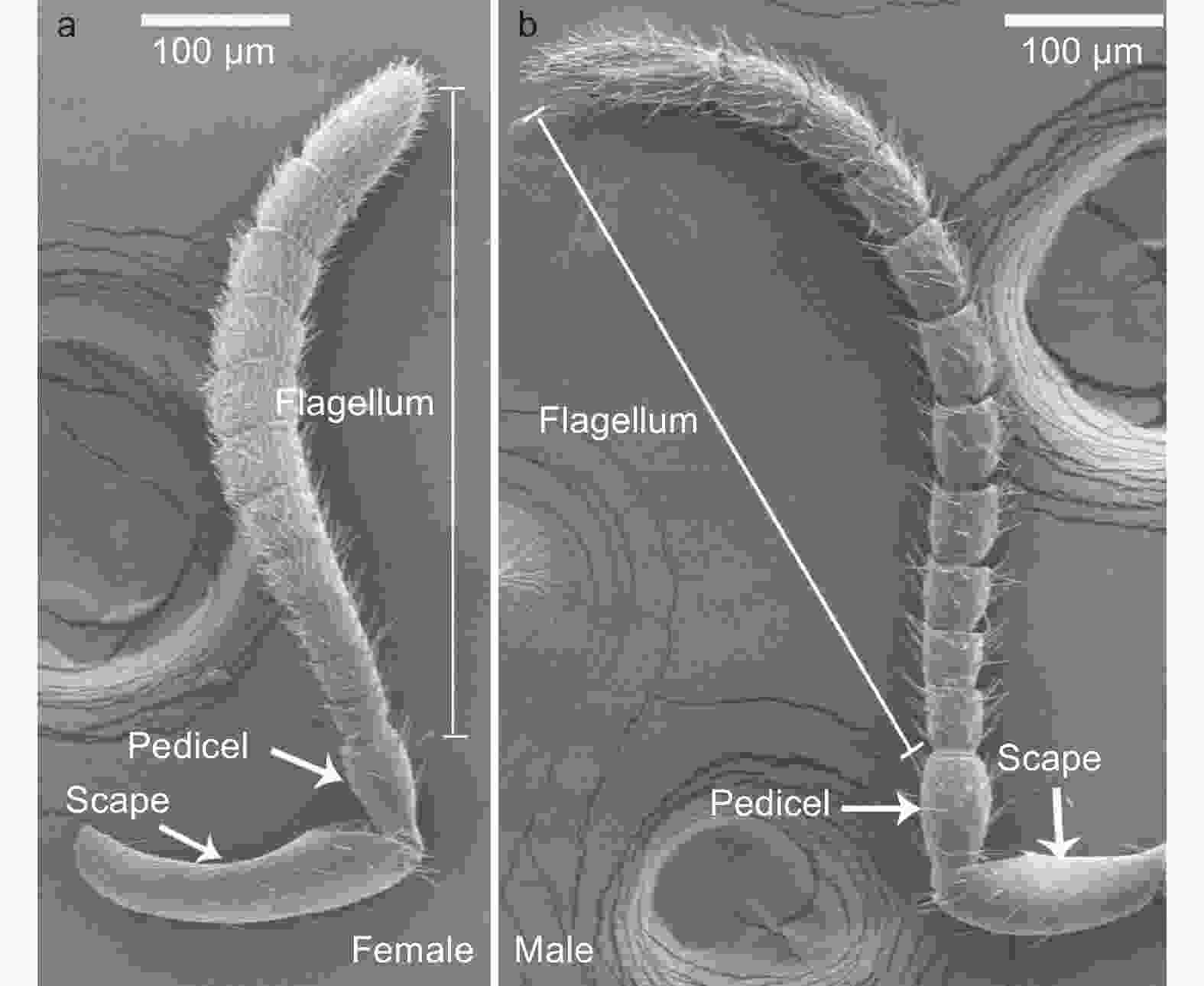
白蜡吉丁肿腿蜂雌雄成虫的触角均呈膝状,由柄节(scape)、梗节(pedicel)和鞭节(flagellum)3部分构成,其中雌雄蜂触角的鞭节均具有11个亚节,雌雄蜂触角在基本构成和节数上无差异(图1)。雌蜂触角平均长度为647.32 μm,显著短于雄蜂触角,后者的平均长度为890.18 μm(t=−13.96,df=18,P<0.01)。雌蜂触角柄节显著长于雄蜂,梗节和鞭节的各亚节均显著短于雄蜂(图2a,表1)。雌蜂触角柄节、鞭小节F6-10较雄蜂更宽,但其梗节和鞭小节F1-3较雄蜂更窄,雌雄蜂鞭小节F4-5、F11的宽度无显著差异(图2b 、表1)。
表 1 雌雄蜂触角各节长和宽的差异检验统计值
Table 1. Difference test parameters of antennal size between female and male parasitoids
变量
Variable统计量
Statistical parameter触角部位
Antennal segmentsSc Pe F1 F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 长度 Length t 2.24 −2.99 −5.47 −6.77 −8.58 −12.66 −10.25 −11.98 −9.12 −8.63 −7.36 −7.73 −6.47 df 18 18 18 18 18 18 18 18 18 18 18 18 18 p 0.03 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 宽度 Width t 4.43 −6.17 −5.59 −4.54 −3.46 −1.79 0.49 2.90 2.77 3.72 3.78 4.83 1.15 df 18 18 18 18 18 18 18 18 18 18 18 18 18 p <0.01 <0.01 <0.01 <0.01 <0.01 0.09 0.63 0.01 0.01 <0.01 <0.01 <0.01 0.27 -
在雌蜂和雄蜂触角上,共发现刺形感器、毛形感器、锥形感器、栓锥形感器、板形感器、腔锥形感器和鳞形感器这7类感受器。雌蜂和雄蜂触角的柄节和梗节上分布的感受器类型较少,绝大多数感受器均分布在鞭节上。
-
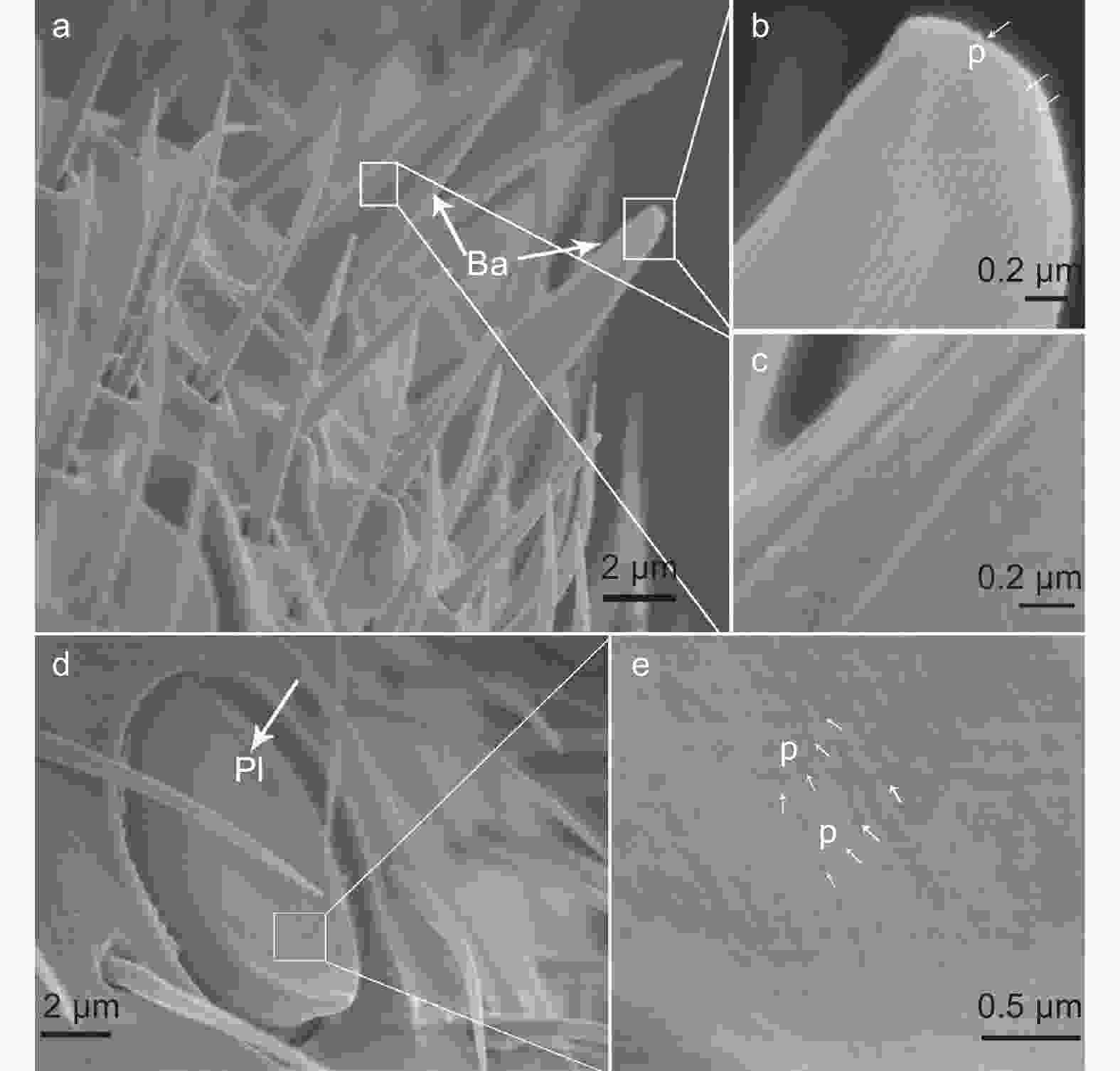
刺形感器是肿腿蜂触角上最常见的一类感器,有2种亚型(Ch1和Ch2)。Ch1密布在触角各节上,呈细长刚毛状,基部着生于触角表皮下陷的基窝内,基部往上1 μm处表面具棱状纹路直至端部,端部尖锐(图3abc)。Ch2较短,基部着生于触角表皮下陷的基窝内,多垂直于触角表面,表面光滑无孔,端部圆钝(图3d)。Ch1和Ch2在雌蜂和雄蜂触角上均有分布,长度在不同性别个体间无显著差异(Ch1:t=−1.608,df=78,P=0.112;Ch2:t=0.477,df=38,P=0.636)(表2)。以单根触角计,Ch1在雌蜂触角上的数量显著多于雄蜂触角(t=47.052,df=18,P<0.001),Ch2的数量在雌雄蜂触角间无显著差异(t=0.728,df=18,P=0.476)(表2)。
表 2 白蜡吉丁肿腿蜂触角上各类感器的长度和数量
Table 2. Length and number of sensilla on the antennae of Sclerodermus pupariae
感器类型
Sensilla type感器长度
Sensilla length/μm感器数量
Sensilla number/n♀ ♂ ♀ ♂ Ch1 13.24 ± 0.73 a 14.66 ± 0.49 a 969.50 ± 11.84 a 388.00 ± 3.56 b Ch2 4.54 ± 0.55 a 4.19 ± 0.48 a 7.20 ± 0.39 a 6.80 ± 0.39 a Tr1 9.50 ± 0.30 — 7.00 ± 0.00 — Tr2 9.64 ± 0.40 — 40.60 ± 0.70 — Tr3 — 16.76 ± 0.74 — 283.10 ± 6.88 Ba 9.37 ± 0.22 — 17.00 ± 0.00 / Pl 9.65 ± 0.24 a 10.16 ± 0.15 a 22.00 ± 0.00 25.00 ± 0.00 St1 12.52 ± 0.25 a 12.33 ± 0.22 a 9.00 ± 0.00 4.00 ± 0.00 St2 6.54 ± 0.21 a 6.81 ± 0.19 a 3.00 ± 0.00 7.00 ± 0.00 Co 0.67 ± 0.03 a 0.67 ± 0.03 a 4.00 ± 0.00 4.00 ± 0.00 Sq 0.88 ± 0.04 a 0.91 ± 0.09 a 97.00 ± 1.10 a 97.40 ± 1.08 a 注:感器长度中,腔锥形感器(Co)所示的数值为其顶端宽度;感器数量均为每根触角上的计数;表中值为均值 ± 标准误;同一行的不同小写字母表示该感器的长度或数量在雌雄蜂触角上存在显著差异;“—”表示无该类型的感受器
Notes: Values of sensilla coeloconica indicate the width of their tips; the numbers of each type of sensillum are presented as one antenna; values in the table are mean ± SEM; different letters in a column mean significant difference at α =0.05; ‘—’ means without this type of sensilla; -
毛形感器呈细长毛发状,有3种亚型(Tr1、Tr2和Tr3)。Tr1垂直于触角表面,基部着生处触角表皮微隆起,无明显凹陷窝,感器表面光滑但密布小孔,端部渐尖(图4ab)。Tr2垂直于触角表面,基部着生处有明显的凹陷窝,感器表面具纵向沟状纹路,同时也具小孔,端部圆钝(图4cde)。Tr1和Tr2仅在雌蜂触角上发现,平均长度分别为9.50 ± 0.30 μm(n=40)和9.64 ± 0.40 μm(n=30)(表2)。Tr1数量保守,每根触角上均为7个;Tr2平均数量为40.60 ± 0.70(表2)。Tr3尖端指向触角末段,基部着生处的触角表皮微隆起呈一圆圈状,无明显凹陷窝,感器表面光滑但密布小孔,端部渐尖(图4fg)。Tr3与Tr1外部形态较类似,但前者更为细长,且仅在雄蜂触角上发现,平均长度为16.76 ± 0.74 μm,每根触角上平均数量为283.10 ± 6.88(表2)。
-
锥形感器呈拇指状,较毛形感器粗短,垂直于触角表面,基部着生处无明显凹陷窝。感器表面具密集纵向沟状纹路,中下部为圆柱状,中部至顶端逐渐扁平,端部有小孔(图5abc)。锥形感器仅分布于雌蜂触角上,平均长度为9.37 ± 0.22 μm(n=30),每根触角上的数量均为17个(表2)。
-
板形感器为瓣状结构,近椭圆形,紧贴触角,着生于触角表皮凹陷的小槽内。感器基部圆钝,端部渐尖且明显高于触角表皮,端部朝向于触角末端。感器表面密布小孔(图5de)。板形感器在雌蜂和雄蜂触角上均有分布,长度无显著差异(t=−1.792,df=58,P=0.078)(表2)。雌蜂每根触角上数量为22个,雄蜂上为25个(表2)。
-
栓锥形感器有2种亚型(St1和St2)。St1着生在凹陷入触角表皮的哑铃状小室内,呈手指状,从基部至端部的感器表面均有纵向沟状纹路,端部圆钝(图6a)。St2着生在凹陷入触角表皮的卵圆形小室内,亦呈手指状,但长度较St1更短(表2)。St2端部明显膨大,膨大的端部表面有纵向沟状纹路,其余部分表面光滑(图6b)。St1和St2的端部均朝向于触角末端。St1和St2在雌蜂和雄蜂触角上均有分布,长度在雌雄蜂间无显著差异(St1:t=0.586,df=38,P=0.561;St2:t=-0.959,df=38,P=0.344)(表2)。St1和St2在雌蜂每根触角上的数量分别为9个和3个,雄蜂上分别为4个和7个(表2)。
-
腔锥形感器着生于凹陷入触角表皮的椭圆形槽内,呈图钉状。感器基部纵向伸入触角表皮,基部着生处可见明显未闭合的基窝。感器端部呈胡桃状,有明显的褶皱,具小孔(图7a)。腔锥形感器在雌蜂和雄蜂触角上均有分布,其顶端宽度无显著差异(t=0.204,df=28,P=0.840),且在雌雄蜂触角上数量相近,每根触角均为4个(表2)。
-
鳞形感器为呈鳞片状的触角表皮端部的一刺形突起,分布在鞭节各亚节的节间处。感器细短,光滑无孔,紧贴触角表面(图7b)。鳞形感器在雌蜂和雄蜂触角上均有分布,其长度无显著差异(t=−0.302,df=38,P=0.764),数量亦无显著差异(t=−0.260,df=18,P=0.798)(表2)。
-
雌性白蜡吉丁肿腿蜂每根触角上各类感受器的总数平均为1 176.30个,显著多于雄蜂的815.30个(t=24.130,df=18,P<0.001)。柄节和梗节上分布的感受器类型较少,绝大部分感受器分布在鞭节(表3)。刺形感器是数量最多的一类感器,其中Ch1在雌雄蜂触角的13个亚节均有分布,Ch2仅分布在梗节。毛形感器Tr1和Tr2特异性分布在雌蜂触角上,其中在鞭小节F6-11有Tr1分布,鞭小节F2-11有Tr2分布。毛形感器Tr3特异性分布在雄蜂触角上,除柄节无该感器外,其余各节均有分布。锥形感器Ba特异性分布在雌蜂触角上,鞭小节F5-11有该感器分布。板形感器Pl在雌蜂触角上可见于鞭小节F4-11,在雄蜂触角上可见于鞭小节F3-11。栓锥形感器虽在雌雄蜂触角上均有分布,但其分布特征差异很大。雌蜂触角鞭小节F5-11可见栓锥形感器St1分布,但该感器在雄蜂触角上仅见于鞭小节F4-7。雄蜂触角鞭小节F4-10均有栓锥形感器St2分布,但该感器在雌蜂触角上仅分布于鞭小节F6、F8和F10。腔锥形感器Co和鳞形感器Sq在雌雄蜂触角上的分布特征一致,前者均仅见于鞭小节F7-10,后者可见于鞭小节F2-11。以上各类感器,包括毛形感器Tr1、锥形感器Ba、板形感器Pl、栓锥形感器St1和St2、腔锥形感器Co,它们各自在触角各节上的数量表现为同一性别的不同个体间均一致(表3)。
表 3 各类感器在雌蜂和雄蜂触角各节的分布和数量
Table 3. Distributions of each type of sensilla on the antennae of female and male wasps
感器类型
Sensilla
type性别
Sex柄节
Scape梗节
Pedicel鞭节
FlagellumF1 F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 Ch1 ♀ 50.90 ± 1.55 24.70 ± 1.61 23.90 ± 1.57 26.10 ± 1.21 37.30 ± 1.90 57.80 ± 2.18 72.50 ± 3.00 79.20 ± 1.79 103.60 ± 2.58 105.10 ± 2.88 101.30 ± 3.27 116.20 ± 2.31 170.90 ± 3.60 ♂ 39.60 ± 1.44 15.50 ± 0.76 11.20 ± 0.63 15.10 ± 0.88 17.20 ± 0.68 21.10 ± 1.05 23.40 ± 1.80 27.80 ± 1.34 38.40 ± 1.13 42.80 ± 1.47 45.30 ± 1.26 40.10 ± 0.64 50.50 ± 1.74 Ch2 ♀ — 7.20 ± 0.39 — — — — — — — — — — — ♂ — 6.80 ± 0.39 — — — — — — — — — — — Tr1 ♀ — — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 2.00 ± 0.00 ♂ — — — — — — — — — — — — — Tr2 ♀ — — — 1.10 ± 0.28 1.00 ± 0.00 2.30 ± 0.26 3.10 ± 0.23 4.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 5.60 ± 0.27 11.50 ± 0.27 ♂ — — — — — — — — — — — — — Tr3 ♀ — — — — — — — — — — — — — ♂ — 6.40 ± 0.45 7.10 ± 0.67 12.10 ± 1.03 20.50 ± 1.07 20.30 ± 1.03 22.80 ± 1.95 26.40 ± 1.77 27.60 ± 2.91 27.80 ± 1.99 31.30 ± 1.83 28.20 ± 1.62 52.60 ± 1.90 Ba ♀ — — — — — — 1.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 6.00 ± 0.00 ♂ — — — — — — — — — — — — — Pl ♀ — — — — — 1.00 ± 0.00 1.00 ± 0.00 2.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 ♂ — — — — 1.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 St1 ♀ — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 3.00 ± 0.00 ♂ — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — — — — St2 ♀ — — — — — — — 1.00 ± 0.00 — 1.00 ± 0.00 — 1.00 ± 0.00 — ♂ — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — Co ♀ — — — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — ♂ — — — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — Sq ♀ — — — 4.30 ± 0.21 8.80 ± 0.39 8.80 ± 0.42 10.40 ± 0.48 10.60 ± 0.40 10.40 ± 0.40 11.00 ± 0.42 10.80 ± 0.44 11.00 ± 0.36 10.90 ± 0.28 ♂ — — — 4.70 ± 0.15 8.60 ± 0.43 10.10 ± 0.38 10.90 ± 0.41 10.10 ± 0.38 11.00 ± 0.36 10.00 ± 0.44 10.40 ± 0.30 10.70 ± 0.34 10.90 ± 0.23 注:“—”表示无该类型感器分布; 表中值为均值 ± 标准误
Notes: ‘—’ means without this type of sensilla; Values in the table are mean ± SEM -
本研究通过扫描电子显微镜观测了白蜡吉丁肿腿蜂触角上感受器的形态结构,发现雌蜂和雄蜂的触角均由柄节、梗节和11个鞭小节组成,雄蜂触角较雌蜂更长。共发现7类11种感受器,包括2种刺形感器、3种毛形感器、1种锥形感器、1种板形感器、2种栓锥形感器、1种腔锥形感器和1种鳞形感器。本研究结果与前期发表的管氏肿腿蜂和松褐天牛肿腿蜂等近缘种触角感受器的组成和分布规律存在较大差异。田慎鹏和徐志强[22]首次对管氏肿腿蜂的触角超微结构进行了研究,提出该蜂触角共14节,包括柄节、梗节和12个鞭小节。我们通过核对该论文附图后发现,作者将管氏肿腿蜂触角与头部相连的支角突(antennifer)鉴定成了柄节,由此得出了触角共14节且柄节最短的结论。同时,该文提出仅发现1种栓锥形感器(同本研究栓锥形感器II型,St2),未发现本研究描述的栓锥形感器I型(St1)。2010年后发表的管氏肿腿蜂和松褐天牛肿腿蜂触角感受器结构的相关结果中,均认为它们的触角由柄节、梗节和11个鞭小节组成[17-18],本研究的结论与其一致。本研究发现白蜡吉丁肿腿蜂触角上的感受器类型与上述两项研究结果基本一致,但一些感器的分布规律存差异。与白蜡吉丁肿腿蜂栓锥形感器I和II型(St1和St2)形态一致的感受器在管氏肿腿蜂[17]和松褐天牛肿腿蜂[18]触角上也均有发现。针对该类感器,管氏肿腿蜂雌蜂触角观察到St1和St2,但雄蜂触角仅有St2分布;松褐天牛肿腿蜂雌蜂触角仅有St1,雄蜂触角仅有St2。本研究中发现,白蜡吉丁肿腿蜂雌蜂和雄蜂触角上均有St1和St2分布。另外,腔锥形感器、锥形感器、板形感器在白蜡吉丁肿腿蜂触角各节上的分布规律与管氏肿腿蜂和松褐天牛肿腿蜂的结果也存在一定的差异。可知即使亲缘关系很近,生物学习性、寄主范围相似的昆虫,但是嗅觉器官的超微结构特征也存在差异。同时,这些器官的显微结构及其分布特征的差异,也可用于近缘种的鉴定。
刺形感器是白蜡吉丁肿腿蜂触角上数量最多的一类。刺形感器I型(Ch1)在雌蜂和雄蜂触角的各亚节均有分布,刺形感器II型(Ch2)仅在它们触角的梗节分布。刺形感器一般被认为是一类机械感觉毛,通常无孔[12]。Ch1作为一种机械感觉毛,其长度在所有感受器中最长,且雌蜂触角上的数量显著地多于雄蜂,可能与雌蜂在寄主搜索时,特别是一些依靠寄主振动信号来探测寄主位置的行为功能相关。Ch2垂直着生于梗节,该类型的感受器在松褐天牛肿腿蜂和白蛾周氏啮小蜂Chouioia cunea中被称为Böhm氏鬃毛[18, 23],但后者在广义上也是一种刺形感器,最早在鳞翅目昆虫触角上被定义[14]。这类感受器在很多昆虫触角上主要分布于头部与触角相连接的支角突和梗节,可能与昆虫感受重力和感受触角相对位置有关[12, 24]。
板形感器、栓锥形感器和腔锥形感器在白蜡吉丁肿腿蜂雌蜂和雄蜂触角上均有分布,且它们各自的数量在同性的不同个体上一致。板形感器是膜翅目昆虫触角上很常见,且数量较大的一类感受器。在一些茧蜂、小蜂等寄生蜂触角上的板形感器多紧贴触角纵轴,在触角表面呈一长条状隆起[25-31]。本研究发现白蜡吉丁肿腿蜂雌蜂每根雌蜂触角上有板型感器22个,雄蜂触角上有25个,形状为马蹄形瓣状,与已报道的寄生蜂触角上的板形感器形状差异较大。板形感器表面一般都具有数量庞大的微孔,是一种典型的化学感受器。对中红侧沟茧蜂(Microplitis mediator)离子型受体MmIR8a免疫定位发现其在板形感器内神经元树突上大量表达,也证实该类感受器具备嗅觉编码功能[32]。本研究中,肿腿蜂板形感器同样有大量的微孔,推测其是一种重要的化学感受器。栓锥形感器在寄生蜂触角上并不常见,前期报道白蜡吉丁柄腹茧蜂(Spathius agrili)[28]和黑卵蜂(Gryon gallardoi)[33]触角上存在栓锥形感器。但是,我们发现上述两项研究中所描述的栓锥形感器,其外形均为图钉状,与多数寄生蜂的腔锥形感器更类似。白蜡吉丁肿腿蜂栓锥形感器的2个亚型(St1和St2),均着生在凹陷入触角表皮的小室内,形状呈手指状。除了前文提到的管氏肿腿蜂、松褐天牛肿腿蜂和川硬皮肿腿蜂之外,在其他寄生蜂触角上尚未报道过与此形态类似的感受器。白蜡吉丁肿腿蜂栓锥形感器表面分布有纵向沟状纹,但是并未发现有明显的微孔。Li et al.[17]在管氏肿腿蜂研究中提出2种栓锥形感器的沟状纹内有微孔,是一种化学感受器,但其提供的图片并没有清晰地展示出微孔结构,仅能从感器切片看到内腔有神经元树突。因此,依据当前结果,我们尚不能确定白蜡吉丁肿腿蜂栓锥形感器是否为化学感器。白蜡吉丁肿腿蜂腔锥形感器数量最少,在雌蜂和雄蜂触角上均为4个。对黑腹果蝇(Drosophila melanogaster)腔锥形感器功能研究中发现其对很多酸和氨类物质有强烈的电生理反应,并进一步证实有15个离子型受体DmIRs表达在其中,发挥着重要的嗅觉功能[8, 34]。白蜡吉丁肿腿蜂腔锥形感器的胡桃状末端分布有众多微孔,也应该是一类化学感受器。
毛形感器和锥形感器在白蜡吉丁肿腿蜂雌蜂和雄蜂触角上存在明显的性二型特征。毛形感器有3个亚型(Tr1、Tr2和Tr3),Tr1和Tr2仅存在于雌蜂触角,Tr3仅存在于雄蜂触角。这3种毛形感器中,Tr1和Tr3外部形态较一致,表面光滑具微孔,但Tr3较Tr1更细长;Tr2表面有纵向纹路,存在不明显的微孔。Li et al.[17]对管氏肿腿蜂触角感器的研究中,未描述Tr1。本研究在Tr1表面发现很多微孔,认为其是一类嗅觉感受器。黑腹果蝇毛形感器是重要的嗅觉感受器,内腔有丰富的嗅觉神经元,神经元膜上表达了大量的气味受体,其中编码性信息素(cis-vaccenyl acetate,cVa)的气味受体DmOr67d就表达在毛形感器I型内[35]。雄性白蜡吉丁肿腿蜂触角上数量最多的化学感受器为Tr3,除柄节无分布外,其余各节均有分布,推测其可能在雄蜂寻找配偶的过程中发挥重要作用。因Tr3在雄蜂触角上分布广泛,且常与刺形感器Ch1混生在一起,延伸方向也一致,容易被误认为是刺形感器。两者的核心区别是Tr3着生处基窝闭合,Ch1着生处有明显的未闭合基窝;微观结构上Tr3表面光滑有孔,Ch1表面具棱无孔。白蜡吉丁肿腿蜂触角锥形感器(Ba)特异性分布于雌蜂触角上,表面具规则的纵向纹路,顶端有孔。该类锥形感器顶端的微孔在管氏肿腿蜂前期研究中有清晰的展示,感器末端腹面可见40余个极孔[17],遗憾的是本研究中因触角粘贴角度原因,未能清晰地拍摄到该感器末端的腹面部分。锥形感器在很多昆虫中是一类典型的化学感受器[5-6, 13]。白蜡吉丁肿腿蜂锥形感器的顶端具多个微孔,我们推测其也可能同时具备嗅觉和味觉识别功能。白蜡吉丁肿腿蜂雌蜂和雄蜂存在一些典型的行为习性差异,如雌蜂极易形成聚集体,有强烈的聚集行为,而雄蜂从不会产生聚集态(作者个人观察)。这些相关的行为差异是否与差异分布的触角感受器相关,未来对这些存在性二型特征的触角感器的化学识别功能的进一步研究,也许可以让我们更好地了解雌雄个体化学感器的分化及其与生物学差异之间的联系。
前期研究在白蜡吉丁柄腹茧蜂触角上发现3类4种感受器,白蜡吉丁啮小蜂(Tetrastichus planipennisi)触角上发现5类8种感受器[28, 36],均少于本研究在白蜡吉丁肿腿蜂触角上发现的感受器种类。白蜡吉丁肿腿蜂的寄主范围较另外2种寄生蜂更广[37-38]。我们推测,多寄主型寄生蜂中观察到更多样化的感受器类型,可能与其更广的寄主范围和营养学关系间更复杂的相互作用需要更精准的感觉器官来充分支持有关。当然,基于外部形态证据,在多寄主型寄生蜂触角上虽可观察到更丰富的感受器组成,但其功能分型尚需结合感受器内神经元数量、神经元上化学感受基因的表达特征和化学感受谱等后续研究来确定。
-
(1)白蜡吉丁肿腿蜂雌蜂和雄蜂触角均为13节,包括柄节、梗节和11个鞭小节,雄蜂触角显著长于雌蜂。
(2)共发现7类11种感受器,包括2种刺形感器、3种毛形感器、1种锥形感器、1种板形感器、2种栓锥形感器、1种腔锥形感器和1种鳞形感器。其中,毛形感器、锥形感器、板形感器和腔锥形感器是典型的化学感受器。
(3)雌蜂触角上有10种感受器,其中毛形感器I型和II型、锥形感器为其特有。雄蜂触角上有8种感受器,其中毛形感器III型为其特有。雌雄蜂触角感受器的类型、分布规律和数量,存在性二型现象。
白蜡吉丁肿腿蜂触角感受器的超微结构
Ultrastructure of the Antennal Sensilla of Parasitic Wasp, Sclerodermus pupariae (Hymenoptera: Bethylidae)
-
摘要:
目的 触角是昆虫最重要的嗅觉器官,触角感受器是昆虫嗅觉识别的最小形态学单元。为了明确多种林木蛀干类吉丁虫和天牛的重要天敌白蜡吉丁肿腿蜂的触角感受器种类和形态。 方法 利用扫描电子显微镜对雌雄蜂触角及其感受器超微结构进行了观察,并进一步分析了各类感受器在不同性别成蜂触角上的分布规律和数量。 结果 白蜡吉丁肿腿蜂触角呈膝状,雌雄蜂触角均为13节,包括柄节、梗节和11个鞭小节,雄蜂触角显著长于雌蜂。在雌雄蜂触角上共发现7类11种感受器,包括2种刺形感器、3种毛形感器、1种锥形感器、1种板形感器、2种栓锥形感器、1种腔锥形感器和1种鳞形感器。雌蜂触角上存在10种感受器,其中毛形感器I型和II型、锥形感器为其特有。雄蜂触角上存在8种感受器,其中毛形感器III型为其特有。毛形感器I型、锥形感器、板形感器、栓锥形感器(I和II型)和腔锥形感器各自数量在同一性别的不同个体间一致,而其余类型感受器的数量在不同个体间不等。在雌雄蜂共有的感器类型中,刺形感器I型、栓锥形感器I型在雌蜂触角上的数量较雄蜂更多;而板形感器、栓锥形感器II型在雄蜂触角上的数量较雌蜂更多;腔锥形感器的数量在雌雄蜂之间相同。毛形感器、锥形感器、板形感器和腔锥形感器表面有微孔,是典型的化学感器。 结论 本研究解析了白蜡吉丁肿腿蜂触角感受器的超微结构,发现雌雄蜂触角感受器的种类、数量和分布特征均存在典型的性二型现象,这些结果为后续研究该寄生蜂的化学通讯机制和雌雄蜂的嗅觉行为差异奠定了基础。 Abstract:Objective Antennae are the most important olfactory organs of insects, and the antennal sensilla are the fundamental morphological units in their olfactory recognitions. To understand the variety and morphology of the antennal sensilla of Sclerodermus pupariae (Hymenoptera: Bethylidae), an important natural enemy of many buprestids and longhorned beetles. Method The ultrastructures of antennae and sensilla of male and female parasitoid adults were investigated by scanning electron microscope, and the distribution and quantity of sensilla on female and male antennae were analyzed. Result The antennae of both male and female parasitoids were knee shaped, and they were consistently constituted with scape, pedicel, and 11 flagellomeres. The antennae of male parasitoids were much longer than those of female wasps. There were seven categories with 11 types of sensilla on the antennae of S. pupariae, including two types of sensilla chaetica (Ch1 and Ch2), three types of sensilla trichodea (Tr1, Tr2, and Tr3), one type of sensilla basiconica (Ba), one type of sensilla placodea (Pl), two types of sensilla styloconica (St1 and St2), one type of sensilla coeloconica (Co), and one type of sensilla squamiforma (Sq). Ten types of sensilla were observed on the antennae of female, and the sensilla Tr1, Tr2, and Ba were restrictedly distributed on female′s antennae. There were eight types of sensilla on the antennae of male, and the sensilla Tr3 were only observed in males. There were six types of sensilla showed consistent quantities across individuals of the same sex, including sensilla Tr1, Ba, Pl, St1, St2, and Co. For other five types of sensilla, their numbers varied among individuals in both females and males. The number of sensilla Ch1 and St1 on the antennae of female adults was significantly higher than that on male adults. Nevertheless, the number of sensilla Pl and St2 on the antennae of male adults was significantly higher than that on female adults. The number of sensilla Co was the same between female and male parasitoids. The sensilla Tr1, Tr2, Tr3, Ba, Pl, and Co were six typical chemosensilla because multiple wall pores could be found on their surfaces. Conclusion We analyzed the ultrastructure of antennal sensilla of S. pupariae, and found that the types, quantities, and distributions of antennal sensilla between female and male parasitoids have typical sexual dimorphism, which will provide fundamental data for the future investigations on the chemoreception mechanism and olfactory behavior differences between the male and female parasitoids. -
Key words:
- antenna
- / sensillum
- / scanning electron microscope
- / parasitic wasp
-
表 1 雌雄蜂触角各节长和宽的差异检验统计值
Table 1. Difference test parameters of antennal size between female and male parasitoids
变量
Variable统计量
Statistical parameter触角部位
Antennal segmentsSc Pe F1 F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 长度 Length t 2.24 −2.99 −5.47 −6.77 −8.58 −12.66 −10.25 −11.98 −9.12 −8.63 −7.36 −7.73 −6.47 df 18 18 18 18 18 18 18 18 18 18 18 18 18 p 0.03 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 宽度 Width t 4.43 −6.17 −5.59 −4.54 −3.46 −1.79 0.49 2.90 2.77 3.72 3.78 4.83 1.15 df 18 18 18 18 18 18 18 18 18 18 18 18 18 p <0.01 <0.01 <0.01 <0.01 <0.01 0.09 0.63 0.01 0.01 <0.01 <0.01 <0.01 0.27 表 2 白蜡吉丁肿腿蜂触角上各类感器的长度和数量
Table 2. Length and number of sensilla on the antennae of Sclerodermus pupariae
感器类型
Sensilla type感器长度
Sensilla length/μm感器数量
Sensilla number/n♀ ♂ ♀ ♂ Ch1 13.24 ± 0.73 a 14.66 ± 0.49 a 969.50 ± 11.84 a 388.00 ± 3.56 b Ch2 4.54 ± 0.55 a 4.19 ± 0.48 a 7.20 ± 0.39 a 6.80 ± 0.39 a Tr1 9.50 ± 0.30 — 7.00 ± 0.00 — Tr2 9.64 ± 0.40 — 40.60 ± 0.70 — Tr3 — 16.76 ± 0.74 — 283.10 ± 6.88 Ba 9.37 ± 0.22 — 17.00 ± 0.00 / Pl 9.65 ± 0.24 a 10.16 ± 0.15 a 22.00 ± 0.00 25.00 ± 0.00 St1 12.52 ± 0.25 a 12.33 ± 0.22 a 9.00 ± 0.00 4.00 ± 0.00 St2 6.54 ± 0.21 a 6.81 ± 0.19 a 3.00 ± 0.00 7.00 ± 0.00 Co 0.67 ± 0.03 a 0.67 ± 0.03 a 4.00 ± 0.00 4.00 ± 0.00 Sq 0.88 ± 0.04 a 0.91 ± 0.09 a 97.00 ± 1.10 a 97.40 ± 1.08 a 注:感器长度中,腔锥形感器(Co)所示的数值为其顶端宽度;感器数量均为每根触角上的计数;表中值为均值 ± 标准误;同一行的不同小写字母表示该感器的长度或数量在雌雄蜂触角上存在显著差异;“—”表示无该类型的感受器
Notes: Values of sensilla coeloconica indicate the width of their tips; the numbers of each type of sensillum are presented as one antenna; values in the table are mean ± SEM; different letters in a column mean significant difference at α =0.05; ‘—’ means without this type of sensilla;表 3 各类感器在雌蜂和雄蜂触角各节的分布和数量
Table 3. Distributions of each type of sensilla on the antennae of female and male wasps
感器类型
Sensilla
type性别
Sex柄节
Scape梗节
Pedicel鞭节
FlagellumF1 F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 Ch1 ♀ 50.90 ± 1.55 24.70 ± 1.61 23.90 ± 1.57 26.10 ± 1.21 37.30 ± 1.90 57.80 ± 2.18 72.50 ± 3.00 79.20 ± 1.79 103.60 ± 2.58 105.10 ± 2.88 101.30 ± 3.27 116.20 ± 2.31 170.90 ± 3.60 ♂ 39.60 ± 1.44 15.50 ± 0.76 11.20 ± 0.63 15.10 ± 0.88 17.20 ± 0.68 21.10 ± 1.05 23.40 ± 1.80 27.80 ± 1.34 38.40 ± 1.13 42.80 ± 1.47 45.30 ± 1.26 40.10 ± 0.64 50.50 ± 1.74 Ch2 ♀ — 7.20 ± 0.39 — — — — — — — — — — — ♂ — 6.80 ± 0.39 — — — — — — — — — — — Tr1 ♀ — — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 2.00 ± 0.00 ♂ — — — — — — — — — — — — — Tr2 ♀ — — — 1.10 ± 0.28 1.00 ± 0.00 2.30 ± 0.26 3.10 ± 0.23 4.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 5.60 ± 0.27 11.50 ± 0.27 ♂ — — — — — — — — — — — — — Tr3 ♀ — — — — — — — — — — — — — ♂ — 6.40 ± 0.45 7.10 ± 0.67 12.10 ± 1.03 20.50 ± 1.07 20.30 ± 1.03 22.80 ± 1.95 26.40 ± 1.77 27.60 ± 2.91 27.80 ± 1.99 31.30 ± 1.83 28.20 ± 1.62 52.60 ± 1.90 Ba ♀ — — — — — — 1.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 2.00 ± 0.00 6.00 ± 0.00 ♂ — — — — — — — — — — — — — Pl ♀ — — — — — 1.00 ± 0.00 1.00 ± 0.00 2.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 4.00 ± 0.00 ♂ — — — — 1.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 3.00 ± 0.00 St1 ♀ — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 3.00 ± 0.00 ♂ — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — — — — St2 ♀ — — — — — — — 1.00 ± 0.00 — 1.00 ± 0.00 — 1.00 ± 0.00 — ♂ — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — Co ♀ — — — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — ♂ — — — — — — — — 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 1.00 ± 0.00 — Sq ♀ — — — 4.30 ± 0.21 8.80 ± 0.39 8.80 ± 0.42 10.40 ± 0.48 10.60 ± 0.40 10.40 ± 0.40 11.00 ± 0.42 10.80 ± 0.44 11.00 ± 0.36 10.90 ± 0.28 ♂ — — — 4.70 ± 0.15 8.60 ± 0.43 10.10 ± 0.38 10.90 ± 0.41 10.10 ± 0.38 11.00 ± 0.36 10.00 ± 0.44 10.40 ± 0.30 10.70 ± 0.34 10.90 ± 0.23 注:“—”表示无该类型感器分布; 表中值为均值 ± 标准误
Notes: ‘—’ means without this type of sensilla; Values in the table are mean ± SEM -
[1] VOSSHALL L B, STOCKER R F. Molecular architecture of smell and taste in Drosophila[J]. Annual Review of Neuroscience, 2007, 30: 505-533. doi: 10.1146/annurev.neuro.30.051606.094306 [2] SUH E, BOHBOT J D, ZWIEBEL L. Peripheral olfactory signaling in insects[J]. Current Opinion in Insect Science, 2014, 6: 86-92. doi: 10.1016/j.cois.2014.10.006 [3] FLEISCHER J, PREGITZER P, BREER H, et al. Access to the odor world: olfactory receptors and their role for signal transduction in insects[J]. Cellular and Molecular Life Sciences, 2018, 75(3): 485-508. [4] VOSSHALL L B, WONG A M, AXEL R. An olfactory sensory map in the fly brain[J]. Cell, 2000, 102(2): 147-159. doi: 10.1016/S0092-8674(00)00021-0 [5] HALLEM E A, HO M G, CARLSON J R. The molecular basis of odor coding in the Drosophila antenna[J]. Cell, 2004, 117(7): 965-979. doi: 10.1016/j.cell.2004.05.012 [6] HALLEM E A, CARLSON J R. Coding of odors by a receptor repertoire[J]. Cell, 2006, 125(1): 143-160. doi: 10.1016/j.cell.2006.01.050 [7] JONES W D, CAYIRLIOGLU P, KADOW I G, et al. Two chemosensory receptors together mediate carbon dioxide detection in Drosophila[J]. Nature, 2007, 445(7123): 86-90. doi: 10.1038/nature05466 [8] BENTON R, VANNICE K S, GOMEZ-DIAZ C, et al. Variant ionotropic glutamate receptors as chemosensory receptors in Drosophila[J]. Cell, 2009, 136(1): 149-162. doi: 10.1016/j.cell.2008.12.001 [9] PASK G M, SLONE J D, MILLAR J G, et al. Specialized odorant receptors in social insects that detect cuticular hydrocarbon cues and candidate pheromones[J]. Nature Communications, 2017, 8: 297. doi: 10.1038/s41467-017-00099-1 [10] SLONE J D, PASK G M, FERGUSON S T, et al. Functional characterization of odorant receptors in the ponerine ant, Harpegnathos saltator[J]. Proceedings of the National Academy of Sciences of the United States of America, 2017, 114(32): 8586-8591. doi: 10.1073/pnas.1704647114 [11] 白鹏华, 王 冰, 张仙红, 等. 昆虫气味受体的研究方法与进展[J]. 昆虫学报, 2022, 65(3):364-385. doi: 10.16380/j.kcxb.2022.03.012 [12] 马瑞燕, 杜家纬. 昆虫的触角感器[J]. 昆虫知识, 2000, 37(3):179-183. [13] 王桂荣, 郭予元, 吴孔明. 棉铃虫触角感器的超微结构观察[J]. 中国农业科学, 2002, 35(12):1479-1482+1584-1586. [14] SCHNEIDER D. Insect antennae[J]. Annual Review of Entomology, 1964, 9: 103-122. doi: 10.1146/annurev.en.09.010164.000535 [15] 杨忠岐, 王小艺, 曹亮明, 等. 管氏肿腿蜂的再描述及中国硬皮肿腿蜂属Sclerodermus (Hymenoptera: Bethylidae) 的种类[J]. 中国生物防治学报, 2014, 30(1):1-12. [16] 胡 霞, 周祖基, 蒋学建, 等. 川硬皮肿腿蜂雌蜂触角超微结构观察[J]. 辽宁林业科技, 2006, 33(6):4-7. [17] LI X R, LU D G, LIU X X, et al. Ultrastructural characterization of olfactory sensilla and immunolocalization of odorant binding and chemosensory proteins from an ectoparasitoid Scleroderma guani (Hymenoptera: Bethylidae)[J]. International Journal of Biological Sciences, 2011, 7(6): 848-868. doi: 10.7150/ijbs.7.848 [18] ZHOU C X, SUN X, MI F, et al. Antennal sensilla in the parasitoid Sclerodermus sp. (Hymenoptera: Bethylidae)[J]. Journal of Insect Science, 2015, 15(36): 1. doi: 10.1093/jisesa/iev024 [19] JIANG Y, YANG Z Q, WANG X Y, et al. Molecular identification of sibling species of Sclerodermus (Hymenoptera: Bethylidae) that parasitize buprestid and cerambycid beetles by using partial sequences of mitochondrial DNA cytochrome oxidase subunit 1 and 28s ribosomal RNA gene[J]. PLoS One, 2015, 10(3): e0119573. doi: 10.1371/journal.pone.0119573 [20] ZACHARUK R Y. Ultrastructure and function of insect chemosensilla[J]. Annual Review of Entomology, 1980, 25: 27-47. doi: 10.1146/annurev.en.25.010180.000331 [21] BLEEKER M A K, SMID H M, VAN AELST A C, et al. Antennal sensilla of two parasitoid wasps: a comparative scanning electron microscopy study[J]. Microscopy Research and Technique, 2004, 63(5): 266-273. doi: 10.1002/jemt.20038 [22] 田慎鹏, 徐志强. 管氏肿腿蜂触角感器的扫描电镜观察[J]. 昆虫知识, 2003, 40(1):59-62. [23] 孙伊阳, 覃东玉, 潘丽娜, 等. 白蛾周氏啮小蜂触角感受器性二型超微结构分析[J]. 林业科学, 2020, 56(10):135-144. doi: 10.11707/j.1001-7488.20201015 [24] 董文霞, 张钟宁. 中红侧沟茧蜂触角感受器的扫描电镜观察[J]. 昆虫学报, 2006, 49(6):1054-1059. doi: 10.3321/j.issn:0454-6296.2006.06.025 [25] ROUX O, VAN BAAREN J, GERS C, et al. Antennal structure and oviposition behavior of the Plutella xylostella specialist parasitoid: Cotesia plutellae[J]. Microscopy Research and Technique, 2005, 68(1): 36-44. doi: 10.1002/jemt.20220 [26] GAO Y, LUO L Z, HAMMOND A. Antennal morphology, structure and sensilla distribution in Microplitis pallidipes (Hymenoptera: Braconidae)[J]. Micron, 2006, 38(6): 684-693. doi: 10.1016/j.micron.2006.09.004 [27] ONAGBOLA E O, FADAMIRO H Y. Scanning electron microscopy studies of antennal sensilla of Pteromalus cerealellae (Hymenoptera: Pteromalidae)[J]. Micron, 2008, 39: 526-535. doi: 10.1016/j.micron.2007.08.001 [28] WANG X Y, YANG Z Q, GOULD J R. Sensilla on the antennae, legs and ovipositor of Spathius agrili Yang (Hymenoptera: Braconidae), a parasitoid of the emerald ash borer Agrilus planipennis Fairmaire (Coleoptera: Buprestidae)[J]. Microscopy Research and Technique, 2010, 73(5): 560-571. [29] DAS P, CHEN L, SHARMA K R, et al. Abundance of antennal chemosensilla in two parasitoid wasps with different degree of host specificity may explain sexual and species differences in their response to host-related volatiles[J]. Microscopy Research and Technique, 2011, 74(10): 900-909. doi: 10.1002/jemt.20974 [30] ZHOU H, WU W J, NIU L M, et al. Antennal sensilla of female Encarsia guadeloupae Viggiani (Hymenoptera: Aphelinidae), a nymphal parasitoid of the spiraling whitefly Aleurodicus dispersus (Hemiptera: Aleyrodidae)[J]. Micron, 2013, 44: 365-372. doi: 10.1016/j.micron.2012.09.001 [31] 刘燕梅, 曾浩源, 王姿烨, 等. 亮腹釉小蜂成虫触角感受器的超微结构[J]. 植物保护学报, 2021, 48(4):822-829. doi: 10.13802/j.cnki.zwbhxb.2021.2020151 [32] WANG S N, PENG Y, LU Z Y, et al. Cloning and expression profile of ionotropic receptors in the parasitoid wasp Microplitis mediator (Hymenoptera: Braconidae)[J]. Journal of Insect Physiology, 2016, 90: 27-35. doi: 10.1016/j.jinsphys.2016.05.002 [33] DA ROCHA L, MOREIRA G R P, REDAELLI L R. Morphology and distribution of antennal sensilla of Gryon gallardoi (Brèthes) (Hymenoptera: Scelionidae) females[J]. Neotropical Entomology, 2007, 36(5): 721-728. doi: 10.1590/S1519-566X2007000500013 [34] YAO C A, IGNELL R, CARLSON J R. Chemosensory coding by neurons in the coeloconic sensilla of the Drosophila antenna[J]. The Journal of Neuroscience, 2005, 25(37): 8359-8367. doi: 10.1523/JNEUROSCI.2432-05.2005 [35] COUTO A, ALENIUS M, DICKSON B J. Molecular, anatomical, and functional organization of the Drosophila olfactory system[J]. Current Biology, 2005, 15(17): 1535-1547. doi: 10.1016/j.cub.2005.07.034 [36] 高 宇, 王志英, 赵红盈, 等. 白蜡吉丁啮小蜂触角感觉器的扫描电镜观察[J]. 中国农业科学, 2013, 46(9):1956-1964. [37] YANG Z Q, WANG X Y, GOULD J R, et al. Host specificity of Spathius agrili Yang (Hymenoptera: Braconidae), an important parasitoid of the emerald ash borer[J]. Biological Control, 2008, 47(2): 216-221. doi: 10.1016/j.biocontrol.2008.08.001 [38] DUAN J J, SCHMUDE J M, WANG X Y, et al. Host utilization, reproductive biology, and development of the larval parasitoid Tetrastichus planipennisi as influenced by temperature: implications for biological control of the emerald ash borer in North America[J]. Biological Control, 2018, 125: 50-56. doi: 10.1016/j.biocontrol.2018.06.009 -




 下载:
下载: