-
重金属土壤污染是引起人类健康和生态系统严重关注的主要环境问题之一[1]。由于重金属对动物和人类具有致癌和致突变作用[2],必须对土壤中的大多数污染物质进行清除,并尽量减少潜在有毒元素进入食物链[3]。土壤中重金属的主要人为来源为采矿业[3]。通常矿物加工会产生大量废弃物,大多数废弃物被认为是有毒或有害的[4]。废弃的金属尾矿地区普遍严重缺乏植被[5],并导致了严峻的环境问题。因此,对废弃尾矿区域进行环境治理是十分紧迫的课题。
筛选合适的植物是能否在废弃尾矿库成功进行植物修复的一个关键因素[6]。与草本植物相比,木本植物特别是速生木本植物具有生物量大、根系发达的特点[7],并在一定程度上有能力富集重金属[8-9]。栓皮栎(Quercus variabilis Bl.)为壳斗科(Fagaceae)栎属(Quercus L.)高大落叶乔木,在我国分布广泛,是重要的乡土树种。研究表明栓皮栎适应性强,具有一定的重金属积累能力[10]。本项目组前期的盆栽试验结果也表明栓皮栎在重金属胁迫下有较好的耐性。目前关于该树种在废弃尾矿区对重金属吸收积累的研究较少。因此,本研究选择栓皮栎为试验材料,探讨其在铅锌尾矿环境下对重金属吸收积累特性的差异,以期筛选出具有土壤修复潜力的植物材料,为尾矿库植被恢复技术的实施提供科学依据。
-
试验地点位于浙江省杭州市富阳区(30°07′42″ N, 119°50′39″ E),属亚热带季风气候。铅锌矿砂样品采集自表层(0~30 cm),其中镉、铅、锌和铜浓度分别为28.5、2380.0、2495.0和117 mg·kg−1;有效磷、水解氮和速效钾浓度分别为1.0、15.9和40.9 mg·kg−1;pH值7.84。通过计算内梅罗综合污染指数来评价铅锌矿砂污染程度,其综合污染指数为21.38,达到重污染水平。
-
栓皮栎种子分别采集自山东泰山(36°12′40″ N, 117°07′13″ E)和徂徕山(36°03′56″ N, 117°14′54″ E),其中包括泰山3个家系(T2、T7和T8)以及徂徕山1个家系(J5)。采种母树生长正常,无病虫害。2014年播种,播种基质为珍珠岩∶泥炭 = 1∶3。2015年5月选取生长一致的各家系1年生容器苗(平均苗高35~40 cm)进行试验。
-
在试验点开挖宽约18 cm、深20 cm种植沟,沟内种植容器苗。株行距为2 m × 2 m。按照随机区组设计,栓皮栎每个家系1行10株,重复3次。2016、2017和2018年春各进行平茬1次。2018年9月对参试材料进行破坏性取样进行生物量、养分和重金属含量分析。
-
试验结束后用卷尺测量每株栓皮栎苗高。用游标卡尺按同一方向测定每株栓皮栎地径。
-
样品收获后,将其分为叶片、茎和根系3部分。根系用去离子水冲洗3次后用5 mmol·L−1 Ca(NO3)2浸泡。所有样品经105℃杀青30 min,75℃烘干3 d后称量。
-
样品烘干粉碎后过筛,称取0.2 g。金属元素浓度根据施翔等[11]方法进行测定。金属元素含量 = 地上部(根系)重金属浓度 × 地上部(根系)生物量。植物重金属富集系数(Bioconcentration factor, BCF)= 植物体内重金属浓度/土壤重金属浓度;重金属转移系数(Translocation factor, TF)= 地上部重金属浓度/根系重金属浓度。植物体内全氮和全磷浓度根据冷华妮等[12]的方法测定。氮(磷)吸收量以每株含量表示。氮、磷利用效率 = 整株生物量/整株氮(磷)吸收量。
-
各指标数据均利用SPSS 26.0软件进行统计分析。采用单因素(One-way ANOVA)和LSD法进行方差分析和多重比较(α = 0.05)。利用OriginPro 2019软件作图。图表中数据为平均值 ± 标准差/标准误。
-
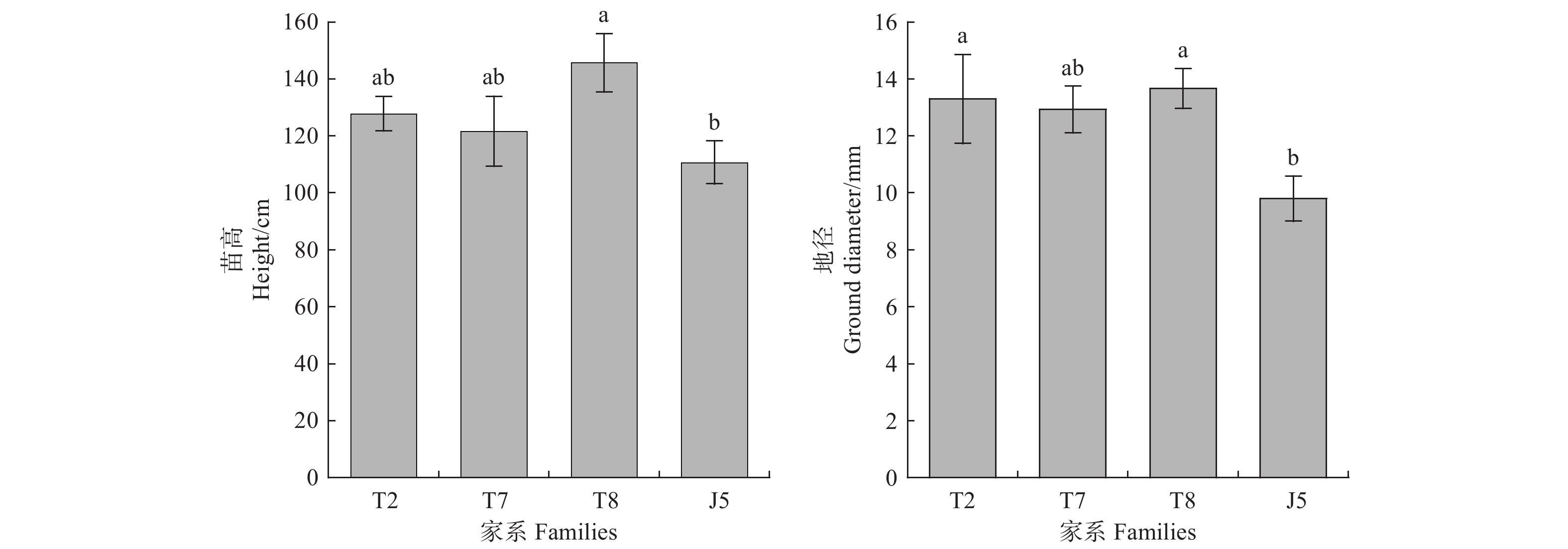
栓皮栎各家系在铅锌矿砂环境中未表现出叶片变黄、萎焉等显著毒害效应。但参试家系苗高表现出一定差异,其中泰山8号家系苗高高于其它参试材料,为145.7 cm;徂徕山5号家系苗高则低于其它家系,仅为110.7 cm(图1)。各参试材料地径在铅锌矿砂中有同样表现。徂徕山5号家系地径仅为9.8 mm,低于其它家系(图1)。
-
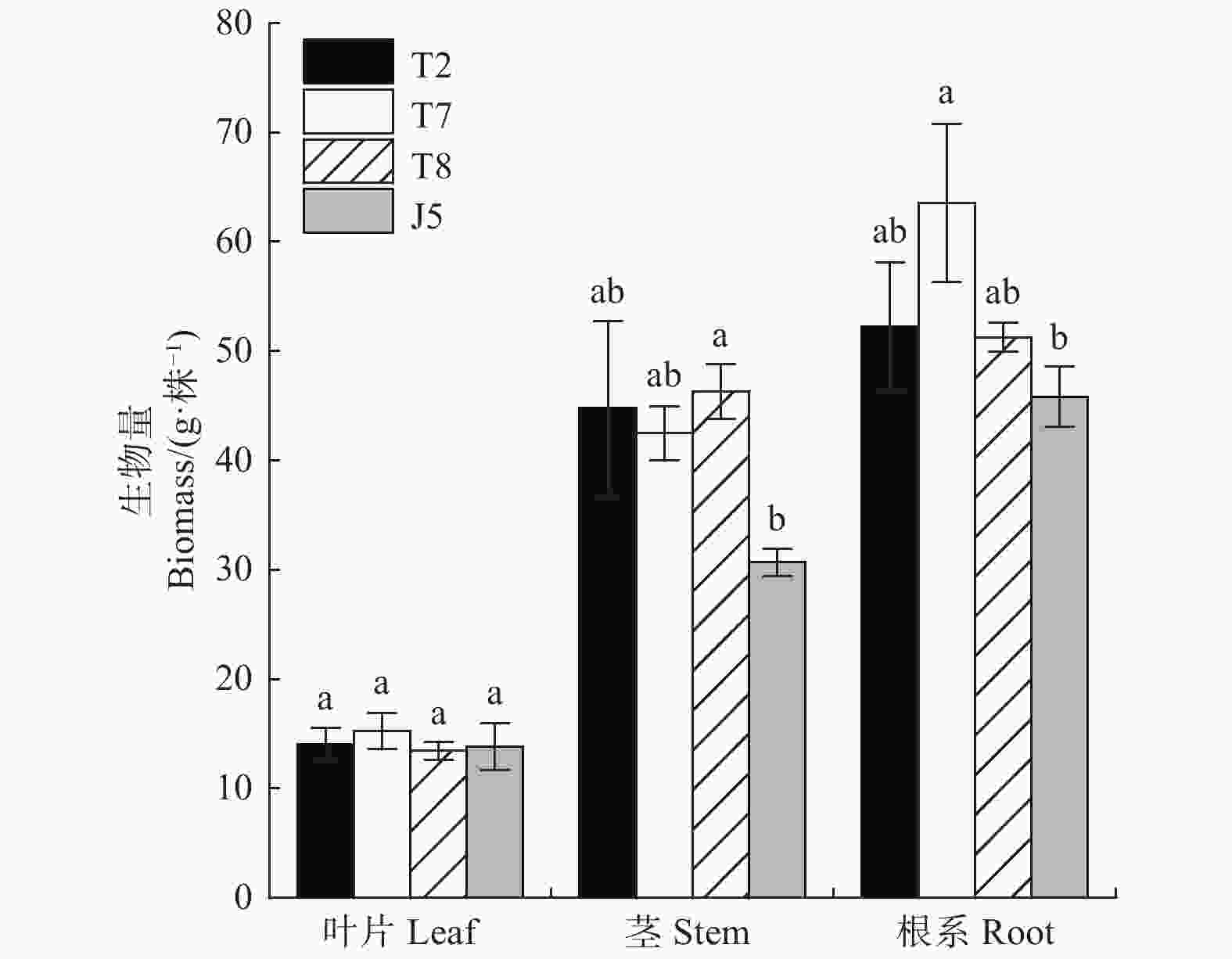
重金属胁迫下参试栓皮栎各家系叶片生物量无显著差异(图2),其中泰山7号家系叶片生物量高于其它家系,为15.2 g·株−1。徂徕山5号家系茎生物量低于其它家系,仅为30.7 g·株−1,其它家系茎生物量无显著差异(图2)。徂徕山5号家系根系生物同样表现为低于其它家系,为45.8 g·株−1。而泰山7号家系根系生物量为63.6 g·株−1,高于其它家系。
-
重金属胁迫下,栓皮栎家系体内营养元素浓度也表现出一定差异(表1)。4个家系栓皮栎叶片氮(N)浓度为18.13~20.27 g·kg−1,其中泰山2号家系叶片N浓度较高。栓皮栎茎N浓度为4.53~6.57 g·kg−1,其中徂徕山5号家系茎N浓度最高。方差分析表明徂徕山5号家系根系N浓度显著高于其它家系,为12.22 g·kg−1。各家系N吸收量无显著差异,为0.85~1.02 g·株−1。N利用效率则在家系间差异显著,为88.8~142.5 g·g−1,其中徂徕山5号家系N利用效率最低。重金属胁迫下,4个家系栓皮栎叶片磷(P)浓度为1.28~1.88 g·kg−1,其中泰山2号家系叶片P浓度最高。栓皮栎茎P浓度为0.69~0.82 g·kg−1,且在家系间无显著差异。方差分析表明泰山8号家系根系P浓度显著高于其它家系,为1.17 g·kg−1。各家系P吸收量为0.079~0.119 g·株−1,其中徂徕山5号家系P吸收量最低。各家系P利用效率则表现出相反趋势,徂徕山5号家系P利用效率为1136.5 g·g−1,高于其它家系。泰山2号家系P利用效率最低,仅为936.7 g·g−1。
表 1 铅锌矿砂环境下4个栓皮栎家系各器官中氮和磷浓度
Table 1. Average N and P concentrations in different organs of four families from Q. variabilis on Pb/Zn mine tailing
器官 Organ 家系 Family N/ (g·kg−1) P/ (g·kg−1) 叶片 Leaf T2 20.27 ± 1.16 a 1.88 ± 0.22 a T7 18.13 ± 1.33 a 1.63 ± 0.06 ab T8 19.60 ± 2.19 a 1.60 ± 0.33 ab J5 18.47 ± 2.40 a 1.28 ± 0.17 b 茎 Stem T2 5.26 ± 1.44 ab 0.82 ± 0.09 a T7 4.59 ± 1.31 ab 0.69 ± 0.08 a T8 4.53 ± 0.59 b 0.80 ± 0.07 a J5 6.57 ± 1.41 a 0.70 ± 0.10 a 根系 Root T2 7.45 ± 1.10 bc 0.76 ± 0.12 b T7 5.99 ± 0.64 c 0.88 ± 0.11 b T8 8.12 ± 0.55 b 1.17 ± 0.11 a J5 12.22 ± 1.25 a 0.88 ± 0.11 b 注:不同小写字母表示家系间差异显著(P < 0.05)。下同。
Note: Lowercase letters in the same row meant significant difference among families at 0.05 level. The same below. -
4个栓皮栎家系对不同重金属的吸收和转运表现出一定差异(表2)。由表2 可知,4个栓皮栎家系体内铅(Pb)浓度表现为根系 > 叶片 > 茎,而镉(Cd)浓度则表现为根系 > 茎 > 叶片(徂徕山5号家系除外)。相比Cd和Pb,栓皮栎各家系体内锌(Zn)和铜(Cu)浓度表现为叶片 > 根系 > 茎。研究表明徂徕山5号家系体内各重金属浓度最高(叶片Zn除外);泰山8号家系体内重金属浓度较低(叶片Zn和茎 Cu除外)。
表 2 铅锌矿砂环境下4个栓皮栎家系各器官中重金属浓度
Table 2. Average heavy metal concentrations in different organs of four families from Q. variabilis on Pb/Zn mine tailing
器官 Organ 家系 Family Cd/(mg·kg−1) Pb/(mg·kg−1) Zn/(mg·kg−1) Cu/(mg·kg−1) 叶片 Leaf T2 0.39 ± 0.17 b 14.01 ± 1.35 b 81.00 ± 8.63 a 13.70 ± 1.23 a T7 0.35 ± 0.05 b 7.17 ± 0.79 c 52.03 ± 5.88 c 14.23 ± 1.25 a T8 0.29 ± 0.04 b 4.59 ± 0.86 c 58.37 ± 2.75 bc 14.33 ± 1.59 a J5 0.87 ± 0.17 a 18.07 ± 2.77 a 68.13 ± 8.56 ab 16.30 ± 3.18 a 茎 Stem T2 0.50 ± 0.07 b 4.97 ± 0.29 b 35.00 ± 1.08 a 4.38 ± 0.91 a T7 0.42 ± 0.04 bc 6.87 ± 0.79 a 34.37 ± 4.35 a 4.72 ± 0.65 a T8 0.34 ± 0.05 c 3.27 ± 0.92 c 29.10 ± 3.67 a 4.43 ± 0.41 a J5 0.69 ± 0.07 a 7.18 ± 0.54 a 36.67 ± 7.49 a 4.60 ± 0.73 a 根系 Root T2 0.71 ± 0.08 b 29.73 ± 3.72 b 43.60 ± 2.46 a 6.62 ± 1.09 b T7 0.60 ± 0.13 b 30.87 ± 7.16 b 47.67 ± 12.48 a 6.39 ± 0.69 b T8 0.65 ± 0.12 b 18.07 ± 1.31 c 42.80 ± 3.66 a 5.72 ± 1.06 b J5 0.93 ± 0.09 a 43.60 ± 5.38 a 58.40 ± 11.78 a 8.64 ± 1.19 a 方差分析表明泰山8号家系地上部Cd和Pb含量显著低于其它家系,分别为0.020和0.21 mg。尽管徂徕山5号家系地上部生物量低于其它家系,但其体内重金属浓度较高,因而其地上部Cd和Pb含量较高,每株分别为0.032和0.45 mg。各家系地上部Zn和Cu含量无显著差异,每株分别为2.02~2.72 mg和0.36~0.41 mg,其中徂徕山5号家系体内含量最低。泰山8号家系根系各重金属含量均低于其它家系,Cd、Pb、Zn和Cu含量分别为0.033、0.92、2.19和0.29 mg。
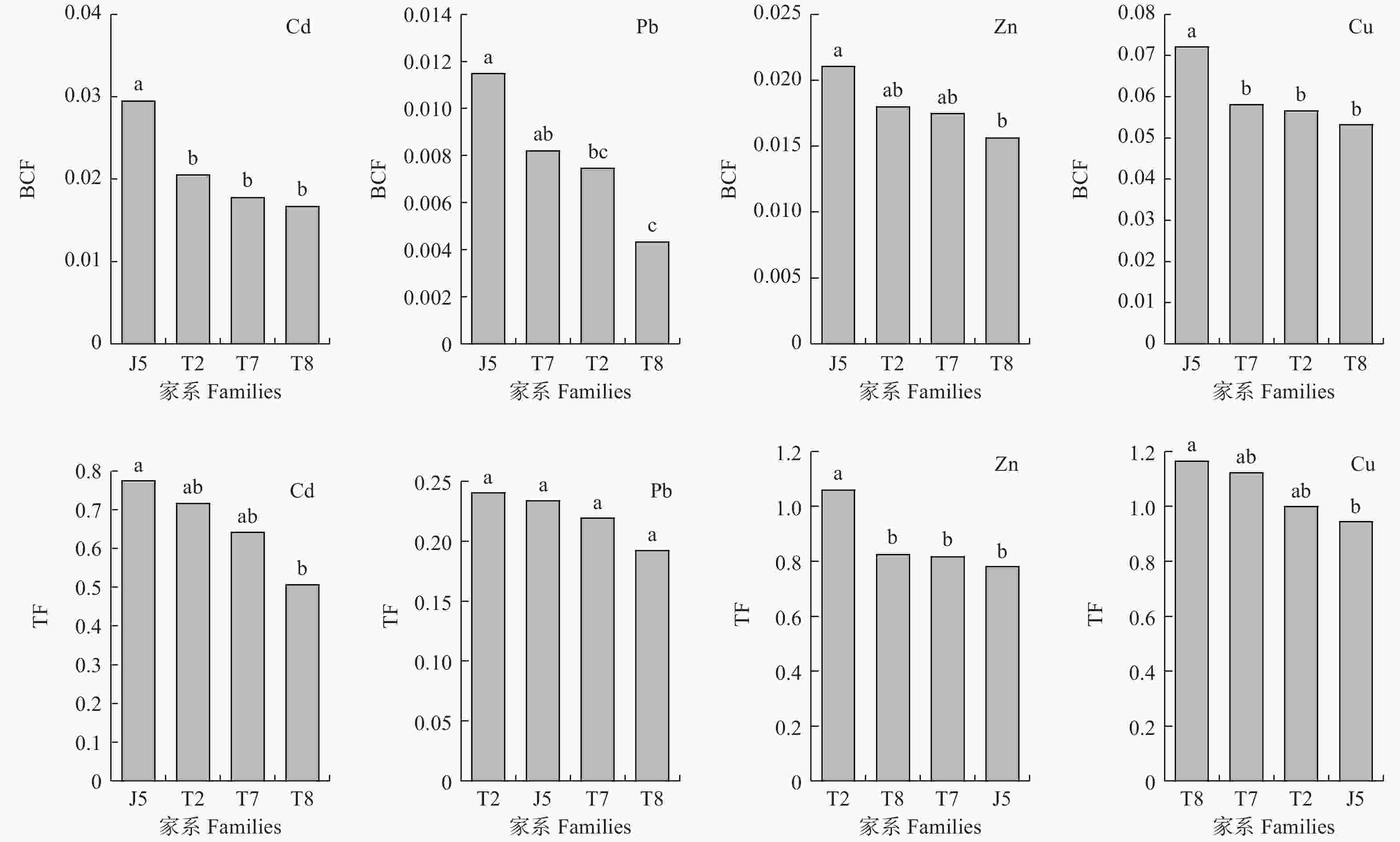
由图3可知,徂徕山5号家系各重金属富集系数(BCF)均最高,表现出较强的重金属富集能力。泰山8号家系各重金属BCF值均低于其它家系,较难富集各重金属。通常各家系较容易富集Cu,而较难富集Pb。参试材料重金属BCF值均小于0.100,特别是铅BCF值均小于0.012。栓皮栎家系对各重金属表现出不同的转移能力(图3),其中徂徕山5号和泰山2号家系对Cd和Pb有较强的转移能力。泰山2号家系对Zn的转移能力较强,其重金属转移系数(TF)为1.06,显著高于其它家系。泰山8号和泰山7号家系对Cu的转移能力较强,其TF值分别为1.16和1.12。总体而言,参试材料较容易转移Cu和Zn,较难转移Pb,其TF值均小于0.25。
-
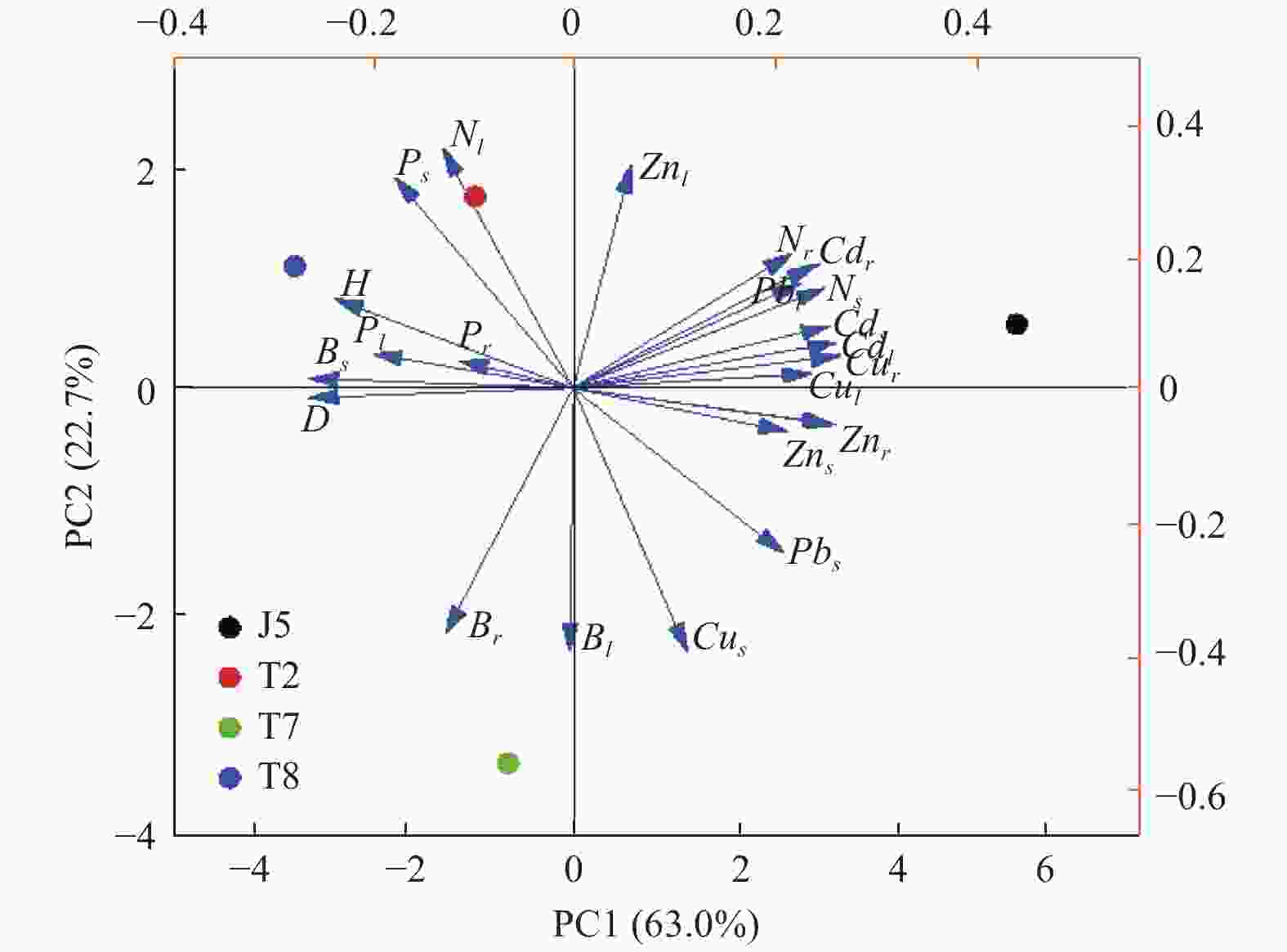
根据主成分分析(图4),前2轴解释率为85.7%,第1主成分(PC1)占63.0%,其中高载荷且为正值的指标有植物体内镉和铅浓度、茎和根系锌和氮浓度、叶片和根系铜浓度,为负值的指标为苗高、地径、茎生物量和叶片磷浓度。说明家系间形态学指标以及茎生物量差异主要由体内重金属浓度决定。第2主成分(PC2)占22.7%,其中高载荷且为负值的指标有叶片和根系生物量,为正值的指标有叶片锌和氮浓度、茎铜和磷浓度。图4展示了标准化的主成分得分和各家系所处的位置。从第1主成分得分排序看,植物体内重金属元素较高的徂徕山5号家系得分最高,而体内重金属浓度较低的泰山8号家系得分最低。表明植物体内重金属浓度水平在第1主成分中起主导作用。从第2主成分得分排序看,植物地上部磷浓度以及叶片氮浓度较高的家系得分较高,泰山7号家系体内氮磷浓度相对较低,因此得分最低。表明植物地上部营养元素浓度在第2主成分中起主导作用。
-
通常植物在高浓度重金属胁迫下会表现出一定的毒害效应[13]。本研究中4个栓皮栎家系在铅锌矿砂中未出现显著毒害效应,表明参试材料对重金属有较强耐性,也初步说明栓皮栎是潜在的重金属耐性植物。研究也发现重金属胁迫下参试材料苗高、地径、茎以及根系生物量表现出一定差异,这表明不同家系对重金属响应有所不同,这可能与其基因型有关。研究也证实来自山东徂徕山地区的栓皮栎家系苗高、地径以及整株生物量均小于泰山地区的3个栓皮栎家系,表明不同种源的环境可塑性以及种源地环境异质性是造成这种现象的可能原因。本研究表明徂徕山5号家系在铅锌矿砂环境下耐性较弱。尽管如此,徂徕山5号家系茎和根系氮浓度高于其它家系,这可能是因为其生物量较低,造成养分浓缩,这与李金波等[14]的研究相似。尽管供试矿砂氮磷浓度低,但本研究中4个栓皮栎家系仍具有较高氮磷利用效率,特别是磷利用效率高于枫香(Liquidambar formosana Hance)[12]和部分木荷(Schima superba Gardn. et Champ.)种源[15],这可能是其具有较高重金属耐性的原因。这与植物耐铝能力与氮磷利用效率具有一定关联的结果相似[16]。
-
本研究中参试4个栓皮栎家系较难从高浓度铅锌矿砂中富集重金属,这与蔡志全等[10]的研究结果一致。此外本研究中铅锌矿砂pH值为7.84,这可能是参试材料体内重金属浓度较低的原因之一[17]。但也有研究表明栓皮栎在锑(Sb)污染土壤中根系Sb浓度可达1623.4 mg·kg−1[18]。这可能是因为栓皮栎对不同重金属的吸收积累有显著差异。通常重金属主要赋存在木本植物根系[19]。本研究各参试栓皮栎家系也有相似表现,其TF值均小于1.00(泰山2号家系锌、泰山7号和泰山8号铜TF值除外)。较低的TF值表明重金属较难转运至栓皮栎地上部,可能是其在重金属胁迫下的另一种保护机制[20]。与麻栎(Q. acutissima Carruth.)[21]相比,栓皮栎镉和铅的TF值较低,可能与试验基质不同有关。而在基质相似的铅锌矿砂胁迫下,栓皮栎重金属TF值较引种的北美栎树(Q. spp.)高(铅除外),特别是其铜TF值显著高于北美栎树[11]。总体而言,参试栓皮栎体内重金属浓度较其它木本植物低[22],其叶片和茎重金属浓度通常在植物正常范围或未达到毒害浓度[23],这可能是栓皮栎在本研究环境下无显著毒害效应的一个重要原因。
研究表明参试栓皮栎家系体内重金属浓度较低,但家系间差异显著。如徂徕山5号家系体内镉和铅浓度较其它家系高。这可能是因为栓皮栎分布广泛,加上长期的地理隔离和自然选择,导致种内分化,从而使得栓皮栎重金属积累机制存在自然变异[24]。
-
本研究中栓皮栎各家系生长速度较快,抗性强,根系较为发达,体内重金属浓度较低。根据Mendez 等[25]的研究,可认为本研究参试的4个栓皮栎家系可作为植物固定修复(Phytostabilisation)的参考植物。有研究报道饲料中允许的最大微量元素浓度(干质量)分别为150(锌)、40(铅)和1(镉)mg·kg−1[22]。而本研究参试栓皮栎叶片生物量分配比例较小,且叶片中重金属浓度均低于以上标准,因此通过食物链向环境释放重金属的风险较低。研究也发现栓皮栎各家系每年生长表现均较为相似,且平茬后的地上部生物量逐年增加。同时栓皮栎体内重金属浓度在年度表现上无显著差异。因此可利用栓皮栎良好的萌蘖能力,适当密植,提高单位面积生物量,在实现尾矿区绿化的同时逐年去除土壤重金属。
-
参试栓皮栎家系在铅锌矿砂环境中的生长未受到抑制,这与其有较高的营养元素利用效率以及体内重金属浓度较低有关。参试栓皮栎家系的重金属富集能力较低,富集系数(BCF)均小于0.100;除铅外,参试栓皮栎对其它重金属表现一定的转移能力。4个栓皮栎家系在铅锌矿砂中的生长、重金属积累转移能力存在一定差异,其中泰山8号家系重金属的积累和转移能力较弱。初步研究表明,参试栓皮栎家系特别是泰山8号家系可作为有潜力的污染土壤修复树种,主要起到植物固定修复作用。今后可通过研究重金属胁迫下栓皮栎体内各元素的相互关系来进一步探究其重金属耐性。
不同栓皮栎家系对重金属的耐性和富集特性
Tolerance and Enrichment Characteristics of Different Families of Quercus variabilis to Heavy Metal
-
摘要:
目的 通过野外试验,评价栓皮栎家系在铅锌尾矿环境中的生长响应、重金属积累能力及土壤修复潜力。 方法 在杭州市富阳区废弃的铅锌尾矿库进行人工植被恢复试验,分析比较4个栓皮栎家系(泰山3号、泰山7号、泰山8号和徂徕山5号)在铅锌矿砂中生长3 a的生物量及其对营养元素和重金属的吸收及转移特征。 结果 4个栓皮栎家系在矿砂环境中均能生长,未表现出毒害效应。重金属胁迫下,栓皮栎家系各器官营养元素浓度表现出一定差异,且各家系均有较高的氮磷利用效率。研究表明参试栓皮栎家系体内重金属浓度较低,且各家系重金属生物富集系数(BCF)均小于0.100。除铅外,各家系重金属转移系数(TF)均大于0.50,表现出一定的重金属转移能力。泰山8号家系平均BCF和TF值均低于其它家系。 结论 3 a的长期试验表明,参试栓皮栎家系耐性较高、重金属生物富集系数和转移系数较低,可作为有潜力的污染土壤修复树种。 Abstract:Objective A filed test was conducted to evaluate the growth response, enrichment and transfer characteristics to heavy metal and vegetation restoration prospect of four families from Quercus variabilis. Method Vegetation restoration test was carried out in the abandoned lead-zinc tailings for 3 years. The seedlings of four families (Taishan 3, Taishan 7, Taishan 8, and Julaishan 5) were transplanted into Pb/Zn mine tailing to compare their biomass, uptake and transfer characteristics of nutrient elements and heavy metals. Result The results showed that all the seedlings could survive in the Pb/Zn tailing and the toxicity symptoms was not observed. Under heavy metal stress, there were statistically significant differences among the seedlings of the four families according to the nutrient concentrations. Meanwhile, all families had high capability of utilization efficiency. Generally, the concentrations of heavy metals in different organs of Q. variabilis were low, and the values of bioconcentration factor (BCF) were less than 0.100. The values of translocation factor (TF) were more than 0.5, except Pb. The average BCF and TF values of family Taishan 8 were lower than those of the other families. Conclusion The results suggest that the families from Q. variabilis have high tolerance with the low BCF and TF, implying that they are better potential candidates for contaminated soil. -
Key words:
- lead/zinc mine tailing
- / Quercus variabilis
- / heavy metal
- / enrichment
-
表 1 铅锌矿砂环境下4个栓皮栎家系各器官中氮和磷浓度
Table 1. Average N and P concentrations in different organs of four families from Q. variabilis on Pb/Zn mine tailing
器官 Organ 家系 Family N/ (g·kg−1) P/ (g·kg−1) 叶片 Leaf T2 20.27 ± 1.16 a 1.88 ± 0.22 a T7 18.13 ± 1.33 a 1.63 ± 0.06 ab T8 19.60 ± 2.19 a 1.60 ± 0.33 ab J5 18.47 ± 2.40 a 1.28 ± 0.17 b 茎 Stem T2 5.26 ± 1.44 ab 0.82 ± 0.09 a T7 4.59 ± 1.31 ab 0.69 ± 0.08 a T8 4.53 ± 0.59 b 0.80 ± 0.07 a J5 6.57 ± 1.41 a 0.70 ± 0.10 a 根系 Root T2 7.45 ± 1.10 bc 0.76 ± 0.12 b T7 5.99 ± 0.64 c 0.88 ± 0.11 b T8 8.12 ± 0.55 b 1.17 ± 0.11 a J5 12.22 ± 1.25 a 0.88 ± 0.11 b 注:不同小写字母表示家系间差异显著(P < 0.05)。下同。
Note: Lowercase letters in the same row meant significant difference among families at 0.05 level. The same below.表 2 铅锌矿砂环境下4个栓皮栎家系各器官中重金属浓度
Table 2. Average heavy metal concentrations in different organs of four families from Q. variabilis on Pb/Zn mine tailing
器官 Organ 家系 Family Cd/(mg·kg−1) Pb/(mg·kg−1) Zn/(mg·kg−1) Cu/(mg·kg−1) 叶片 Leaf T2 0.39 ± 0.17 b 14.01 ± 1.35 b 81.00 ± 8.63 a 13.70 ± 1.23 a T7 0.35 ± 0.05 b 7.17 ± 0.79 c 52.03 ± 5.88 c 14.23 ± 1.25 a T8 0.29 ± 0.04 b 4.59 ± 0.86 c 58.37 ± 2.75 bc 14.33 ± 1.59 a J5 0.87 ± 0.17 a 18.07 ± 2.77 a 68.13 ± 8.56 ab 16.30 ± 3.18 a 茎 Stem T2 0.50 ± 0.07 b 4.97 ± 0.29 b 35.00 ± 1.08 a 4.38 ± 0.91 a T7 0.42 ± 0.04 bc 6.87 ± 0.79 a 34.37 ± 4.35 a 4.72 ± 0.65 a T8 0.34 ± 0.05 c 3.27 ± 0.92 c 29.10 ± 3.67 a 4.43 ± 0.41 a J5 0.69 ± 0.07 a 7.18 ± 0.54 a 36.67 ± 7.49 a 4.60 ± 0.73 a 根系 Root T2 0.71 ± 0.08 b 29.73 ± 3.72 b 43.60 ± 2.46 a 6.62 ± 1.09 b T7 0.60 ± 0.13 b 30.87 ± 7.16 b 47.67 ± 12.48 a 6.39 ± 0.69 b T8 0.65 ± 0.12 b 18.07 ± 1.31 c 42.80 ± 3.66 a 5.72 ± 1.06 b J5 0.93 ± 0.09 a 43.60 ± 5.38 a 58.40 ± 11.78 a 8.64 ± 1.19 a -
[1] Singh R, Gautam N, Mishra A, et al. Heavy metals and living systems: An overview[J]. Indian Journal Pharmacology, 2011, 43(3): 246-253. doi: 10.4103/0253-7613.81505 [2] Sánchez-Chardi A, Ribeiro C A O, Nadal J. Metals in liver and kidneys and the effects of chronic exposure to pyrite mine pollution in the shrew Crocidura russula inhabiting the protected wetland of Doñana[J]. Chemosphere, 2009, 76(3): 387-394. doi: 10.1016/j.chemosphere.2009.03.036 [3] Álvarez-Mateos P, Alés-Álvarez F-J, García-Martín J F. Phytoremediation of highly contaminated mining soils by Jatropha curcas L. and production of catalytic carbons from the generated biomass[J]. Journal of Environmental Management, 2019, 231: 886-895. doi: 10.1016/j.jenvman.2018.10.052 [4] Jiménez-Moraza C, Iglesias N, Palencia I. Application of sugar foam to a pyrite-contaminated soil[J]. Minerals Engineering, 2006, 19(5): 399-406. doi: 10.1016/j.mineng.2005.10.011 [5] 施 翔, 陈益泰, 王树凤, 等. 废弃尾矿库15种植物对重金属Pb、Zn的积累和养分吸收[J]. 环境科学, 2012, 33(6):2021-2027. [6] Midhat L, Ouazzani N, Hejjaj A. Accumulation of heavy metals in metallophytes from three mining sites (Southern Centre Morocco) and evaluation of their phytoremediation potential[J]. Ecotoxicology and Environmental Safety, 2019, 169: 150-160. doi: 10.1016/j.ecoenv.2018.11.009 [7] Mendez M O, Maier R M. Phytostabilization of mine tailings in arid and semiarid environments-an emerging remediation technology[J]. Environmental Health Perspectives, 2008, 116(3): 278-283. doi: 10.1289/ehp.10608 [8] Zhou L Y, Zhao Y, Wang S F, et al. Lead in the soil-mulberry (Morus alba L.)-silkworm (Bombyx mori) food chain: translocation and detoxification[J]. Chemosphere, 2015, 128: 171-177. doi: 10.1016/j.chemosphere.2015.01.031 [9] Salam M M A, Kaipiainen E, Mohsin M, et al. Effects of contaminated soil on the growth performance of young Salix (Salix schwerinii E. L. Wolf) and the potential for phytoremediation of heavy metals[J]. Journal of Environmental Management, 2016, 183: 467-477. doi: 10.1016/j.jenvman.2016.08.082 [10] 蔡志全, 阮宏华, 叶镜中. 栓皮栎林对城郊重金属元素的吸收和积累[J]. 南京林业大学学报: 自然科学版, 2001, 25(1):18-22. [11] 施 翔, 王树凤, 陈益泰, 等. 5种栎树幼苗对铅锌尾矿砂的耐性与植被恢复前景[J]. 应用生态学报, 2019, 30(12):4091-4098. [12] 冷华妮, 陈益泰, 段红平, 等. 磷胁迫对不同种源枫香生长及氮、磷吸收利用率的影响[J]. 应用生态学报, 2009, 20(4):754-760. [13] Malar S, Manikandan R, Favas P J C, et al. Effect of lead on phytotoxicity, growth, biochemical alterations and its role on genomic template stability in Sesbania grandiflora: A potential plant for phytoremediation[J]. Ecotoxicology and Environmental Safety, 2014, 108: 249-257. doi: 10.1016/j.ecoenv.2014.05.018 [14] 李金波, 李诗刚, 宋桂龙, 等. 砷胁迫对黑麦草根系形态及养分吸收的影响[J]. 草业科学, 2018, 35(6):1385-1392. doi: 10.11829/j.issn.1001-0629.2017-0434 [15] 林 磊, 周志春. 水分和磷素对木荷不同种源苗木生长和磷效率的影响[J]. 应用生态学报, 2009, 20(11):2617-2623. [16] 赵学强, 沈仁芳. 提高铝毒胁迫下植物氮磷利用的策略分析[J]. 植物生理学报, 2015, 51(10):1583-1589. [17] Bolan N, Kunhikrishnan A, Thangarajan R, et al. Remediation of heavy metal(loid)s contaminated soils – To mobilize or to immobilize[J]. Journal of Hazardous Materials, 2014, 266: 141-166. doi: 10.1016/j.jhazmat.2013.12.018 [18] Zhao X L, Zheng L Y, Xia X L, et al. Responses and acclimation of Chinese cork oak (Quercus variabilis Bl.) to metal stress: the inducible antimony tolerance in oak trees[J]. Environmental Science and Pollution Research, 2015, 22(15): 11456-11466. doi: 10.1007/s11356-015-4304-2 [19] Luo Z B, He J L, Polle A et al. Heavy metal accumulation and signal transduction in herbaceous and woody plants: Paving the way for enhancing phytoremediation efficiency[J]. Biotechnology Advances, 2016, 34(6): 1131-1148. doi: 10.1016/j.biotechadv.2016.07.003 [20] de Souza S C R, de Andrade S A L, de Souza L A, et al. Lead tolerance and phytoremediation potential of Brazilian leguminous tree species at the seedling stage[J]. Journal of Environmental Management, 2012, 110: 299-307. doi: 10.1016/j.jenvman.2012.06.015 [21] 王 君, 严小莉, 李 凌. 不同种源麻栎幼苗对Cd2+-Pb2+复合污染的吸收累积特性[J]. 林业科学, 2014, 50(7):23-30. [22] Evangelou M W H, Robinson B H, Gunthardt-Goerg M S, et al. Metal uptake and allocation in trees grown on contaminated land: implications for biomass production[J]. International Journal of Phytoremediation, 2013, 15(1): 77-90. doi: 10.1080/15226514.2012.670317 [23] Kabata-Pendias A. Trace element in soils and plants[M]. Fourth Edition.Boca Raton, Florida: CRC Press, 2011: 105. [24] Chen D Q, Zhang H, Wang Q L, et al. Intraspecific variations in cadmium tolerance and phytoaccumulation in giant duckweed (Spirodela polyrhiza)[J]. Journal of Hazardous Materials, 2020, 395: 122672. doi: 10.1016/j.jhazmat.2020.122672 [25] Mendez M O, Maier R M. Phytoremediation of mine tailings in temperate and arid environments[J]. Reviews in Environmental Science and Bio/Technology, 2008, 7(1): 47-59. doi: 10.1007/s11157-007-9125-4 -




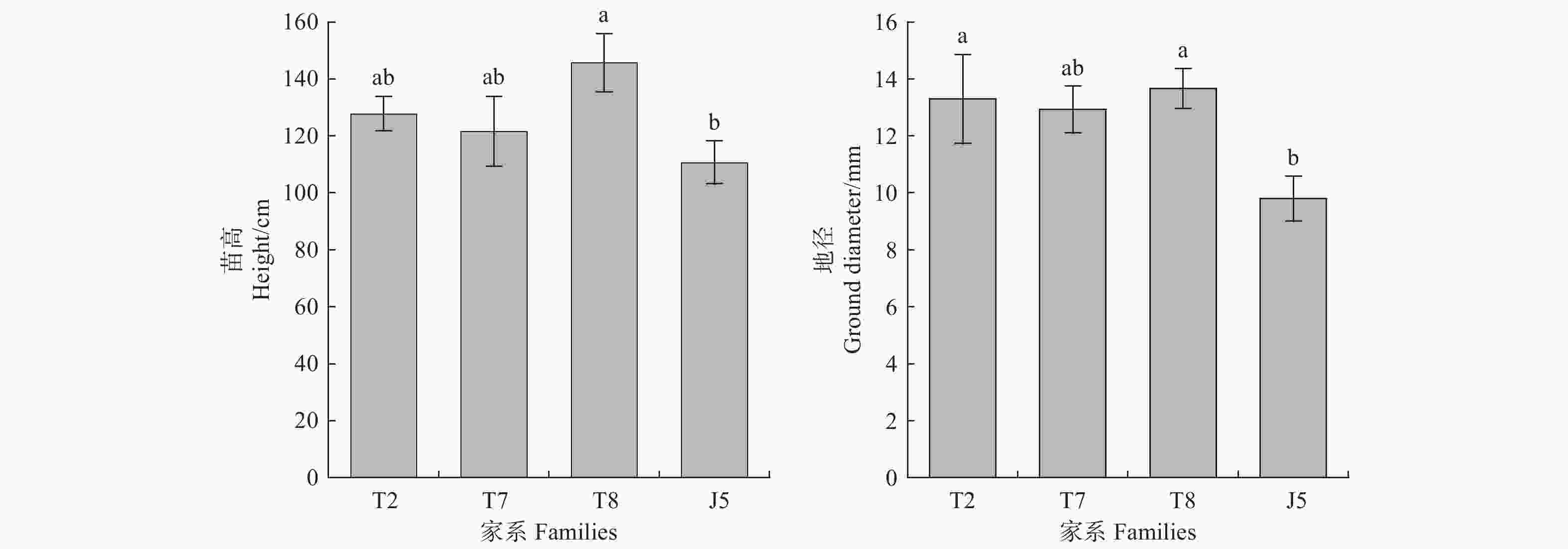
 下载:
下载: