-
肠道为昆虫的一个重要器官,由于肠道的特殊生态环境,其中栖息着大量的微生物。昆虫肠道微生物与宿主昆虫的生命活动密切相关,参与摄食、消化、排泄及繁殖等一系列过程,同时还与有毒物质的降解、信息素的合成以及宿主免疫反应等有关[1-3]。植食性昆虫肠道微生物群落的结构与多样性受到所食植物的影响。在对棉铃虫(Helicoverpa armigera)[4]、菜粉蝶(Pieris rapae)[5]、小菜蛾(Plutella xylostella)[6]、松异舟蛾(Thaumetopoea pityocampa)[7]等昆虫的研究中证实,肠道细菌群落的组成与昆虫所食植物叶际细菌群落的组成相似,表明食物是植食性昆虫肠道微生物的重要来源。植食性昆虫啃食植物的同时会给植物造成大量伤口,这些伤口使植物更易受到病原微生物的侵入[8-9]。同时,肠道是病原微生物定殖的重要场所,部分病原微生物可在昆虫肠道内存活,昆虫则成为了病原微生物的越冬场所或来年病害的初侵染来源[10]。
油桐尺蛾(Buzura suppressaria)是为害桉树的重要灾害性食叶害虫,近年来的为害率和为害面积均呈快速上升趋势[11]。在以往的林间调查过程中,我们发现油桐尺蛾虫害与叶部病害经常在同一片桉树林中发生,但究竟是油桐尺蛾先为害然后引起病害侵入?还是病害先侵染桉树,引起植株产生生理生化改变,继而吸引油桐尺蛾为害?或者上述现象仅仅只是一个巧合?由于目前尚未有相关方面的报道,因此有必要对上述问题开展研究。目前对油桐尺蛾的研究主要集中在生物学、生理学、生态学、化学或生物防治等方面[12-14]。鉴于肠道微生物对油桐尺蛾的生命活动有重要影响,因此分析油桐尺蛾肠道微生物群落的结构和多样性,寻找与桉树叶部微生物的关联,对于研究油桐尺蛾与桉树间的互作机理、明确为害规律、寻找新的防治方法均具有重要意义。
-
油桐尺蛾幼虫采集自广西国有东门林场桉树林地。随机选取20头5龄油桐尺蛾幼虫,饥饿24 h后于75%酒精中浸泡2 min,无菌水清洗3次并用滤纸擦干。实验中所有解剖工具均经高压灭菌,于超净工作台中解剖油桐尺蛾幼虫,采集中肠组织。将中肠组织随机分为5组,保存于−80℃冰箱备用。
-
在采集油桐尺蛾的同一片桉树林地采集桉树叶片。通过多点取样的方式采集样品,同组样品等量混合成一个样品(5 g·组−1)。叶片用无菌水洗涤30 s,浸泡于70%酒精中洗涤2 min,2.5% NaClO(含0.1% Tween 80)浸泡5 min,随后将叶片转移至70%酒精中浸泡30 s,用无菌水洗涤3次;叶片样品经上述步骤处理后即可视为表面无菌,用于检测叶片内生菌,保存于−80℃备用。
-
分别将油桐尺蛾中肠组织和经无菌化处理的桉树叶样品用液氮磨成粉末,采用E.Z.N.ATM Mag-Bind Soil DNA Kit试剂盒(OMEGA,M5635-02)提取样品基因组DNA,具体操作参照试剂盒说明书。待基因组DNA抽提完成后,用1%琼脂糖凝胶电泳检测抽提的基因组DNA。每组样品均设5个重复,油桐尺蛾中肠样品编号为“BS”,桉树叶样品编号为“BCN”。
-
样品细菌16s rDNA的扩增区域为16SV3-V4,采用细菌16s rDNA基因通用引物341F:5′-CCTACGGGNGGCWGCAG-3′和805R:5′-GACTACHVGGGTATCTAATCC-3′加接头后进行PCR扩增。样品真菌ITS rDNA扩增区域为ITS1-2,采用真菌ITS rDNA通用引物ITS1F:5′-CTTGGTCATTTAGAGGAAGTAA-3′和ITS2R:5′-GCTGCGTTCTTCATCGATGC-3′加接头后进行PCR扩增。第一轮PCR反应体系(30 μL):2 × Taq master Mix 15 μL,F/R引物(10 μmol·L−1)各1 μL,DNA10~20 ng,ddH2O补齐总体积至30 μL。第一轮PCR扩增条件:94℃ 3 min;94℃ 30 s,45℃ 20 s,65℃ 30 s,共5个循环;94℃ 20 s,55℃ 20 s,72℃ 30 s,共20个循环;72℃ 5 min。上述PCR反应结束后进行第二轮PCR扩增,引入Illumina桥式PCR兼容引物。第二轮PCR反应体系(30 μL):2 × Taq master Mix15 μL,F/R引物(10 μmol·L−1)各1 μL,上一轮PCR产物DNA 20 ng,ddH2O补齐总体积至30 μL。第二轮PCR扩增条件:95℃ 3 min;94℃ 20 s,55℃ 20 s,72℃ 30 s,共5个循环;72℃ 5 min。上述PCR反应结束后,用1%琼脂糖凝胶电泳检测PCR产物。使用磁珠纯化方法对PCR产物进行纯化,将纯化产物保存于−80℃ 冰箱备用。
-
利用Qubit3.0 DNA检测试剂盒(Life,Q10212)对纯化的PCR产物精确定量,以方便按照1∶1等量混合后测序。等量混合时,每个样品DNA量取10 ng,最终上机测序浓度为20 pmol。样品委托生工生物工程(上海)股份有限公司基于IlluminaMiseq技术测序平台构建文库,进行高通量双末端测序。根据相似度,将序列聚类为操作分类单元(operational taxonomic unit,OTU),采用RDP(ribosomal database project)classifier贝叶斯算法对97%相似度水平的OTU代表序列进行分类学分析。使用RDP分类器对序列进行系统分析,并在各分类级别上对物种进行统计,依照OTU数据进行Alpha和Beta多样性分析、物种分类树分析、菌群差异分析。
-
结果显示(表1),5个BS样品的细菌16s rDNA文库测序共获得3 271条有效序列,基于97%的序列相似性进行聚类分析,得到199个OTUs,共注释到6门、11纲、19目、38科、43属;真菌ITS rDNA文库测序共获得280 392条有效序列,得到4 279个OTUs,共注释到8门、29纲、67目、159科、257属。5个BCN样品细菌16s rDNA文库测序共获得11 413条有效序列,得到379个OTUs,共注释到10门、19纲、26目、54科、74属;真菌ITS rDNA文库为287 330条有效序列,获得4 236个OTUs,共注释到8门,31纲,79目,171科,291属。从属水平上看,BCN样品的细菌和真菌数目均多于BS样品。
表 1 BCN和BS样品OTU聚类及RDP分类水平统计
Table 1. OTU clustering and RDP classification level statistics of BCN and BS samples
样品
Sample检测项目
Test items有效reads数目/条
Reads numOTU数目/个
OUT numRDP分类
Classification of RDP门
Phylum纲
Class目
Order科
Family属
GenusBS 细菌 Bacteria 3 271 199 6 11 19 38 43 真菌 Fungus 280 392 4 279 8 29 67 159 257 BCN 细菌 Bacteria 11 413 379 10 19 26 54 74 真菌 Fungus 287 330 4 236 8 31 79 171 291 -
细菌和真菌群落的α多样性检测结果显示(表2),BCN和BS样品物种覆盖率均达97%以上,表明样品所构建的细菌和真菌文库均能有效反应其物种的多样性,且各组样品多样性指数组内偏差较小,数据可靠。BCN样品的细菌ACE指数和Chao l指数均明显高于BS样品,说明病害桉树叶内生细菌的群落丰富度较高,而上述两组样品的真菌ACE指数和Chao l指数则无显著差异,即两者的真菌群落丰富度差异较小。此外,BCN细菌和真菌的Shannon指数、覆盖率、Simpson指数较BS样品均无统计学差异,表明两组样品在细菌、真菌群落多样性、优势菌种的集中程度方面无明显差异。
表 2 BCN和BS样品α多样性指数统计
Table 2. Alpha diversity index statistics of BCN and BS samples
样品
Sample检测项目
Test itemsOTU数量
OUT numSimpson指数
Simpson indexShannon指数
Shannon indexACE指数
ACE indexChao1指数
Chao1 index覆盖率
coverageBS 细菌 Bacteria 39.80 ± 14.82 0.30 ± 0.10 1.90 ± 0.43 61.37 ± 24.07 53.55 ± 14.68 0.97 ± 0.01 真菌 Fungus 855.80 ± 168.88 0.10 ± 0.05 3.60 ± 0.47 1 098.59 ± 164.90 1 058.26 ± 156.59 1.0 ± 0.00 BCN 细菌 Bacteria 75.80 ± 32.71 0.21 ± 0.07 2.33 ± 0.57 109.82 ± 33.31a 104.12 ± 38.89a 0.98 ± 0.01 真菌 Fungus 847.20 ± 142.69 0.13 ± 0.13 3.50 ± 0.71 1 167.82 ± 103.06 1 094.02 ± 147.21 1.0 ± 0.00 注:a为与BS样品细菌检测组相比,P < 0.05
Note: P is compared with the BS samples in the bacterial detection group, P < 0.05 -
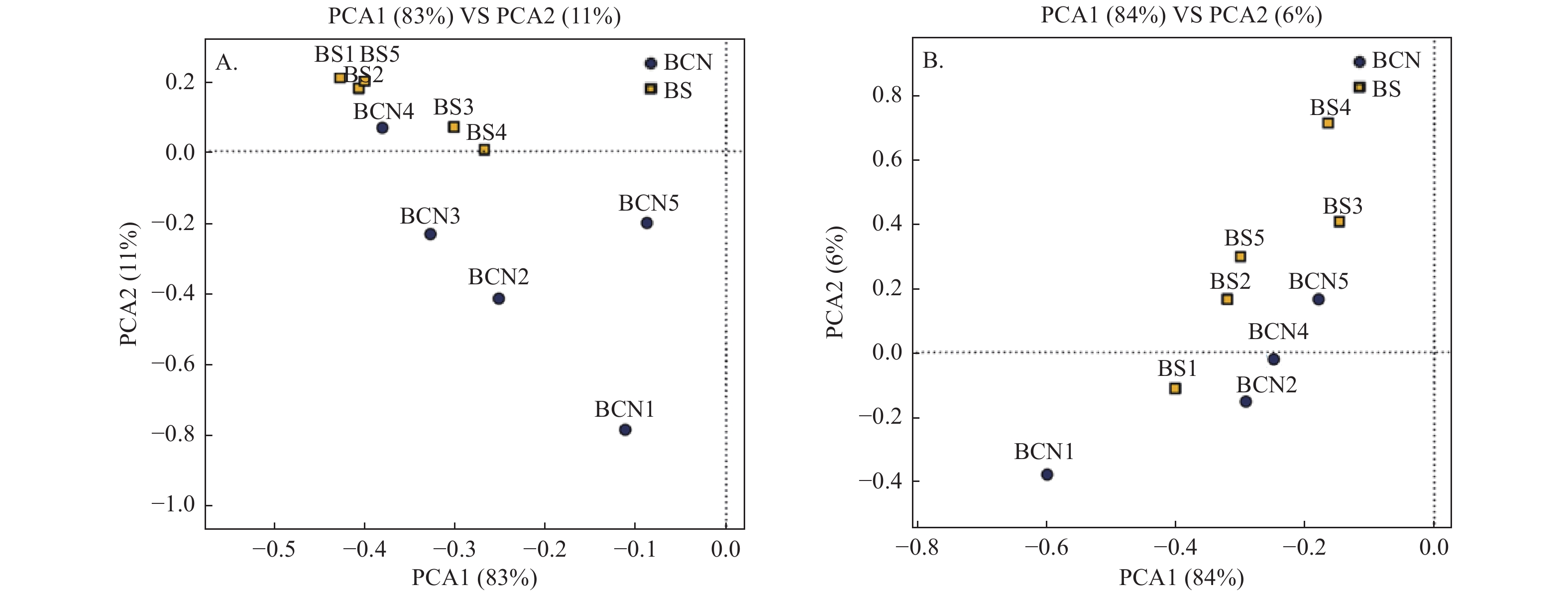
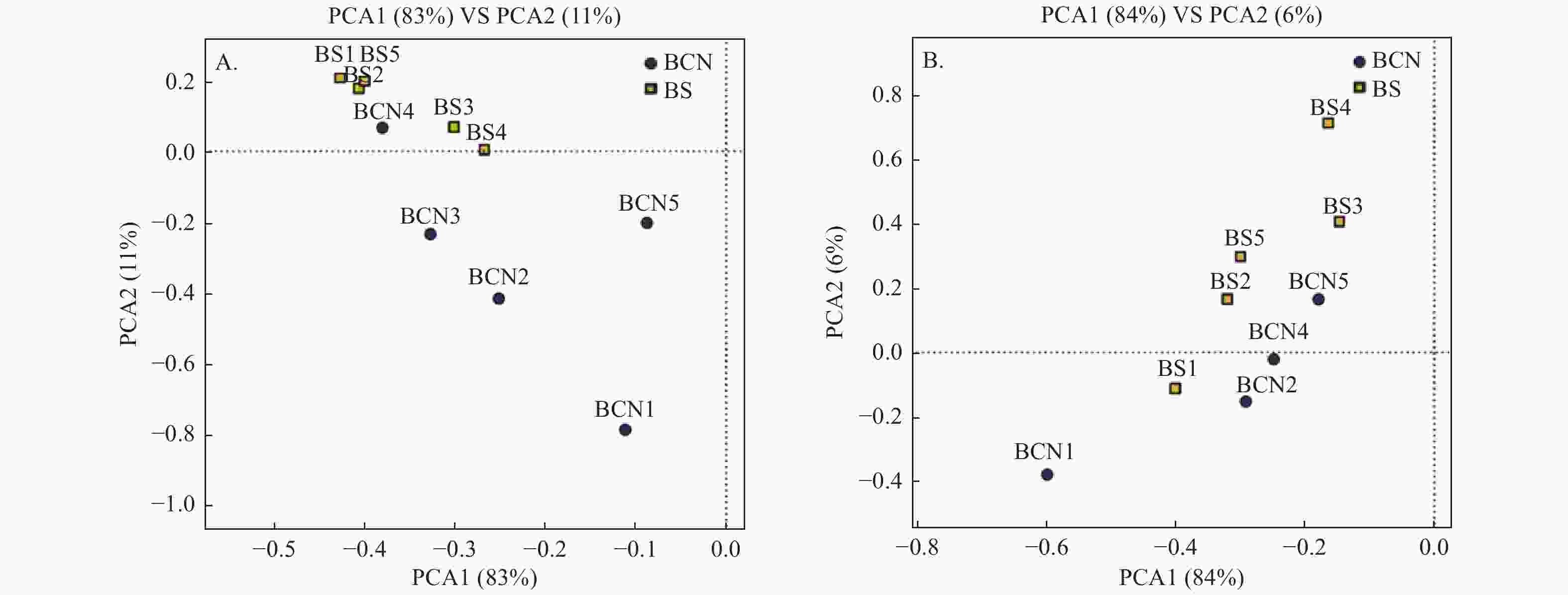
采用PCA法对BCN和BS样品的细菌和真菌群落进行主成分分析(图1A、1B)。从总体上看,BCN与BS样品大致分布在不同区域,表明BCN与BS样品间细菌、真菌的组成和结构存在较大差异。其中,细菌群落分析结果中有1组重复、真菌群落分析结果中有2组重复与其各自所属组别不在同一区域,可能是因为上述3组重复存在的偏差较大,从而导致PCA分析时出现误差,但还需做进一步验证。
-
在门水平上(图2A),BCN样品和BS样品的细菌均主要由变形菌门(Proteobacteria)组成,分别占(92.66%~99.65%)和(91.16%~97.56%),即变形菌门为两组样品的优势菌门。
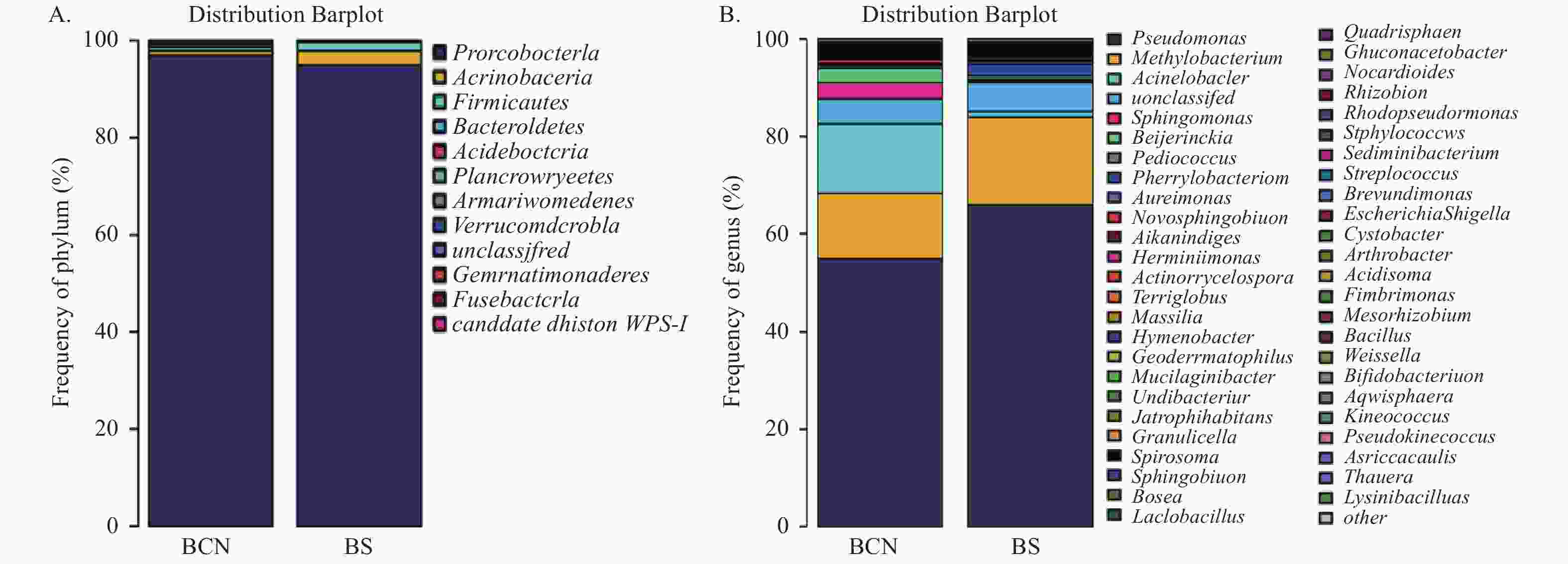
图 2 BCN和BS样品细菌在门(A)和属(B)水平的组成
Figure 2. Composition of bacteria in BCN and BS samples at the level of phylum (A) and genus (B)
在属水平上(图2B),BCN样品排名前五位的优势菌属分别为假单胞菌属(Pseudomonas)(12.24%~59.14%)、不动杆菌属(Acinetobacter)(6.37%~39.86%)、甲基杆菌属(Methylobacterium)(8.76%~39.11%)、拜叶林克氏菌属(Beijerinckia)(0.85%~18.53%)、鞘氨醇单胞菌属(Sphingomonas)(1.22%~12.09%);BS样品的优势菌属为假单胞菌属(52.10%~86.97%)和甲基杆菌属(4.42%~27.97%),而不动杆菌属(0.41%~1.66%)、拜叶林克氏菌属(0.2%~0.87%)和鞘氨醇单胞菌属(0.2%~0.62%)的占比较少。
-
在真菌门分类水平上(图3A),BCN和BS样品的优势菌门均为担子菌门(Ascomycota),分别占78.68%~93.44%和64.42%~91.55%。
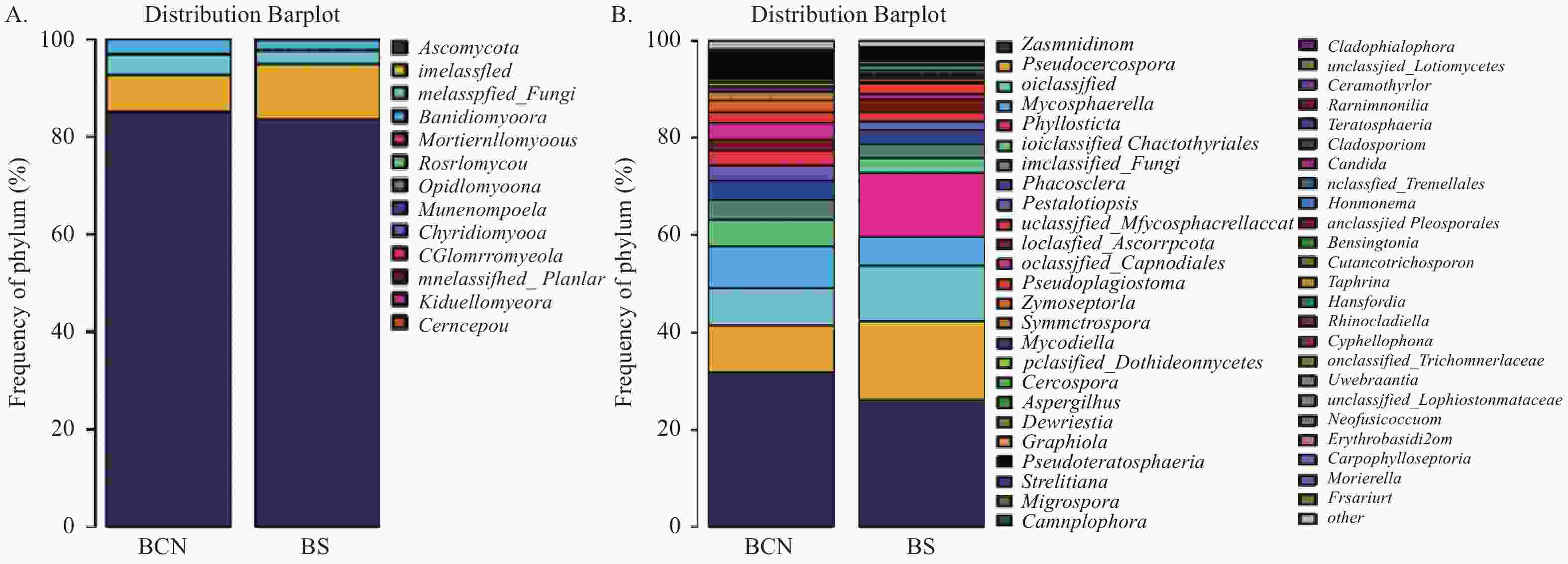
图 3 BCN和BS样品真菌在门(A)和属(B)水平的组成
Figure 3. Composition of BCN and BS samples at the phylum (A) and genus (B) levels
在属水平上(图3),BCN样品中排名前三位的优势菌属分别为平脐疣孢属(Zasmidium)(16.18%~60.82%)、假尾孢属(Pseudocercospora)(5.53%~16.58%)、球腔菌属(Mycosphaerella)(0.84%~10.47%),均为球腔菌科真菌;BS样品中排名前三位的优势菌属分别为平脐疣孢属(12.88%~40.68%)、假尾孢属(10.63%~28.59%)和叶点霉属(Phyllosticta)(4.06%~34.50%),球腔菌属(2.45%~12.33%)则排名第四位。
-
BCN和BS样品细菌菌群丰度的T-Test分析结果表明(图4),BCN样品中硝化杆菌属(Nitrobacter)和苯基杆菌属(Phenylobacterium)的丰度明显低于BS样品(P < 0.05),而Terriglobus菌属和鞘脂菌属(Sphingobium)的丰度显著高于BS样品(P < 0.05)。真菌菌群丰度的T-Test分析结果显示(图5),BCN样品中叉丝单囊壳属(Podosphaera)、Pseudoteratosphaeria属、Zymoseptoria属的丰度均明显高于BS样品(P < 0.05);而炭疽菌属(Colletotrichum)、节担菌属(Wallemia)、曲霉菌属(Aspergillus)、Graphiola属、黑孢属(Nigrospora)、青霉菌属(Penicillium)、梗孢酵母属(Sterigmatomyces)的丰度均明显低于BS样品(P < 0.05)。
-
采用T-Test分析法分别对BCN和BS样品中桉树主要病害病原菌所属菌属的丰度进行比较。结果显示(表3),BS样品中引起桉树炭疽病的炭疽菌属丰度明显高于BCN样品(P < 0.05)。而BCN和BS样品中引起青枯病的假单胞菌属(Pseudomonas)、引起叶斑病类的叶点霉属和假尾孢属(Pseudocercospora)、引起花斑病的短梗霉属(Aureobasidium)、引起紫斑病的壳针孢属(Septoria)、引起枝枯病的毛色二孢属(Lasiodiplodia)、引起梢枯病的新壳梭孢属(Neofusicoccum)的菌群丰度则无显著差异。
表 3 BCN和BS样品中桉树病害病原菌菌群丰度差异T-Test比较(属级水平)
Table 3. Comparison of abundance difference of Eucalyptus disease pathogenic bacteria in BCN and BS samples by T-Test (genus level)
菌属 Genus Freq1 Freq2 P值
P valueq值
q value效应量
Effect size95.0% 置信区间 95.0% CI 下限
Lower上限
UpperPseudomonas 40.534 71.146 0.071 1.000 −30.612 −64.790 3.566 Phyllosticta 0.081 14.517 0.059 1.000 −14.436 −29.774 0.903 Colletotrichum 0.005 0.020 0.046 1.000 −0.015 −0.029 0.000 Aureobasidium 0.000 0.009 0.115 1.000 −0.009 −0.021 0.003 Septoria 0.023 0.001 0.192 1.000 0.022 −0.017 0.062 Pseudocercospora 10.120 15.362 0.225 1.000 −5.242 −14.630 4.146 Lasiodiplodia 0.000 0.007 0.264 1.000 −0.007 −0.023 0.008 Neofusicoccum 0.083 0.033 0.267 0.990 0.050 −0.056 0.156 注:Freq1:桉树叶内菌属丰度;Freq2:油桐尺蛾中肠菌属丰度;q值:P值的校验值。
Note:Freq1: Abundance of bacteria or fungus in Eucalyptus leaves (genus level); Freq2: Abundance of bacteria or fungus inmidgut of Buzura suppressaria larvae (genera level); q value: The check value of the P value. -
本研究采用16s rDNA、ITS rDNA和Illumina Miseq技术分别对油桐尺蛾中肠和桉树叶部内生的细菌、真菌群落组成和结构进行分析。其中,油桐尺蛾肠道细菌共注释到6门、11纲、19目、38科、43属,真菌共注释到8门、29纲、67目、159科、257属;桉树叶部内生细菌共注释到10门、19纲、26目、54科、74属,真菌共注释到8门,31纲,79目,171科,291属。Alpha多样性分析表明,桉树叶部内生细菌的群落多样性和丰富度均高于油桐尺蛾肠道细菌。在森林生态系统中,植物与环境中的多种因素均具有交互作用,如植物与土壤、植物与昆虫、植物与植物之间等,这些复杂的交互作用形成了植物微生物的多样性。与植物相比,昆虫因活动区域的限制和肠道微环境条件的影响,体内的微生物种群相对单一。因此,尽管寄主植物是植食性昆虫肠道微生物的主要来源[15],但昆虫肠道微生物群落的多样性和丰富度均低于寄主植物。这一点在对BCN和BS样品的PCA分析中也得到证实,两组样品在细菌和真菌的组成和结构上存在较大差异。上述结果表明,桉树叶部微生物群落多样化与油桐尺蛾肠道微生物群落的相对单一化均是对各自生存环境和功能适应的结果。
在桉树叶部内生细菌和油桐尺蛾肠道细菌的组成中,变形菌门均占优势地位。变形菌门细菌广泛存在于自然界中,在土壤和动植物中均能检测到[16-18]。变形菌门也广泛存在于昆虫体内,研究表明变形菌门在舞毒蛾(Lymantria dispar)[19]、家蚕(Bombyx mori)[20]、小菜蛾[21]、棉铃虫[22]等多种鳞翅目昆虫幼虫的肠道中均为优势细菌。此外,变形菌门细菌的丰度在双翅目的泽兰实蝇(Procecidochares utilis)[23]、直翅目的沙漠蝗(Schistocerca gregaria)[24]、半翅目的点蜂缘椿象(Riptortus clavatus)[25]、鞘翅目的光肩星天牛(Anoplophora glabripennis)[26]等昆虫的肠道中也位居前列。上述研究表明,变形菌门细菌在以自然植物为食的昆虫中广泛存在,通过昆虫进食寄主植物进入至肠道并定殖,其丰度可能与昆虫所食植物上携带菌群的多少有关。在细菌属水平上,变形菌门中的假单孢菌属、甲基杆菌属均为BCN和BS样品中的优势细菌,其中又以假单胞菌的丰度最高。研究表明,假单胞菌能够通过固氮作用为宿主提供营养物质[27],在如克里角梢小蠹(Trypophloeus klimeschi Eggers)等以缺乏氮营养的植物为食的昆虫中,假单胞菌属可通过固氮作用为虫体提供更多的氮营养[28]。由此我们推测,油桐尺蛾肠道中高丰度的假单胞菌属细菌可能来自于所食的桉树叶片,假单胞菌属细菌在肠道中发挥的功能可能与营养代谢有关。此外,本研究还发现桉树叶部内生细菌和油桐尺蛾肠道细菌中的硝化杆菌属、苯基杆菌属、Terriglobus菌属、鞘脂菌属细菌的丰度存在显著差异,但上述细菌在样品中均占比较小,是否与寄主的生命活动相关也尚未明确,因此还需进一步探索研究。
担子菌门是桉树叶部和油桐尺蛾肠道的优势真菌,前人在对克里角梢小蠹[29]和致倦库蚊(Culex quinquefasciatus)[30]等的研究中也发现担子菌门在肠道真菌中排名前列。担子菌门是环境中的常见真菌,与部分昆虫存在共生关系,如担子菌门真菌中的Entomocorticium存在于松小蠹(Ips avulus)和食菌小蠹(Pityoborus comatus)的体表或体内,与上述小蠹虫所食植物有关,并与寄主形成共生关系[31]。因此我们推测,油桐尺蛾肠道中高丰度的担子菌门真菌可能来源自桉树叶,且作为肠道优势真菌可能在油桐尺蛾的生命活动中发挥重要作用。在属水平上,本研究发现平脐疣孢属、假尾孢属、球腔菌属均为桉树叶部和油桐尺蛾肠道的优势真菌。在非优势真菌中,叉丝单囊壳属、Pseudoteratosphaeria属、Zymoseptoria属、炭疽菌属、节担菌属、曲霉菌属、Graphiola属、黑孢属、青霉菌属、梗孢酵母属的丰度在两种样品中存在差异。
除此之外,我们发现在检测的细菌和真菌群落中存在多个桉树病害相关的病原菌属。其中,假单胞菌属是引起桉树青枯病的茄青枯极毛杆菌(Pseudomonas solanacearum)的所属菌属[32]。青枯病可引起桉树大面积枯萎坏死,是桉树常见的细菌病害[33]。尽管假单胞菌属为桉树叶的优势内生细菌,但并未在采集样品的林地中发现桉树感染青枯病的症状。同时,我们还在桉树叶和油桐尺蛾肠道中检测到了炭疽菌属真菌的存在。炭疽菌属的某些种类可以引起桉树炭疽病[34],本研究发现油桐尺蛾肠道中炭疽菌属真菌的丰度显著高于桉树叶,可能在油桐尺蛾啃食桉树叶的过程中或排出的粪便接触健康叶片时导致该真菌侵染桉树叶。叶点霉属真菌在油桐尺蛾肠道中属于优势菌属,而在桉树叶内则丰度较低。叶点霉属下的叶点霉菌(Phyllosticta lindericola)是引起桉树叶斑病的病原之一[35],但我们在林间调查时并未发现桉树感染叶斑病的情况。此外,我们还在桉树叶内和油桐尺蛾肠道中发现了其他可能引起桉树病害的病原菌属[36],包括引起桉树叶斑病的假尾孢菌(Pseudocercospora sp.)所属的假尾孢属,引起花斑病的出芽短梗霉菌(Aureobasidium pullulans)所属的短梗霉属,引起紫斑病的桉壳针孢菌(Septoria mortarlensis)所属的壳针孢属,引起枝枯病的可可毛色二孢菌(Lasiodiplodia theobromae)所属的毛色二孢属,引起梢枯病的新壳梭孢菌(Neofusicoccum parvum)所属的新壳梭孢属,这些菌属在样品中均非优势菌属,占比极小。尽管我们在采集的桉树叶和油桐尺蛾肠道样品中均检测到了上述病原菌相关菌属的存在,且其中的部分菌属的丰度也较高,但在林间并未发现相关病害症状。推测可能这些病原菌的丰度并未达到造成病害的条件,或检测到的菌属中并不存在病害相关的病原菌。此外,一些细菌或真菌对上述病原菌具有拮抗作用,如恶臭假单孢杆菌(P. putida)、荧光假单孢杆菌(P. Fluorescens)和铜绿假单孢杆菌(P. aeruginosa)产生的抗生素能拮抗茄青枯极毛杆菌的生长[37],这也可能是在林间并未发现相关病害的原因。但上述结论仅为推测,还需通过对样品中相关细菌、真菌进行分离培养做进一步鉴定。
-
本研究推测,生存环境和功能适应是导致桉树叶部与油桐尺蛾肠道中细菌、真菌群落存在较大差异的原因。同时,油桐尺蛾肠道中的细菌和真菌的群落结构也受到所食桉树叶的影响,而携带某些病原菌的油桐尺蛾啃食或其排泄物接触可能是导致该病原菌侵染健康桉树叶的原因。
油桐尺蛾肠道菌群与桉树叶片内生菌差异分析
Comparison and Analysis of Diversified Intestinal Flora of Buzura suppressaria Larvae and Endophytes in Eucalyptus Leaves
-
摘要:
目的 植食性昆虫肠道微生物的多样性与其食物密切相关,本研究通过探讨油桐尺蛾(Buzura suppressaria)幼虫肠道菌群及其取食的桉树叶内生菌群的多样性,分析两者之间的关系。 方法 利用Illumina Miseq技术对油桐尺蛾5龄幼虫肠道和桉树叶部内生的细菌16s rDNA和真菌ITS rDNA进行测序,分析油桐尺蛾肠道和桉树叶部内生的细菌和真菌的多样性和丰度。 结果 油桐尺蛾肠道细菌共注释到6门、11纲、19目、38科、43属,真菌共注释到8门、29纲、67目、159科、257属;桉树叶部内生细菌共注释到10门、19纲、26目、54科、74属,真菌共注释到8门,31纲,79目,171科,291属。桉树叶内和油桐尺蛾肠道中均发现桉树主要病害病原菌所属菌属,分别为炭疽菌属(Colletotrichum)、假单胞菌属(Pseudomonas)、假尾孢属(Pseudocercospora)、短梗霉属(Aureobasidium)、壳针孢属(Septoria)、毛色二孢属(Lasiodiplodia)、新壳梭孢属(Neofusicoccum)。 结论 油桐尺蛾肠道的细菌、真菌的多样性和丰度与桉树叶之间存在差异,桉树叶对油桐尺蛾肠道微生物的群落结构有重要影响,而携带某些病原菌的油桐尺蛾啃食或其排泄物接触可能是导致该病原菌侵染健康桉树叶的原因。 Abstract:Objective This study is to investigate the diversified intestinal flora of Buzura suppressaria larvae and Eucalyptus leaves which are the major food of B. suppressaria, and to analyze the inner relationship between them. Method Illumina Miseq technology was applied to sequence the bacteria 16S rDNA and fungi ITS rDNA in the intestinal of the 5th instar larvae and Eucalyptus leaves. Both the diversity and abundance of the bacteria and fungi in the intestinal and Eucalyptus leaves were analyzed. Result The results of bioinformatics analysis showed that the bacteria populations in the intestinal tract of B. Suppressaria larvae were annotated into 6 phyla, 11 classes, 19 orders, 38 families and 43 genus, whereas the fungi populations were categorized into 8 phyla, 29 classes, 67 orders, 159 families and 257 genus. The Endophytic bacteria found in Eucalyptus leaves included 10 phyla, 19 classes, 26 orders, 54 families and 74 genus, and the fungi included 8 phyla, 31 classes, 79 orders, 171 families and 291 genus. The main pathogenic bacteria of Eucalyptus were found in both of the Eucalyptus leaves and the larvae intestinal tract, including the orders of Colletotrichum, Pseudomonas, Pseudocercospora, Aureobasidium, Septoria, Lasiodiplodia and Neofusicoccum. Conclusion The diversity and abundance of bacteria and fungi populations in the intestinal tract of B. suppressaria larvae were different from those of the Eucalyptus leaves, which had an important influence on the intestinal microbial community structure. The gnawing or fecal contact of B. suppressaria larvae carrying certain pathogens may be the cause of the pathogen infecting healthy Eucalyptus leaves. -
Key words:
- Buzura suppressaria
- / Eucalyptus
- / intestinal flora
- / endophyte
- / microbial diversity
- / high-throughput sequencing
-
表 1 BCN和BS样品OTU聚类及RDP分类水平统计
Table 1. OTU clustering and RDP classification level statistics of BCN and BS samples
样品
Sample检测项目
Test items有效reads数目/条
Reads numOTU数目/个
OUT numRDP分类
Classification of RDP门
Phylum纲
Class目
Order科
Family属
GenusBS 细菌 Bacteria 3 271 199 6 11 19 38 43 真菌 Fungus 280 392 4 279 8 29 67 159 257 BCN 细菌 Bacteria 11 413 379 10 19 26 54 74 真菌 Fungus 287 330 4 236 8 31 79 171 291 表 2 BCN和BS样品α多样性指数统计
Table 2. Alpha diversity index statistics of BCN and BS samples
样品
Sample检测项目
Test itemsOTU数量
OUT numSimpson指数
Simpson indexShannon指数
Shannon indexACE指数
ACE indexChao1指数
Chao1 index覆盖率
coverageBS 细菌 Bacteria 39.80 ± 14.82 0.30 ± 0.10 1.90 ± 0.43 61.37 ± 24.07 53.55 ± 14.68 0.97 ± 0.01 真菌 Fungus 855.80 ± 168.88 0.10 ± 0.05 3.60 ± 0.47 1 098.59 ± 164.90 1 058.26 ± 156.59 1.0 ± 0.00 BCN 细菌 Bacteria 75.80 ± 32.71 0.21 ± 0.07 2.33 ± 0.57 109.82 ± 33.31a 104.12 ± 38.89a 0.98 ± 0.01 真菌 Fungus 847.20 ± 142.69 0.13 ± 0.13 3.50 ± 0.71 1 167.82 ± 103.06 1 094.02 ± 147.21 1.0 ± 0.00 注:a为与BS样品细菌检测组相比,P < 0.05
Note: P is compared with the BS samples in the bacterial detection group, P < 0.05表 3 BCN和BS样品中桉树病害病原菌菌群丰度差异T-Test比较(属级水平)
Table 3. Comparison of abundance difference of Eucalyptus disease pathogenic bacteria in BCN and BS samples by T-Test (genus level)
菌属 Genus Freq1 Freq2 P值
P valueq值
q value效应量
Effect size95.0% 置信区间 95.0% CI 下限
Lower上限
UpperPseudomonas 40.534 71.146 0.071 1.000 −30.612 −64.790 3.566 Phyllosticta 0.081 14.517 0.059 1.000 −14.436 −29.774 0.903 Colletotrichum 0.005 0.020 0.046 1.000 −0.015 −0.029 0.000 Aureobasidium 0.000 0.009 0.115 1.000 −0.009 −0.021 0.003 Septoria 0.023 0.001 0.192 1.000 0.022 −0.017 0.062 Pseudocercospora 10.120 15.362 0.225 1.000 −5.242 −14.630 4.146 Lasiodiplodia 0.000 0.007 0.264 1.000 −0.007 −0.023 0.008 Neofusicoccum 0.083 0.033 0.267 0.990 0.050 −0.056 0.156 注:Freq1:桉树叶内菌属丰度;Freq2:油桐尺蛾中肠菌属丰度;q值:P值的校验值。
Note:Freq1: Abundance of bacteria or fungus in Eucalyptus leaves (genus level); Freq2: Abundance of bacteria or fungus inmidgut of Buzura suppressaria larvae (genera level); q value: The check value of the P value. -
[1] 陈勃生, 鲁兴萌, 邵勇奇. 鳞翅目昆虫肠道微生物的多样性及其与宿主的相互作用[J]. 昆虫学报, 2017, 60(6):710-722. [2] Douglas A E. The microbial dimension in insect nutritional ecology[J]. Functional Ecology, 2010, 23(1): 38-47. [3] 梅 承, 范 硕, 杨 红. 昆虫肠道微生物分离培养策略及研究进展[J]. 微生物学报, 2018, 58(6):985-994. [4] Priya N G, Ojha A, Kajla M K, et al. Host plant induced variation in gut bacteria of Helicoverpaarmigera[J]. PLoS ONE, 2012, 7(1): e30768. doi: 10.1371/journal.pone.0030768 [5] Robinson C J, Schloss P, Ramos Y, et al. Robustness of the bacterial bommunity in the Cabbage white butterfly larval midgut[J]. Microbial Ecology, 2010, 59(2): 199-211. doi: 10.1007/s00248-009-9595-8 [6] 吴晓露, 夏晓峰, 陈俊晖, 等. 取食不同食物对小菜蛾幼虫肠道细菌多样性的影响[J]. 昆虫学报, 2019, 62(10):1172-1185. [7] Strano C P, Antonino M, Campolo O, et al. Influence of host plant on Thaumetopoea pityocampa gut bacterial community[J]. Microbial Ecology, 2018, 75(2): 487-494. doi: 10.1007/s00248-017-1019-6 [8] Dillard H R, Cobb A C. Relationship between leaf injury and colonization of cabbage by Sclerotinia sclerotiorum[J]. Crop Protection, 1995, 14(8): 677-682. doi: 10.1016/0261-2194(95)00039-9 [9] Telsa W, Syama C, Cárcamo H. Interactions of Root-feeding insects with fungal and oomycete plant pathogens[J]. Front Plant, 2017, 8: 1764. doi: 10.3389/fpls.2017.01764 [10] Polenogova O V, Kabilov M R, Tyurin M V, et al. Parasitoid envenomation alters the Galleria mellonella midgut microbiota and immunity, thereby promoting fungal infection[J]. Scientific Reports, 2019, 9(1): 4012. doi: 10.1038/s41598-019-40301-6 [11] 廖旺姣, 盛 双, 常明山, 等. 油桐尺蠖幼虫对3种桉树无性系的取食选择性初探[J]. 广西林业科学, 2011, 40(4):328-329. [12] 孙艳娟, 韦金英, 杨振德, 等. 油桐尺蠖幼虫龄期的划分[J]. 应用昆虫学报, 2009, 46(6):892-895. [13] 刘有莲, 黄寿昌, 牙 璋, 等. 油桐尺蠖越冬蛹在桉树林空间分布型及其生物学特性研究[J]. 中国森林病虫, 2018, 37(2):14-17. [14] 钟雅婷, 罗 辑, 蒋学建, 等. BsNPV和BmNPV重组毒株生物学特性[J]. 森林与环境学报, 2018, 38(4):494-498. [15] Pinto-Tomás Adrián A, Ana S, Uribe-Lorío Lorena, et al. Comparison of midgut bacterial diversity in Tropical caterpillars (Lepidoptera: Saturniidae) fed on different diets[J]. Environmental Entomology, 2011(5): 1111-1122. [16] 彭 雯, 谭 玲, 明安刚, 等. 南亚热带典型人工纯林土壤剖面细菌群落组成差异分析[J]. 土壤通报, 2018, 49(6):1361-1369. [17] 郭玉倩, 角建林, 袁 鑫, 等. 树鼩大肠内容物细菌的群落多样性分析[J]. 中国比较医学杂志, 2018, 28(9):7-13, 32. [18] Haldar S, Sengupta S. Impact of plant development on the rhizobacterial population of Arachishypogaea: a multifactorial analysis[J]. Journal of Basic Microbiology, 2015, 55(7): 922-928. doi: 10.1002/jobm.201400683 [19] Zeng J Y, Shi Z B, Shi J H, et al. Ambient temperature‐mediated enzymic activities and intestinal microflora in Lymantria dispar larvae[J]. Archives of Insect Biochemistry and Physiology, 2019, 102(2): e21597. [20] Kumar D, Sun Z, Cao G, et al. Bombyx moribidensovirus infection alters the intestinal microflora of fifth instar silkworm (Bombyx mori) larvae[J]. Journal of Invertebrate Pathology, 2019, 163: 48-63. doi: 10.1016/j.jip.2019.03.004 [21] Lin X L, Pan Q J, Tian H G, et al. Bacteria abundance and diversity of different life stages of Plutella xylostella (Lepidoptera: Plutellidae), revealed by bacteria culture-dependent and PCR-DGGE methods[J]. Insect Sci, 2015, 22(3): 375-385. doi: 10.1111/1744-7917.12079 [22] Ranjith M T, Mani C, Harish E R, et al. Bacterial communities associated with the gut of tomato fruit borer, Helicoverpaarmigera (Hübner) (Lepidoptera: Noctuidae) based on Illumina Next-Generation Sequencing[J]. Journal of Asia-Pacific Entomology, 2016: 333-340. [23] 张 某, 杨 璞, 朱家颖, 等. 基于16SrDNA基因序列的泽兰实蝇幼虫肠道细菌多样性分析[J]. 昆虫学报, 2016,59(2):200-208. [24] Dillon R J, Webster G, Weightman A J, et al. Diversity of gut microbiota increases with aging and starvation in the desert locust[J]. Antonie Van Leeuwenhoek, 2010, 97(1): 69-77. doi: 10.1007/s10482-009-9389-5 [25] Kikuchi Y, Meng X Y, Fukatsu T. Gut symbiotic bacteria of the genus Burkholderia in the broad-headed bugs Riptortus clavatus and Leptocorisa chinensis (Heteroptera: Alydidae)[J]. Appl Environ Microbiol, 2005, 71(7): 4035-4043. doi: 10.1128/AEM.71.7.4035-4043.2005 [26] Schloss P D, Italo D, Jo H, et al. Bacteria associated with the guts of two wood-boring beetles: Anoplophora glabripennis and Saperda vestita(Cerambycidae)[J]. Environmental Entomology, 2006(3): 625-629. [27] Desnoues, Nicole, M in, et al. Nitrogen fixation genetics and regulation in a Pseudomonas stutzeri strain associated with rice[J]. Microbiology (13500872), 2003, 149(8): 2251-2262. doi: 10.1099/mic.0.26270-0 [28] Gao G, Dai L, Gao J, et al. Electroantennogram, behavioural responses, and field trapping of Trypophloeus klimeschi (Coleoptera: Curculionidae: Scolytinae) to eight host volatiles[J]. The Canadian Entomologist, 2019: 1-15. [29] 高冠群. 克里角梢小蠹发生规律与寄主选择机制的研究[D]. 杨凌, 西北农林科技大学, 2018 [30] Bawin T, Seye F, Boukraa S, et al. Histopathological effects of Aspergillusclavatus (Ascomycota: Trichocomaceae) on larvae of the southern house mosquito, Culex quinquefasciatus (Diptera: Culicidae)[J]. Fungal Biol, 2016, 120(4): 489-499. doi: 10.1016/j.funbio.2016.01.002 [31] 胡 霞. 华山松大小蠹肠道微生物群落多样性与幼虫肠道纤维素降解菌的研究[D]. 杨凌, 西北农林科技大学, 2014. [32] 林绪平, 林雪坚. 桉树青枯病的流行规律研究[J]. 中南林业科技大学学报, 1996(3):49-55. [33] 向妙莲, 冉隆贤. 桉树青枯病研究进展[J]. 中国森林病虫, 2004, 23(1):37-40. [34] 张国辉, 宋盛英, 刘德波, 等. 桉树褐斑病和炭疽病病原鉴定与病害分析[J]. 中国植保导刊, 2014, 34(12):10-13. [35] 郝海坤, 曹艳云, 黄志玲, 等. 桉树扦插苗主要病虫害及其防治技术[J]. 林业科技开发, 2007(4):102-103. [36] 罗基同, 薛振南, 廖旺姣, 等. 广西速生丰产桉树病害调查[J]. 中国森林病虫, 2012(4):21-24. [37] 吴志华, 谢耀坚, 罗联峰, 等. 我国桉树青枯病研究进展[J]. 林业科学研究, 2007, 20(4):569-575. -




 下载:
下载: