-
桉树焦枯病是由丽赤壳属(Calonectria spp.)真菌引起的世界性病害,主要发生在桉树幼苗和4年生以下的人工幼林[1-2]。该病原菌可侵染桉树幼茎、叶片、枝杆、树梢等多个部位,使苗木猝倒、根腐,叶片焦枯、卷曲脱落,枝条枯死,造成桉苗腐烂病以及生长后期的焦枯、顶枯、叶枯病。在我国广东、广西、海南、福建、台湾等桉树苗圃和人工林都有不同程度的发生,每次病害流行均导致大面积桉树林枯萎死亡,造成桉树人工林生产量明显下降[3-4]。据估计,福建省因该病害造成的年经济损失达0.518亿元[5]。桉树具有丰富的遗传资源,不同品系对焦枯病菌的抗性存在明显差异[6-8]。
苯丙烷代谢途径是植物体中一条重要的代谢途径[9-10]。苯丙氨酸解氨酶(PAL)是苯丙烷类代谢的关键酶和限速酶,主要负责催化L-苯丙氨酸脱氨形成反式肉桂酸,后者是植物合成木质素、植保素、黄酮、类黄酮等多种酚类次级代谢产物的前体物质,这些次级产物在调节植物生长发育及提高抗逆性中起着重要作用。目前,已从植物中克隆到多个PAL基因并研究了相应功能[11-12]。大多数植物PAL基因的编码序列长度约为2 100 bp,包含2个外显子和1个内含子,其中,外显子长度分别约为400、1 700 bp,内含子的位置相对保守,但长度差异很大。植物PAL蛋白一般含有PAL-HAL、PLN02457及phe_am_lyase三个保守结构域,其中,酶活性位点在PAL-HAL结构域上。植物PAL活性与其抗病性密切相关[13],在病原物侵染后,PAL活性往往呈现出先升高后下降的趋势,且抗病品系PAL活性高于感病品系[14]。水稻感染稻瘟病、小麦感染白粉病和赤霉病、玉米感染黑穗病、锥栗感染疫病后的PAL活性均出现上述变化趋势[15-19]。桉树PAL基因较少研究,PAL基因对桉树抗焦枯病菌的作用也未见报道。本研究从尾细桉M1基因组中克隆到1个PAL基因,并进行了生物信息学分析,探讨了接种Ca. pseudoreteaudii后不同品系PAL基因的表达特征,为进一步研究PAL基因在桉树抵抗焦枯病菌侵染的作用机制奠定基础。
-
抗病品系尾细桉M1(Eucalyptus urophylla × E. tereticornis M1)和中感品系尾巨桉3229号(E. urophylla × E. grandis 3229)、感病品系巨桉5号(E. grandis 5)扦插苗及桉树焦枯病菌(Calonectria pseudoreteaudii YA51)由福建农林大学森林保护研究所提供[6]。质粒pDoner207由福建农林大学功能基因组研究中心惠赠。孢子悬浮液的配制参照Feng等[20]的方法。
将15棵桉树扦插苗(苗龄3个月,苗高25 cm)栽植于带土花盆,并置于人工气候箱(昼夜温度分别为25℃和23℃,相对湿度90%,光暗时间比14/10)中适应性培养7 d后,用75%酒精消毒,再用ddH2O冲洗23遍,将浓度为2.0×105 个·mL-1的分生孢子悬浮液均匀地喷洒于植株上,再置于人工气候箱保湿培养。分别于接种后0、12、24、48、72 h采集叶片,每个时间点从不同幼苗上采集的叶片混合在一起,液氮速冻后-80℃冰箱保存备用。
-
按照Plant Genomic DNA Kit(TIANGEN)的说明书提取尾细桉M1的基因组DNA。
-
采用改良CTAB法提取桉树总RNA [21]。检测合格后,以总RNA为模板,按照ScriptTM RT reagent Kit with gDNA Eraser (TaKaRa)说明书合成cDNA,置于-20℃备用。
-
根据巨桉(E. grandis)PAL基因(Eucgr.J01079.1)cDNA序列在其可能的开放阅读框两端设计引物(表 1)。以尾细桉M1的基因组DNA为模板,分两段克隆PAL基因。反应程序:95℃预变性3 min;95℃变性15 s,60℃退火15 s,72℃延伸1 min,30个循环;72℃保温5 min,4℃保存。琼脂糖凝胶电泳检测PCR产物,并回收目的片段。采用重叠延伸扩增技术(SOE)连接两个目的片段。第一步反应体系:10× LA Buffer 5 μL,dNTP mix 2 μL,Templax 1 1 μL,Templax 2 0.5 μL,LA Tag 0.25 μL,ddH2O补足48 μL,反应程序:95℃预变性3 min;94℃变性30 s,64℃退火1 min,72℃延伸1 min,10个循环;72℃保温7 min,4℃保存。第二步反应体系:第一步反应产物48 μL,引物F1 1 μL,R2 1 μL,反应条件:94℃预变性4 min;94℃变性40 s,60℃退火40 s,72℃延伸2 min,30个循环;72℃保温7 min,4℃保存。电泳检测目的条带,回收纯化后连接到pDoner207,并用Gateway技术进行BP反应,转化DH5α感受态细胞,菌液PCR鉴定为阳性克隆后,送至上海华大基因生物技术有限公司测序。
表 1 基因克隆及荧光定量PCR引物
Table 1. Primers used for cloning and real-time PCR
基因名称
Gene name用途
Function序列
Primer sequence (5’-3’)片段长度
Length of fragment/bpPAL Clone F1:ATGGAGATGGAGAGCACC
R1:CTTATAAGTTCCTTCTGCAAAG398 PAL Clone F2:TTGCAGAAGGAACTTATAAGGTTCTTGAATGCCGGGAT
R2:CTAAGAGATGGGAAGAGGAG1 750 PAL RT-PCR F3:TCGAGTCCAGTACGAGACAGG
R3:CTAAGAGATGGGAAGAGGAGCAC241 Actin RT-PCR F:TTCTACTATGTTCCCAGGTATCGC
R:GACCAGATTCATCATACTCACCCT194 -
利用ORF finder预测基因的开放阅读框(ORF)和编码序列;采用NCBI的Blast工具分析核苷酸和氨基酸序列相似性;利用ProtParam(http://expasy.org/tools/protparam.html)在线分析氨基酸序列的理化性质;采用ProtScale分析蛋白质的疏水性/亲水性;采用NetPhos分析蛋白的磷酸化位点;利用SOPMA分析蛋白的二级结构;利用MEGA7.0软件及邻接(NJ)法构建进化树。
-
以Actin(Eucgr.I00241.1)为内参基因,根据SYBR Premix Ex TaqTM(Tli RNaseH Plus)(TaKaRa)的操作说明,在荧光定量PCR仪(Eppendorf)上分析不同桉树品系PAL基因在焦枯病菌胁迫下的表达情况。反应体系:1 μL cDNA,0.3 μL F3,0.3 μL R3,5 μL SYBR Premix ExTaq,ddH2O补足10 μL。反应条件:95℃ 2 min;95℃ 30 s,60℃ 15 s,68℃ 20 s,40 cycles。每个样品技术重复3次,以0 h为对照,以2-△△CT方法分析相对表达量。
-
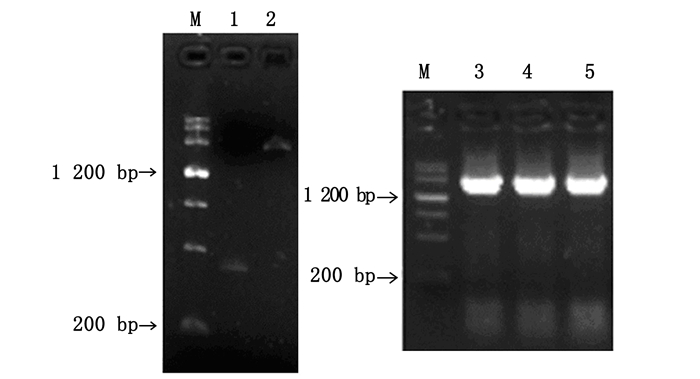
根据巨桉PAL基因片段序列,设计特异性引物,获得长度分别约为400、1 800 bp的目的条带,SOE连接获得2 200 bp左右的条带(图 1)。对随机取得的3个重组质粒进行双向测序,得到长度为2 350 bp的基因序列,其中,含1个长达2 142 bp的ORF,推测编码713个氨基酸,含有苯丙氨酸和组氨酸解氨酶保守结构(序列第195211位)(图 2)。
-
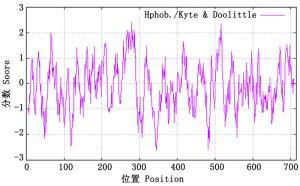
ProtParam分析表明:尾细桉PAL蛋白的理论分子量为76 942.79 Da,等电点(pI)为6.02,分子式为C3396H5441N949O1036S25,负电荷氨基酸残基总数(Asp + Glu)为78,正电荷氨基酸残基总数(Arg + Lys)为67,不稳定系数为34.04,是一类稳定蛋白。脂溶性指数为94.29,总平均疏水性为-0.099,预测为亲水性蛋白。ProtScale进一步分析PAL蛋白的疏水曲线(大于0为疏水,小于0为亲水)(图 3),PAL蛋白序列存在明显的疏水区和亲水区,其中,第279位最高,为2.422,第345位最低,为-2.656,因此,PAL蛋白为亲水性蛋白。
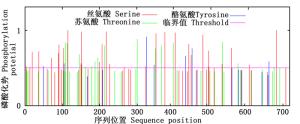
NetPhos分析(图 4)表明:PAL蛋白存在丝氨酸、苏氨酸和酪氨酸3个磷酸化位点,其中,丝氨酸磷酸化位点最多,有29个,其次是苏氨酸磷酸化位点16个,酪氨酸磷酸化位点最少,仅7个。
SOPMA分析(图 5)发现:共有387个氨基酸参与形成α-螺旋,占总氨基酸的54.28%,235个氨基酸参与形成无规则卷曲,占总氨基酸的32.96%。另外,有45个氨基酸参与形成延伸链(6.31%),46个氨基酸参与形成β-转角(6.45%),表明尾细桉M1 PAL蛋白的二级结构以α-螺旋和无规卷曲为主。
-
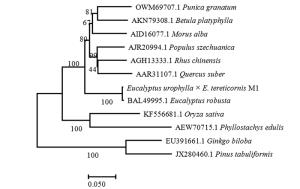
利用NCBI的BLAST进行序列比对,结果表明:尾细桉PAL基因的核苷酸序列与大叶桉(E. robusta,ID: AB696677.1)的相似性为99.39%以上,此外与桑树(Morus alba,ID: KX387372.1)和石榴(Punica granatum,ID: KY094504.2)等的相似性也较高[22-24]。该基因编码的氨基酸序列与大叶桉(E. robusta,ID: BAL49995.1)的相似性最高,达到99.30%,与桑树(Morus alba,ID: AID16077.1)、石榴(Punica granatum,ID: OWM69707.1)、漆树(Rhus chinensis,AGH13333.1)和川杨(Populus szechuanica,AJR20994.1)相似性也较高,均在84%以上[25-26]。以尾细桉M1及其他11植物的PAL蛋白序列为基础,构建系统进化分析,结果(图 6)表明:尾细桉M1的PAL蛋白序列与大叶桉亲缘关系最近,单独为一小分支,再与桑树、石榴、漆树和川杨等其他双子叶植物聚为一类,单子叶植物水稻和毛竹聚为一类,裸子植物银杏和油松聚为一类。
-
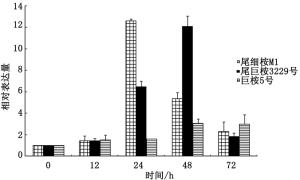
对桉树焦枯病菌侵染下的抗病品系尾细桉M1、中感品系尾巨桉3229号和感病品系巨桉5号等叶片的表达情况进行分析。结果(图 7)表明:以0 h为对照,随着侵染时间的推移,3个桉树品系的PAL基因均呈上调表达趋势,但抗病品系的PAL基因表达量于24 h就出现最高点,中感品系和感病品系则在48 h出现高峰,且表达高峰的水平呈现为抗病品系>中感品系>感病品系。说明PAL基因的早表达、高表达可能在桉树抵御焦枯病菌侵染的过程中起着重要作用。
-
PAL是调节和控制植物体苯丙烷类代谢途径的关键酶,在植物的生长发育、抗逆及抗病反应中起重要作用[27-29]。甜椒PAL沉默后,该突变体对病原菌的敏感性增加,而过表达CaPAL1基因则提高拟南芥对病原菌的抗性[30]。大豆GmPAL敲除突变体SA的合成减少,对病原菌的抗性随之减弱[31]。一般认为,植物受病原菌侵染后,PAL基因上调表达,从而合成一些次级代谢产物来提高自身的抗病能力,如大量积累木质素以强化细胞壁,产生植保素抑制病原菌的生长,合成水杨酸(SA)以诱发系统获得性抗性(SAR),进而产生一种更持久的广谱抗性反应[32-33]。因此,PAL基因在植物抗病反应中发挥着重要角色。
本研究通过SOE-PCR技术从尾细桉M1中克隆到一个PAL基因,其开放阅读框长2 142 bp,编码一个具有713个氨基酸的亲水性蛋白,该蛋白具有典型的苯丙氨酸和组氨酸解氨酶保守结构域、PLN02457特异结合位点和PLN02457超家族保守结构域。进而分析桉树不同品系PAL基因的表达趋势,结果发现,不同品系PAL基因在焦枯病菌诱导下均表现为明显的上调表达,且抗性越强,PAL基因越早上调,上调表达量越大。该结果进一步佐证了笔者的前期研究,即焦枯病菌入侵后,桉树多酚类物质和黄酮类化合物含量增加,SOD及POD等防御酶活性也会提高,且抗病品系的增加水平均超过中感品系和感病品系[34-35]。综合PAL在其他植物抗病中的研究结果,推断桉树PAL可能通过促进植保素、木质素及酚类化合物等防御性物质的合成来提高不同品系的抗病能力。此外,PAL也可能通过合成信号分子SA,启动SAR反应,进而增强桉树对焦枯病菌的抵抗能力,但其具体的作用机制还有待进一步的研究。
-
从尾细桉M1中获得1个PAL基因,其编码的蛋白序列具有典型的PAL家族成员特征。根据其在焦枯病菌诱导下的表达情况推测,该基因可能通过参与酚类防御性物质或者信号分子SA等物质的合成,进而在桉树抵抗焦枯病菌侵染的过程中发挥重要作用。
桉树PAL基因克隆及焦枯病菌诱导下的表达分析
Cloning of PAL Gene from Eucalyptus and Its Expression under Calonectria pseudoreteaudii Stress
-
摘要:
目的 克隆桉树苯丙氨酸解氨酶基因(PAL),分析其序列特征及在焦枯病菌诱导下的表达特征,为探究桉树抗焦枯病机理提供理论支撑。 方法 通过SOE-PCR技术从尾细桉M1中克隆PAL基因,利用real-time PCR分析其在焦枯病菌诱导下的表达特性。 结果 克隆得到尾细桉M1 PAL基因,其ORF序列长2 142 bp,编码713个氨基酸,与其他已发表的桉树PAL相似性均在99%以上。序列分析发现,其编码蛋白是一类稳定的亲水性蛋白,含有丝氨酸、苏氨酸和酪氨酸3个磷酸化位点,二级结构以α-螺旋和β-折叠为主,具有典型的苯丙氨酸和组氨酸解氨酶保守结构域。进一步分析桉树不同品系PAL基因的表达趋势,结果发现,不同品系PAL基因在焦枯病菌诱导下均表现为明显的上调表达,且抗性越强,PAL基因越早上调,上调表达量越大。 结论 从尾细桉M1中克隆得到的PAL基因具有典型的PAL家族成员特征,推测该基因可能通过参与酚类防御性物质或者信号分子SA等物质的合成,进而在桉树抵抗焦枯病菌侵染的过程中发挥重要作用。 Abstract:Objective This study aimed at cloning the cDNA sequence of Eucalyptus phenylalanin ammonia-lyase (PAL) gene, analyzing its characteristics, and investigating its expressions in Eucalyptus under the infection of Calonectria pseudoreteaudii. Method SOE-PCR technology was used to clone PAL gene from E. urophylla×E. tereticornis M1. The expression level of PAL gene in different Eucalyptus cultivars infected with C. pseudoreteaudii were analyzed by RT-PCR. Result One PAL gene was cloned. Its ORF sequence was 2 142 bp, and encoded 713 amino acids, which showed high similarity to the PAL from other Eucalyptus, with 99% identity. Amino acids sequence analysis indicated that the predicted PAL protein was a stable hydrophilic protein, including three phosphorylation sites (serine, threonine and tyrosine). Its secondary structure was rich in α-helix and random coil. The predicted protein contained phenylalanine and histidine ammonia-lyase conserved structure. The results showed that the PAL gene was significantly up-regulated in all tested cultivars. Moreover, the PAL gene was earlier and more significantly up-regulated in the stronger resistant cultivars. Conclusion The PAL gene which was cloned from E. urophylla×E. tereticornis M1 has typical characteristics of PAL family. It is speculated that PAL gene may participant in the synthesis of phenolic compounds or signal molecule salicylic acid, then improves the resistance of Eucalyptus to C. pseudoreteaudii. -
Key words:
- Eucalyptus
- / phenylalanine ammonia-lyase
- / SOE-PCR
- / bioinformatics
- / Calonectria pseudoreteaudii
-
表 1 基因克隆及荧光定量PCR引物
Table 1. Primers used for cloning and real-time PCR
基因名称
Gene name用途
Function序列
Primer sequence (5’-3’)片段长度
Length of fragment/bpPAL Clone F1:ATGGAGATGGAGAGCACC
R1:CTTATAAGTTCCTTCTGCAAAG398 PAL Clone F2:TTGCAGAAGGAACTTATAAGGTTCTTGAATGCCGGGAT
R2:CTAAGAGATGGGAAGAGGAG1 750 PAL RT-PCR F3:TCGAGTCCAGTACGAGACAGG
R3:CTAAGAGATGGGAAGAGGAGCAC241 Actin RT-PCR F:TTCTACTATGTTCCCAGGTATCGC
R:GACCAGATTCATCATACTCACCCT194 -
[1] Lombard L, Crous P W, Wingfield B D, et al. Species concepts in Calonectria (Cylindrocladium)[J]. Studies in Mycology, 2010, 66:1-13. doi: 10.3114/sim.2010.66.01 [2] Lombard L, Chen S F, Mou X, et al. New species, hyper-diversity and potential importance of Calonectria spp. from Eucalyptus in South China[J]. Studies in Mycology, 2015, 80:151-188. doi: 10.1016/j.simyco.2014.11.003 [3] 陈全助, 郭文硕, 叶小真, 等.福建省桉树焦枯病菌分类鉴定[J].福建林学院学报, 2013, 33(2):176-182. doi: 10.3969/j.issn.1001-389X.2013.02.015 [4] Chen S F, Lombard L, Roux J, et al. Novel species of Calonectria associated with Eucalyptus leaf blight in Southeast China[J]. Persoonia, 2011, 26:1-12. doi: 10.3767/003158511X555236 [5] 朱建华, 郭文硕, 陈红梅, 等.桉树焦枯病对桉树生长量的损失估计研究[J].中国森林病虫, 2011, 30(05):6-10. doi: 10.3969/j.issn.1671-0886.2011.05.002 [6] 陈全助, 桉树种系对焦枯病抗性的初步测定[J].福建林学院学报, 2010, 30(4):297-299. doi: 10.3969/j.issn.1001-389X.2010.04.003 [7] 李国清, 李洁琼, 刘菲菲, 等. 12种桉树焦枯病病原菌对10种桉树无性系致病性初步分析[J].桉树科技, 2014, 31(4):1-7. doi: 10.3969/j.issn.1674-3172.2014.04.001 [8] Rodas C A, Lombard L, Gryzenhoinf M, et al. Cylindrocladium blight of Eucalyptus grandis in Colombia[J]. Australasian Plant Pathology, 2005, 34(2):143-149. doi: 10.1071/AP05012 [9] Dixon R A and Paiva N L.Stress-induced phenylpropanoid metabolism[J]. Plant Cell, 1995, 7(7):1085. doi: 10.2307/3870059 [10] Naoumkina M A, Zhao Q, Gallego-Giraldo L, et al. Genome-wide analysis of phenylpropanoid defence pathways[J]. Molecular Plant Pathology, 2010, 11(6):829-846. [11] Huang J, Gu M, Lai Z, et al. Functional analysis of the Arabidopsis PAL gene family in plant growth, development, and response to environmental stress[J]. Plant Physiology, 2010, 153(4):1526-1538. doi: 10.1104/pp.110.157370 [12] 徐晓梅, 杨署光.苯丙氨酸解氨酶研究进展[J].安徽农业科学, 2009, 37(31):15115-15119, 15122 doi: 10.3969/j.issn.0517-6611.2009.31.012 [13] 曾永三, 王振中.苯丙氨酸解氨酶在植物抗病反应中的作用[J].仲恺农业技术学院学报, 1999, 12(3):56-65. [14] Vaganan M M, Ravi I, Nandakumar A, et al. Phenylpropanoid enzymes, phenolic polymers and metabolites as chemical defenses to infection of Pratylenchus coffeae in roots of resistant and susceptible bananas (Musa spp.)[J]. Indian Journal of Experimental Biology, 2014, 52(3):252-260. [15] 张树武, 徐秉良, 刘佳, 等.白粉病菌侵染对美洲南瓜不同抗性品种PAL基因表达量的影响[J].核农学报, 2015, 29(10):1876-1883. doi: 10.11869/j.issn.100-8551.2015.10.1876 [16] 虞光辉, 王桂平, 王亮, 等.小麦PAL基因的克隆及赤霉菌诱导下的表达分析[J].植物遗传资源学报, 2015, 16(5):1055-1061. [17] 张江涛.苯丙氨酸解氨酶(PAL)与水稻抗稻瘟病的关系[J].植物生理学通讯, 1987(6):34-37. [18] 贺字典, 高增贵, 庄敬华, 等.玉米丝黑穗病菌对寄主防御相关酶活性的影响[J].玉米科学, 2006, 14(2):150-151. doi: 10.3969/j.issn.1005-0906.2006.02.048 [19] 郭文硕.锥栗对栗疫病的抗性与苯丙氨酸解氨酶的关系[J].林业科学, 2001, 37(1):90-93. doi: 10.3321/j.issn:1001-7488.2001.01.013 [20] Feng L Z, Guo W S, Xie W F, et al. Construction and analysis of a SSH cDNA library of Eucalyptus grandis×Eucalyptus urophylla 9224 induced by Cylindrocladium quinqueseptatum[J]. Botany, 2012, 90(12):1277-1283. doi: 10.1139/b2012-099 [21] 杨婕, 郭文硕, 叶小真, 等.巨桉叶片总RNA提取方法比较[J].基因组学与应用生物学, 2015, 34(6):1272-1276. [22] Akimoto S, Ishino T, Terada T, et al. A phenylalanine ammonia-lyase gene (ErPAL1) from Eucalyptus robusta: molecular cloning, expression and characterization[J]. Bulletin University of Tokyo Forest, 2013, 128:121-137. [23] Zhao S, Park C H, Li X, et al. Accumulation of rutin and betulinic acid and expression of phenylpropanoid and triterpenoid biosynthetic genes in mulberry (Morus alba L.)[J]. Journal Agricultural and Food Chemistry, 2015, 63(38):8622-8630. doi: 10.1021/acs.jafc.5b03221 [24] 冯立娟, 尹燕雷, 焦其庆, 等.石榴PAL基因的克隆与表达分析[J].核农学报, 2018, 32(7):1320-1329. [25] Ma W, Wu M, Wu Y, et al. Cloning and characterisation of a phenylalanine ammonia-lyase gene from Rhus chinensis[J]. Plant Cell Reports, 2013, 32(8):1179-1190. doi: 10.1007/s00299-013-1413-6 [26] Chen Z J, Cao Z M, Yu Z D, et al. Cloning and characterization of defense-related genes from Populus szechuanica infected with rust fungus Melampsora larici-populina[J]. Genetics and Molecular Research, 2016, 15(1). doi:10.4238/gmr.15017314. [27] Cass C L, Peraldi A, Dowd P, et al. Effects of PHENYLALANINE AMMONIA LYASE (PAL) knockdown on cell wall composition, biomass digestibility, and biotic and abiotic stress responses in Brachypodium[J]. Journal of Experimental Botany, 2015, 66(14):4317-4335. doi: 10.1093/jxb/erv269 [28] Tian L, Shi S, Nasir F, et al. Comparative analysis of the root transcriptomes of cultivated and wild rice varieties in response to Magnaporthe oryzae infection revealed both common and species-specific pathogen responses[J]. Rice, 2018, 11(1):26 doi: 10.1186/s12284-018-0211-8 [29] Giberti S, Bertea C M, Narayana R, et al. Two phenylalanine ammonia lyase isoforms are involved in the elicitor-induced response of rice to the fungal pathogen Magnaporthe oryzae[J]. Journal of Plant Physiology, 2012, 169(3):249-254. [30] Kim D S and Hwang B K. An important role of the pepper phenylalanine ammonia-lyase gene (PAL1) in salicylic acid-dependent signalling of the defence response to microbial pathogens[J]. Journal of Experimental Botany, 2014, 65(9):2295-2306. doi: 10.1093/jxb/eru109 [31] Shine M B, Yang J W, El-Habbak M, et al. Cooperative functioning between phenylalanine ammonia lyase and isochorismate synthase activities contributes to salicylic acid biosynthesis in soybean[J]. New Phytology, 2016, 212:627-636. doi: 10.1111/nph.14078 [32] Tonnessen BW, Manosalva P, Lang J M, et al. Rice phenylalanine ammonia-lyase gene OsPAL4 is associated with broad spectrum disease resistance[J]. Plant Molecular Biology, 2015, 87(3):273-286 doi: 10.1007/s11103-014-0275-9 [33] Li Y X, Zhang W, Dong H X, et al. Salicylic acid in Populus tomentosa is a remote signalling molecule induced by Botryosphaeria dothidea infection[J].Science Report, 2018, 8(1):14059 doi: 10.1038/s41598-018-32204-9 [34] 冯丽贞, 陈全助, 郭文硕, 等.桉树的次生代谢及其对焦枯病抗性的关系[J].中国生态农业学报, 2008, 16(2):426-430. [35] 冯丽贞, 陈全助, 郭文硕, 等.植物防御酶与桉树对焦枯病抗性的关系[J].中国生态农业学报, 2008, 16(5):1188-1191. -




 下载:
下载: