-
林下植被管理是人工林经营中较重要的管理措施之一。在长期的林业生产中,经营者普遍对林下植被进行清除,从而减少其与上层乔木的水分和养分竞争[1]。大量研究表明,林下植被尽管在森林总生物量只占很小部分,但由于具有较高的养分积累量,其凋落物的化学组成以及生物量周转速率明显优于上层乔木,因而,在结构相对单一的人工林生态系统的养分循环中起重要作用[2-3]。Qiao等[4]和Lin等[5]等研究发现,林下植被的存在提高了森林土壤的有机质含量,有利于人工林的养分周转及长期生产力的保持,但Tripathi等[6-7]研究发现,通过去除林下植被,使白桦(Betula platyphylla Suk.)人工林的总有机碳、总氮、微生物生物量碳和氮含量均显著提升,土壤质量得到改善。此外,也有相关研究结果表明,在我国亚热带地区马占相思(Acacia mangium Willd.)人工林中进行林下植被去除后,土壤理化性质并未发生显著变化[8-10]。可见,林下植被在人工林生态系统养分循环中的作用及其机制受到地理位置和植被类型等诸多因素的影响,探究林下植被不同管理措施的影响效益对于人工林的经营和管理具有重要的理论和实践价值。
土壤酶来自微生物、植物或动物的活体或残体,既是生物体又是生物过程的产物[11]。土壤酶作为土壤生物化学过程的积极参与者,直接参与土壤的物质迁移转化与养分的固定释放[12-14],其活性受土壤理化性质、植被类型和微生物群落结构等诸多因素的影响,被视为一种比土壤微生物区系、群落组成以及土壤呼吸等更敏感、可靠的生物活性指标,可作为构建土壤质量评价体系的指标之一[15]。目前,关于林下植被的研究普遍集中于林下植被不同管理措施下土壤理化性质的变化,且往往在林下植被管理措施实施当年或次年对土壤进行采样分析,而以土壤酶为角度探究林下植被不同管理措施长期效益的研究仍有待深入。
杉木(Cunninghamia lanceolata(Lamb.)Hook.)属于亚热带的常绿乔木,具有生长快,材质好,病害少等优点,是我国南方人工林造林的主要树种,种植面积已达17×106 hm2,占全国人工林面积的24%和世界人工林面积的6.5%,对林业的生产和开发具有至关重要的作用[16-17]。当前,经济发展和人民生活水平日益提高,国内木材消费需求处于不断增长之中,中小型径材的需求趋于饱和,而大径级用材的供需矛盾较为紧张[18],为此,加强杉木大径材培育林的经营与管理显得尤为重要。由于大径材培育林林分结构相对稳定,林下植被与上层乔木竞争关系趋于减弱,长期以来有关此阶段杉木人工林林下植被管理效益的研究少有报道,特别关于林下植被不同管理措施对于杉木林地土壤酶活性的研究十分薄弱。鉴于此,本研究以亚热带地区培育杉木大径材林分为研究对象,在林分内设置林下植被保留、林下植被去除以及林下植套种3种林下植被管理措施,探究林下植被不同管理措施下杉木人工林土壤酶活性差异,并采用主成分分析法将土壤酶活性作为土壤质量指标,对土壤质量进行综合评价,为探索科学合理的杉木大径材林经营管理提供理论依据。
-
研究区位于福建省洋口国有林场南元管护站南山下林厂(117°92′~118°64′ E,26°87′~27°13′ N),系武夷山脉南部,属低山丘陵区,属于典型亚热带气候,海拔252~339 m,平均坡度26~32°。该区年平均气温18.6℃,极端最高气温41.4℃,极端最低气温−5.8℃,年降水量1 600~1 900 mm,年平均蒸发量1 308~1 587 mm,无霜期277~285 d,光照充足,雨量充沛。土壤类型为丘陵红壤。
-
2018年6月在福建省洋口国有林场南元管护站,根据海拔、坡度、坡向等立地条件尽量一致的原则,选择林下植被保留(UP)、林下植被去除(UR)和林下套种楠木(Phoebe bourmei S. Lee)(IP)3种林下植被管理措施培育的杉木大径材林分。3种林分造林时间均为1984年,种苗来源为杉木第1代种子园的优良家系壮苗,初植密度为2 505 株·hm−2,前3年每年全面锄草抚育2次,于1998年进行抚育间伐,间伐强度约50%,保留株数1 187 株·hm−2,后在保障杉木个体生长的基础上使林下植被自然演化,减少人为干扰,2012年被选为杉木大径材林进行培育。UP处理的林分采取林下植被保留方式至今,林下植被保留完好;UR处理的林分于2012年7月起,每年进行1次林下植被清理,林下灌木清除采用割灌机,林下草本采用镰刀手工割除,清除的林下植被残体原样保留在试验地内;IP处理的林分于2012年林下植被清除后实施林下套种,清除的林下植被残体经耙带(带宽1 m,带与带间距7 m)整理后保留在试验地内,楠木套种密度为450 株·hm−2,并定期抚育。
-
2018年7月在每种林分类型中分别设置3个20 m×20 m样地进行每木调查,并在每块标准样地内的四周和中心设置5个5 m×5 m的灌木小样方和随机设置10个1 m×1 m的草本小样方,分别调查灌木和草本的种类、株数、胸径、高度、盖度等。不同林分类型样地基本情况见表1,UP和IP处理的林分样地林下植被情况见表2。在每块样地内按对角线随机设置5个采样点,去除表层枯枝落叶后挖取土壤剖面,分别取0~20、20~40、40~60 cm土层的土壤样品用于测定物理性质和化学性质。将同一样地同一土层获取的土壤样品去除石块等杂物后等量混匀,采取4分法分为2份,1份经快速过筛后置于0~4℃冰箱保存备用测定土壤酶活性,1份用于自然风干过筛用于测定土壤化学性质。
表 1 林下植被不同管理措施的样地基本情况
Table 1. Basic information of stands under different understory vegetation management measures
措施
Treatment坡向
Aspect坡位
Position坡度
Slope/(°)林分密度
Stand density/(株·hm−2)平均树高
Average height/m平均胸径
Average diameter/cmUP 西南WS 中 31 958 22.68±2.70 31.86±4.29 UR 南S 中 35 983 22.30±2.12 30.26±3.17 IP 西W 中 34 928 22.32±1.97 31.75±3.92 注:表中数值为平均值±标准误,下同。
Note: Values in the table are average ± standard error, the same blow.表 2 林下植被不同管理措施的样地林下植被
Table 2. Understory vegetation of stands under different understory vegetation management measures
措施
Treatment灌木
Shrub layer草本层
Herb layer总盖度
Total
coverage
/%物种名称
Species平均高度
Average
height/m平均胸径
Average
diameter/cm重要值
Important
value/%物种名称
Species平均高度
Average
height/m盖度
Coverage
/%频度
Frequency重要值
Important
value/%鲜生物量
Fresh
biomass/
(t·hm−2)UP 紫麻
Oreocnide
frutescenssp2.31 2.22 23.87 薄盖短肠蕨
Allantodia
hachijoensis0.70 0.45 0.67 8.25 6.70 92 粗叶榕
Ficus hirta1.93 1.70 17.49 江南短肠蕨
Allantodia
metteniana0.44 0.19 0.73 6.14 杜虹花
allicarpa
formosana1.43 0.61 15.36 三叶青
Tetrastigma
hemsleyanum0.46 0.26 0.60 5.11 IP 楠木
Phoebe bourmei2.98 2.46 100 深绿卷柏
Selaginella
doederleinii0.21 0.19 0.67 9.78 6.29 88 薄盖短肠蕨
Allantodia
hachijoensis0.54 0.38 0.87 7.13 金星蕨Parathelypteris glanduligera 0.33 0.18 0.87 5.73 注:表中所列重要值为前3的植物种。
Note: The table lists the plants with the first 3 important values. -
蔗糖酶活性采用水杨酸比色法测定,结果以24 h后1 g土壤中葡萄糖的毫克数表示;脲酶活性采用靛酚蓝比色法测定,结果以24 h后1 g土壤中NH3-N的毫克数表示;酸性磷酸酶采取4-硝基苯酚磷酸盐法(C6H5NO3)测定,结果以24 h后1 g土壤中对硝基苯酚的毫克数表示;过氧化氢酶采取紫外分光光度法测定,结果以每20 min内1 g土壤分解的过氧化氢的毫克数表示;多酚氧化酶活性采用邻苯三酚比色法测定,结果以2 h后1 g土壤中没食子素的毫克数表示。
土壤容重、含水率和孔隙度等物理性质采用环刀法取样测定,土壤有机质含量采用重铬酸钾氧化-外加热法测定,全氮采用半微量凯氏法测定,全磷采用碱熔-钼锑抗比色法测定,全钾采用碱熔-火焰光度法测定,水解性氮用碱解-扩散法测定,有效磷采用NaHCO3浸提钼锑抗显色法测定,速效钾采用中性乙酸铵提取-火焰光度计法测定,以上测定方法参考文献[18-20]。
-
数据采用Microsoft Excel 2016进行整理作图。使用SPSS 23进行采用单因素方差分析及Pearson相关性分析和主成分分析,方差分析显著性水平均设定为P=0.05。通过主成分分析,得到林下植被管理措施土壤的各主成分得分,采用加权法计算土壤质量指数(SQI),其表达式为[21]:
$ SQI = \sum {{W_i} \times {F_i}} $
式中:Wi为各主成分贡献率,Fi为各样地的主成分得分,如得分为正,表明该林分的土壤综合指数在平均水平之上,反之表明在平均水平以下。
-
表3表明:3种林下植被管理措施,0~20 cm土层的土壤蔗糖酶活性高低排序为IP>UP>UR,IP处理的土壤蔗糖酶活性比UP和UR处理分别提高了27.9%和64.4%,其中,IP和UR处理的土壤蔗糖酶活性差异显著;20~40、40~60 cm土层,3种林下植被管理措施的土壤蔗糖酶活性间差异不显著。可见,IP处理对杉木人工林0~20 cm土层的土壤蔗糖酶活性具有促进作用,对于更深层的土壤,3种林下植被管理措施的影响较小。
表 3 林下植被管理措施对于土壤酶活性的影响
Table 3. Soil enzyme activity under understory vegetation management measures
措施
Treatment土层
Soillayer/ cm蔗糖酶
Sucrase/(mg·g−1)脲酶
Urease/(mg·g−1)多酚氧化酶
Polyphenol oxidase/(mg·g−1)过氧化氢酶
Catalase/(mg·g−1)酸性磷酸酶
Acid phosphatase/(mg·g−1)UP 0~20 39.08±7.05ABa 2.38±0.68Aa 0.26±0.05Aa 18.45±3.22Aa 0.46±0.02Ba 20~40 25.92±4.25Ab 1.33±0.67Bb 0.15±0.02Ab 17.19±2.63Aa 0.45±0.23Ba 40~60 21.35±5.11Ab 1.29±0.27Ab 0.12±0.02Ab 16.68±4.79Aa 0.46±0.07Aa UR 0~20 30.39±6.02Bb 2.98±0.98Aa 0.10±0.07Ca 17.61±4.34Aa 0.48±0.04Ba 20~40 25.73±2.12Aab 2.37±0.60Aab 0.07±0.01Bb 15.12±3.19Ba 0.49±0.95ABa 40~60 19.32±6.21Ab 1.54±0.60Ab 0.04±0.04Bb 14.52±2.55Ab 0.47±0.12Aa IP 0~20 49.97±10.72Aa 3.05±0.33Aa 0.17±0.03Ba 18.24±4.90Aa 0.54±0.14Aa 20~40 30.82±2.31Ab 2.31±0.42Aab 0.11±0.06ABb 15.21±2.36Ba 0.53±0.16Aa 40~60 18.19±4.22Ac 1.39±0.34Ab 0.10±0.04ABb 13.39±3.58Ba 0.48±0.07Aa 注:不同大写字母表示林下植被不同管理措施同一土层酶活性差异显著(p<0.05);不同小写字母表示同一林下植被管理措施不同土层酶活性差异显著(p<0.05)。
Notes: Different capital letters indicated significant differences in enzyme activity in the same soil layer under different understory vegetation management measures (p<0.05). Different lowercase letters indicate significant differences in enzyme activity in different soil layers under the same understory vegetation management measure (p<0.05).UP处理的土壤脲酶活性均低于UR和IP处理,UR和IP处理的土壤脲酶活性间差异较小;而UP处理的土壤脲酶活性在20~40 cm土层迅速降低,分别比UR和IP处理降低了43.9%和42.4%,且差异显著。
UP处理的土壤过氧化氢酶活性均高于UR和IP处理;20~40 cm土层,UP处理的土壤过氧化氢酶活性分别比UR和IP处理提高了13.7%和13.0%;40~60 cm土层,UP处理的土壤过氧化氢酶活性分别比UR和IP处理提高了14.9%和24.6%;20~40 cm和40~60 cm土层,UP和IP处理的土壤过氧化氢酶活性差异显著,而UR和IP处理的土壤过氧化氢酶活性差异较小。
3种林下植被管理措施各土层的土壤酸性磷酸酶活性高低排序为IP>UR>UP;0~20 cm土层,IP处理的土壤酸性磷酸酶活性显著高于UP和UR处理,分别提高了17.4%和12.5%。值得注意的是,与其他种类土壤酶不同,各处理的土壤酸性磷酸酶活性随土层深度的增加下降幅度较小,甚至略有提升,可见,土层深度对于杉木人工林土壤酸性磷酸酶活性的影响较小。
3种林下植被管理措施各土层的土壤多酚氧化酶活性高低排序为UP>IP>UR;0~20 cm土层,UP和IP处理的土壤多酚氧化酶活性分别比UR处理提高了160.0%和70.0%,且UP处理的土壤多酚氧化酶活性与UR和IP处理间差异显著。
综合而言,林下植被不同管理措施的土壤脲酶、蔗糖酶和多酚氧化酶活性差异较大,其中,多酚氧化酶对于各林下植被管理措施的响应更灵敏,且能反映于较深土层。除酸性磷酸酶活性外,其它4类土壤酶活性均具明显的表聚性,随土层的加深而递减。
-
结合各林分土壤物理性质(表4)和化学性质(表5)进行相关性分析,由表6可知:有机质含量与蔗糖酶、脲酶、过氧化氢酶和多酚氧化酶活性均呈极显著正相关,与酸性磷酸酶活性呈显著正相关;土壤水解性氮含量与5类土壤酶活性具有极显著或显著正相关;有效磷含量与蔗糖酶、脲酶和酸性磷酸酶活性呈极显著正相关;速效钾含量与蔗糖酶、过氧化氢酶和多酚氧化酶活性呈极显著正相关;土壤容重与各类土壤酶活性均为负相关;蔗糖酶与土壤总孔隙度呈显著正相关,与非毛管孔隙度呈极显著正相关;脲酶活性与土壤含水率和非毛管孔隙度呈极显著正相关。过氧化氢酶活性与土壤总孔隙度呈显著正相关;酸性磷酸酶活性与总孔隙度和非毛管孔隙度呈极显著正相关,与毛管孔隙度呈显著正相关;多酚氧化酶活性与土壤含水率呈显著正相关。相关性分析表明:杉木人工林土壤的理化性质均与土壤酶活性具有紧密联系,可见土壤指标之间相关性比较强,存在信息上的重叠,因此,为明确林下植被不同管理措施杉木人工林土壤质量的差异,本试验对土壤各指标进行数据降维的统计分析,即主成分分析。
表 4 林下植被不同管理措施培育杉木大径材林分土壤物理性质
Table 4. Soil physical properties of Cunninghamia lanceolata large-diameter timber cultivated forestunder different understory vegetation management measures
措施
Treatment土层
Soil layer/cm容重
Bulk density/(g·m−3)含水率
Moisture content/%总孔隙度
Total porosity/%毛管孔隙度
Capillary porosity/%非毛管孔隙度
Noncapillary porosity/%UP 0~20 1.21±0.05 25.4±2.78 50.2±2.35 39.5±5.91 10.7±2.32 20~40 1.24±0.05 24.4±1.17 45.8±3.12 39.3±5.36 6.5±0.03 40~60 1.36±0.02 23.2±0.67 45.0±5.72 36.3±3.79 5.4±0.17 UR 0~20 1.24±0.02 24.7±0.98 46.5±2.57 36.8±6.74 10.8±1.24 20~40 1.37±0.12 24.5±1.20 44.2±3.51 36.7±2.09 8.8±0.95 40~60 1.38±0.01 20.6±2.60 44.1±1.74 36.0±1.00 8.0±0.82 IP 0~20 1.02±0.27 22.7±0.54 53.2±2.35 41.6±4.90 11.6±2.44 20~40 1.23±0.01 20.4±0.62 50.7±7.28 41.5±10.96 9.2±1.06 40~60 1.26±0.02 20.1±0.34 44.5±1.83 35.4±0.58 9.1±2.17 表 5 林下植被不同管理措施培育杉木大径材林分土壤化学性质
Table 5. Soil chemical properties of Cunninghamia lanceolata large-diameter timber cultivated forest under different understory vegetation management measure
措施
Treatment土层
Soil layer/cm有机质
Organic content/
(g·kg−1)水解性氮
Hydrolysis nitrogen/
(mg·kg−1)有效磷
Available phosphate/
(mg·kg−1)速效钾
Rapidly availablepotassium/
(mg·kg−1)UP 0~20 32.72±2.57 7.68±0.33 0.57±0.27 62.99±2.61 20~40 28.93±2.47 5.86±0.39 0.47±0.03 52.44±3.44 40~60 19.07±0.22 5.70±0.44 0.42±0.04 49.82±1.99 UR 0~20 29.83±0.90 8.31±0.17 0.60±0.05 43.98±2.03 20~40 23.34±1.63 5.96±0.58 0.53±0.10 32.11±1.59 40~60 18.13±1.49 5.83±0.20 0.48±0.01 28.49±1.16 IP 0~20 31.77±0.40 9.69±0.86 0.79±0.03 52.58±3.82 20~40 27.31±1.28 6.47±0.73 0.65±0.01 44.52±2.90 40~60 19.74±0.92 6.35±0.36 0.60±0.03 35.15±1.73 表 6 土壤酶活性与土壤性质的相关性分析
Table 6. Pearson’s correlation coefficients between soil enzyme activities and properties
指标
Indicator有机质
Organiccontent水解性氮
Hydrolysis
nitrogen有效磷
Available
phosphate速效钾
Rapidly
available
potassium容重
Bulkdensity含水率
Moisture
content总孔隙度
Totalporosity毛管孔隙度
Capillary
porosity非毛管孔隙度
Noncapillary
porosity蔗糖酶Suarese 0.744** 0.703** 0.605** 0.532** −0.805** 0.224 0.482* 0.372 0.496** 脲酶Urease 0.599** 0.603** 0.528** 0.261 −0.463* 0.489** 0.352 0.376 0.609** 过氧化氢酶Catalase 0.586** 0.409* 0.209 0.605** −0.409* 0.374 0.398* 0.264 −0.027 酸性磷酸酶Acid phosphatase 0.395* 0.409* 0.744** 0.016 −0.358 0.011 0.652** 0.484* 0.560** 多酚氧化酶Polyphenol oxidase 0.679** 0.494** 0.342 0.828** −0.549** 0.390* 0.341 0.375 0.286 注Note:** P <0.01,* P <0.05.
-
为保证主成分分析结果的客观性和科学性,将本研究中14个土壤指标进行标准化处理,以排除不同量纲和数量级对土壤质量评价的影响。由表7可知:特征根值≥1的主成分有3个,3个主成分解释百分比分别为26.99%、24.91%和23.41%,共可解释土壤参数总变量的75.31%,其中,脲酶、酸性磷酸酶、有效磷、总孔隙度、毛管孔隙度和非毛管孔隙度在PC1上有较高因子负荷,过氧化氢酶、多酚氧化酶、有机质和速效钾在PC2上有较高因子负荷,蔗糖酶、水解性氮、有效磷和容重在PC3上有较高因子负荷。
表 7 土壤质量因子主成分的贡献率及因子载荷、权重
Table 7. Eigenvalue and contribution rate of principal factors
指标
Indicator主成分因子载荷
Principal component factor loadingPC1 PC2 PC3 蔗糖酶 Suarese 0.480 0.454 0.645 脲酶 Urease 0.710 0.232 0.395 过氧化氢酶 Catalase −0.030 0.725 0.277 酸性磷酸酶 Acid phosphatase 0.880 −0.069 0.205 多酚氧化酶 Polyphenol oxidase 0.188 0.796 0.342 有机质 Organic content 0.399 0.649 0.520 水解性氮 Hydrolysis nitrogen 0.309 0.212 0.805 有效磷 Available phosphate 0.643 −0.043 0.708 速效钾 Rapidly available potassium 0.106 0.882 0.258 容重 Bulk density −0.226 −0.287 −0.876 含水率 Moisture content 0.171 0.714 −0.184 总孔隙度 Total porosity 0.734 0.311 0.143 毛管孔隙度 Capillary porosity 0.626 0.359 0.085 非毛管孔隙度 Noncapillary porosity 0.742 0.018 0.392 特征根值 Eigenvalue 3.778 3.487 3.277 百分比 Percent/% 26.99 24.91 23.41 累积贡献率 Cumulative percent/% 26.99 51.90 75.31 -
表8是各林下植被管理措施土壤各指标在相应主成分上的因子得分系数矩阵,对应表7的3个主成分,可获得各主成分的函数:
表 8 土壤指标成分得分系数矩阵
Table 8. Component score coefficient matrix of soil index
指标
Indicator因子得分系数
Component score coefficient matrixPC1 PC2 PC3 蔗糖酶 Suarese 0.002 0.043 0.172 脲酶 Urease 0.207 −0.010 −0.025 过氧化氢酶 Catalase −0.140 0.238 0.059 酸性磷酸酶 Acid phosphatase 0.365 −0.102 −0.148 多酚氧化酶 Polyphenol oxidase −0.056 0.245 0.014 有机质 Organic content −0.005 0.146 0.085 水解性氮 Hydrolysis nitrogen −0.127 −0.074 0.377 有效磷 Available phosphate 0.081 −0.172 0.249 速效钾 Rapidly available potassium −0.072 0.298 −0.028 容重 Bulk density 0.195 0.054 −0.438 含水率 Moisture content 0.116 0.308 −0.305 总孔隙度 Total porosity 0.300 0.066 −0.210 毛管孔隙度 Capillary porosity 0.265 0.100 −0.220 非毛管孔隙度 Noncapillary porosity 0.233 −0.095 0.001 PC1=蔗糖酶×0.002+脲酶×0.207+过氧化氢酶×(−0.140)+酸性磷酸酶×0.365+多酚氧化酶×(−0.056)+有机质×(−0.005)+水解性氮×(−0.127)+有效磷×0.081+速效钾×(−0.072)+容重×0.195+含水率×0.116+总孔隙度×0.300+毛管孔隙度×0.265+非毛管孔隙度×0.233。
PC2=蔗糖酶×0.043+脲酶×(−0.010)+过氧化氢酶×0.238+酸性磷酸酶×(−0.102)+多酚氧化酶×0.245+有机质×0.146+水解性氮×(−0.074)+有效磷×(−0.172)+速效钾×0.298+容重×0.054+含水率×0.308+总孔隙度×0.066+毛管孔隙度×0.100+非毛管孔隙度×(−0.095)。
PC3=蔗糖酶×0.172+脲酶×(−0.025)+过氧化氢酶×0.059+酸性磷酸酶×(−0.148)+多酚氧化酶×0.014+有机质×0.085+水解性氮×0.377+有效磷×0.249+速效钾×(−0.028)+容重×(−0.438)+含水率×(−0.305)+总孔隙度×(−0.210)+毛管孔隙度×(−0.220)+非毛管孔隙度×0.001。
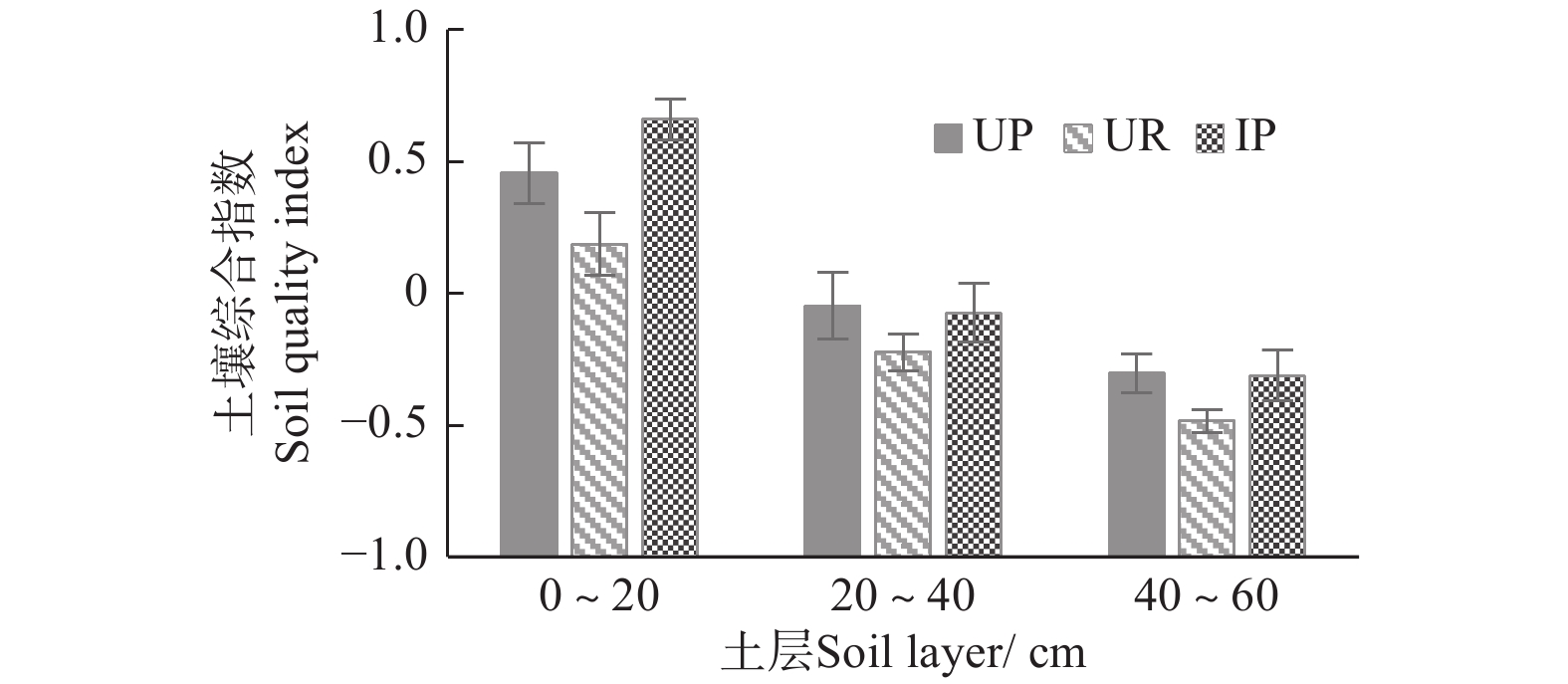
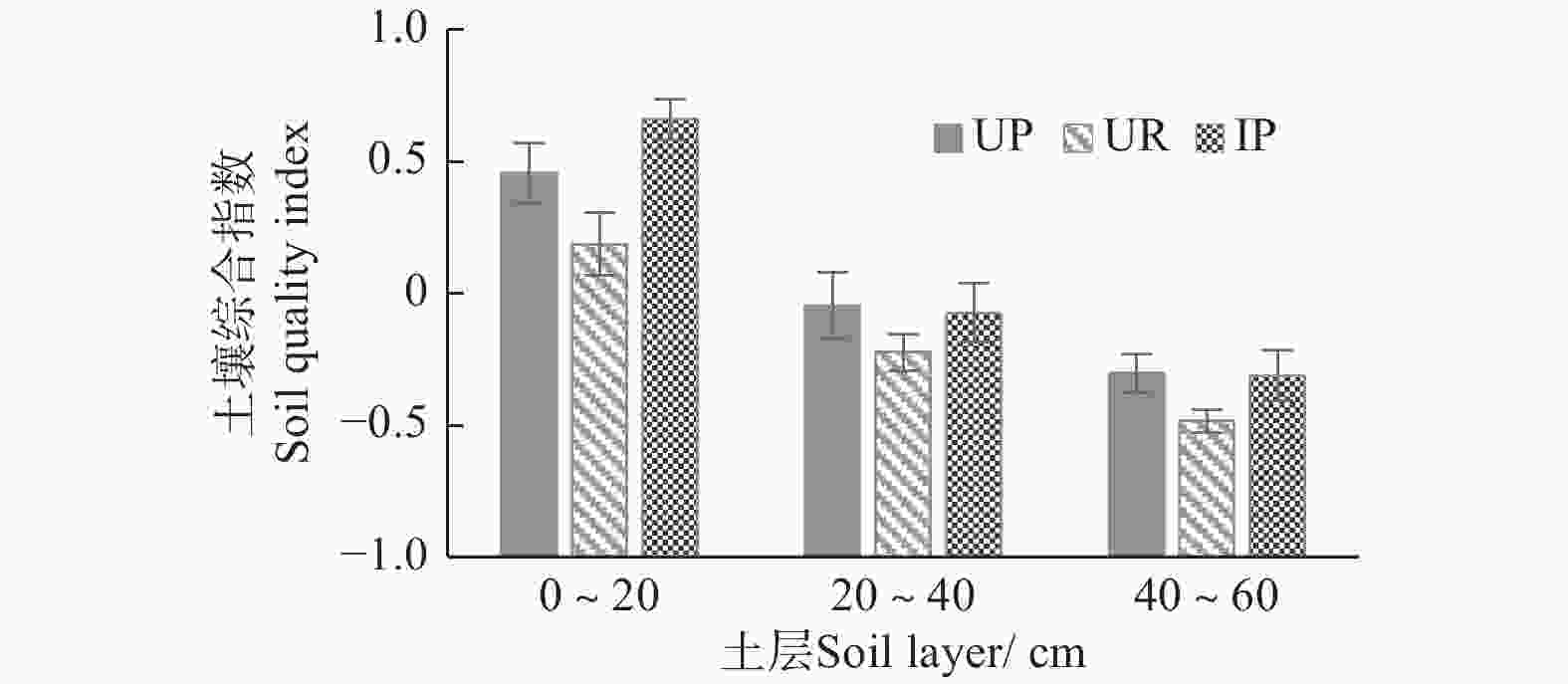
将经过标准化的数据分别代入主成分函数表达式中,得到3种林下植被不同管理措施土壤得分情况,图1显示:3种林下植被管理措施的杉木人工林土壤质量均随着土层深度的增加而降低;同一土层土壤质量综合指数高低排序均呈IP>UP>UR,可知林下套种楠木对于土壤质量提升效果最佳。
-
土壤酶活性的高低与林下植物群落生物量、植被盖度、植物多样性具有密切关系[11,21-22],林下植被的根系物质分泌、凋落物积累等生长代谢过程以及对林内土壤性质和水热条件等环境因素间接改变,均导致森林生态系统的土壤酶活性发生改变[23-24]。本研究中,5类土壤酶对林下植被管理措施响应表现不一,与林下植被保留措施相比,林下植被去除降低了土壤蔗糖酶、过氧化氢酶和多酚氧化酶活性,增加了土壤脲酶和酸性磷酸酶活性,这与杨洋等[25]、郑琳琳等[26]的研究结果有差异。林下植被细根周转及其根际分泌物能够为土壤提供不稳定性有机化合物[27],与根系相连的菌根也能产生胞外酶。林下植被去除直接减少了根际分泌物、细根周转以及来自其凋落物的养分输入[28],降低了凋落物分解速率[29-31],导致土壤中酶促反应底物含量的下降,从而引起土壤酶活性的降低。脲酶为土壤中的聚积酶,主要来自于动植物残体所形成的土壤有机质。本研究区域位于亚热带地区,水热条件较好,植被恢复速度较快,林下植被去除管理措施中所残留的林下植被清理剩余物使土壤中植物残体增多,可能是土壤脲酶活性增加的原因之一。杨万勤等[31]研究表明,林下植被的种类和数量对土壤酸性磷酸酶活性不具有明显影响,可见,酸性磷酸酶受植物的生理代谢过程的影响较小。本试验中,与林下植被保留措施相比,杉木人工林下套种楠木,营造杉楠复层混交林提高了土壤蔗糖酶、脲酶和酸性磷酸酶活性。长期的林业生产实践已证实,杉木和楠木的优化组合改善了土壤结构和养分,进而增加土壤酶活性,对杉木大径材林地生态系统起到积极作用,这与部分学者研究结果基本相同[32-33]。以土壤酶活性的垂直分布差异而言,除酸性磷酸酶活性外,其它4类土壤酶活性均具明显表聚性,这与Song等[34]和Chen等[35]研究基本一致,造成这种现象的主要原因为随着土层深度的增加,土壤的理化性质随之下降,微生物种类和数量递减,因而,土壤酶活性随土层深度的增加逐渐下降[36]。
本试验相关性分析结果表明,蔗糖酶活性与水解性氮、有效磷和速效钾含量均呈极显著正相关,脲酶活性与有效磷含量也具有极显著正相关,酸性磷酸酶活性与水解性氮含量呈显著正相关,而过氧化氢酶和多酚氧化酶活性与水解性氮和速效钾含量均具有显著或极显著正相关,可知,土壤酶活性与土壤养分并非简单的一一对应关系,不同种类的酶和土壤养分之间具有协同交互作用,有待进一步研究。众多研究表明,土壤酶活性与土壤物理性质和养分含量有密切的相关性[36]。通过对林下植被不同管理措施土壤质量进行了优势排序,各土层均呈现为IP>UP>UR。可知,相比于自然演化形成的自然植被,林下套种楠木的管理措施是缓解杉木大径材人工林地力衰退的有效途径之一,而林下植被去除不利于土壤质量的保持和提升。
本研究对象为培育杉木大径材林分,培育时间较长,其林下植被处于不断更新演替中;随着楠木的生长发育,林下所套种的楠木在杉木人工林生态系统中的影响效益也将进一步加大。因此,林下植被不同管理措施对土壤质量的长期影响有待进一步跟踪研究。此外,由于不同种类的林下植被具有不同的物候,其生长发育节律导致土壤性质在不同季节间产生差异,为更好研究林下植被与林分土壤的相互作用,在后续的研究中应加强不同季节林下植被管理措施对于土壤性质的影响。
-
(1)相比UR和IP处理,UP处理提高了杉木大径材培育林土壤过氧化氢酶活性,脲酶活性则有所降低,UR和IP处理的脲酶和过氧化氢酶活性之间差异较小,3种林下植被管理措施的杉木人工林土壤酸性磷酸酶活性高低排序为IP>UR>UP,多酚氧化酶活性高低排序为UP>IP>UR。
(2)以土壤酶作为土壤生物活性指标,结合土壤物理和化学性质利用主成分分析对土壤质量综合定量评价,结果表明:3种林下植被管理措施下的杉木大径材培育林各土层土壤质量水平指数排序均为:IP>UP>UR,林下套种楠木对于土壤质量的提升效果最佳。
林下植被不同管理措施培育杉木大径材林分土壤酶活性差异及质量评价
The Differences of Soil Enzyme Activity and Quality Evaluation of Cunninghamia lanceolata Large-diameter Industrial Plantation under Different Understory Vegetation Management Measures
-
摘要:
目的 探讨林下植被不同管理措施对培育杉木大径材林分土壤酶活性及土壤质量的影响。 方法 本研究以培育杉木大径材林分为研究对象,分析了林下植被保留(UP)、林下植被去除(UR)和林下套种(IP)3种林下植被管理措施培育杉木大径材林分土壤酶活性差异,并以土壤酶作为土壤生物活性指标,结合土壤物理和化学性质,利用主成分分析法对土壤质量进行综合定量评价。 结果 IP处理提高了0~20 cm土层蔗糖酶活性,而20~40、40~60 cm土层3种林下植被管理措施间土壤蔗糖酶活性差异较小;相比于UR和IP处理,UP处理提高了土壤过氧化氢酶活性,脲酶活性则相反,UR和IP处理间土壤脲酶和过氧化氢酶活性差异较小;3种林下植被管理措施下,土壤酸性磷酸酶活性高低排序为IP>UR>UP,多酚氧化酶活性高低排序为UP>IP>UR;林下植被不同管理措施间土壤脲酶、蔗糖酶和多酚氧化酶活性差异较大,其中,多酚氧化酶对于林下植被管理措施的响应更灵敏,且能反映于较深土层;除酸性磷酸酶活性外,其余土壤酶活性均具明显表聚性,且随土层加深而递减。有机质和水解性氮含量与各种土壤酶活性具有极显著或显著正相关;有效磷含量与蔗糖酶、脲酶和酸性磷酸酶活性呈极显著正相关;速效钾含量与蔗糖酶、过氧化氢酶和多酚氧化酶活性呈极显著正相关;土壤物理性质与土壤酶活性相关性较弱。将土壤酶活性作为土壤质量指标之一,结合土壤物理性质和化学性质,通过主成分分析提取出3个主成分,反映了原信息量的75.31%。林下植被不同管理措施的土壤质量指数排序均为:IP>UP>UR。 结论 培育杉木大径材林分中,林下套种楠木的林下植被管理措施对于保持和提升土壤质量效果最佳,其次为林下植被保留措施,林下植被去除措施的效果较差。 Abstract:Objective To study the effects of different understory vegetation management measures on soil enzyme activities and quality in Cunninghamia lanceolata large-diameter industrial plantation. Method Taking Cunninghamia lanceolata large-diameter industrial plantation as the research object, This study analyzed soil enzyme activities of plantation under three kinds of understory vegetation management measures, i.e. understory vegetation protection (UP), understory vegetation removal (UR) and understory interplanting (IP). The integrated quantitative evaluation on soil quality was made by principle components analysis taking soil enzymes as soil biological activity indexes combining with soil physical and chemical characteristics. Result The IP increased the sucrase activity in the soils with the depth of 0-20 cm, and there was no obvious difference between the sucrase activity in the soils with the depths of 20-40 and 40~60 cm under these understory vegetation management measures. Compared with UR and IP, the UP showed better effect in increasing soil catalase activity and decreasing soil urease activity, and the soil urease and catalase activity under UR and IP showed no obvious difference. The ranking of soil acid phosphatase activity under the three kinds of understory vegetation management measures was IP>UR>UP, and the ranking of soil polyphenol oxidase activity was UP>IP>UR. The activities of urease, sucrase and polyphenol oxidase in soil under different understory vegetation management measures showed a big difference, and the response of polyphenol oxidase to understory vegetation management measures was more sensitive and was reflected in deeper soil layer. Except acid phosphatase activity, the activities of other soil enzyme showed a obvious property of surface-aggregation, and decreased with the deepening of soil layer. The contents of organic matter and hydrolyzed nitrogen showed extremely significant or significant positive correlation with the activities of the 5 kinds of soil enzyme, the content of available phosphorus showed extremely significant positive correlation with the activities of sucrase, urease and acid phosphatase, and the content of available potassium showed extremely significant positive correlation with the activities of sucrase, catalase and polyphenol oxidase, the activities of soil enzymes were less dependent on the soil physical properties. Soil enzyme activity was taken as one of soil quality indexes, combining with soil physical and chemical characteristics, three principal components were extracted through principal component analysis, reflecting 75.31% of the original information amount. The ranking of soil quality index among different understory vegetation management measures was IP>UP>UR. Conclusion To sum up, in the large diameter Cunninghamia lanceolata plantation, the effect of planting Phoebe bourmei under the plantation is the best to maintain and improve the soil quality, followed by protecting understory vegetation, and the effect of understory vegetation removal is poor. -
表 1 林下植被不同管理措施的样地基本情况
Table 1. Basic information of stands under different understory vegetation management measures
措施
Treatment坡向
Aspect坡位
Position坡度
Slope/(°)林分密度
Stand density/(株·hm−2)平均树高
Average height/m平均胸径
Average diameter/cmUP 西南WS 中 31 958 22.68±2.70 31.86±4.29 UR 南S 中 35 983 22.30±2.12 30.26±3.17 IP 西W 中 34 928 22.32±1.97 31.75±3.92 注:表中数值为平均值±标准误,下同。
Note: Values in the table are average ± standard error, the same blow.表 2 林下植被不同管理措施的样地林下植被
Table 2. Understory vegetation of stands under different understory vegetation management measures
措施
Treatment灌木
Shrub layer草本层
Herb layer总盖度
Total
coverage
/%物种名称
Species平均高度
Average
height/m平均胸径
Average
diameter/cm重要值
Important
value/%物种名称
Species平均高度
Average
height/m盖度
Coverage
/%频度
Frequency重要值
Important
value/%鲜生物量
Fresh
biomass/
(t·hm−2)UP 紫麻
Oreocnide
frutescenssp2.31 2.22 23.87 薄盖短肠蕨
Allantodia
hachijoensis0.70 0.45 0.67 8.25 6.70 92 粗叶榕
Ficus hirta1.93 1.70 17.49 江南短肠蕨
Allantodia
metteniana0.44 0.19 0.73 6.14 杜虹花
allicarpa
formosana1.43 0.61 15.36 三叶青
Tetrastigma
hemsleyanum0.46 0.26 0.60 5.11 IP 楠木
Phoebe bourmei2.98 2.46 100 深绿卷柏
Selaginella
doederleinii0.21 0.19 0.67 9.78 6.29 88 薄盖短肠蕨
Allantodia
hachijoensis0.54 0.38 0.87 7.13 金星蕨Parathelypteris glanduligera 0.33 0.18 0.87 5.73 注:表中所列重要值为前3的植物种。
Note: The table lists the plants with the first 3 important values.表 3 林下植被管理措施对于土壤酶活性的影响
Table 3. Soil enzyme activity under understory vegetation management measures
措施
Treatment土层
Soillayer/ cm蔗糖酶
Sucrase/(mg·g−1)脲酶
Urease/(mg·g−1)多酚氧化酶
Polyphenol oxidase/(mg·g−1)过氧化氢酶
Catalase/(mg·g−1)酸性磷酸酶
Acid phosphatase/(mg·g−1)UP 0~20 39.08±7.05ABa 2.38±0.68Aa 0.26±0.05Aa 18.45±3.22Aa 0.46±0.02Ba 20~40 25.92±4.25Ab 1.33±0.67Bb 0.15±0.02Ab 17.19±2.63Aa 0.45±0.23Ba 40~60 21.35±5.11Ab 1.29±0.27Ab 0.12±0.02Ab 16.68±4.79Aa 0.46±0.07Aa UR 0~20 30.39±6.02Bb 2.98±0.98Aa 0.10±0.07Ca 17.61±4.34Aa 0.48±0.04Ba 20~40 25.73±2.12Aab 2.37±0.60Aab 0.07±0.01Bb 15.12±3.19Ba 0.49±0.95ABa 40~60 19.32±6.21Ab 1.54±0.60Ab 0.04±0.04Bb 14.52±2.55Ab 0.47±0.12Aa IP 0~20 49.97±10.72Aa 3.05±0.33Aa 0.17±0.03Ba 18.24±4.90Aa 0.54±0.14Aa 20~40 30.82±2.31Ab 2.31±0.42Aab 0.11±0.06ABb 15.21±2.36Ba 0.53±0.16Aa 40~60 18.19±4.22Ac 1.39±0.34Ab 0.10±0.04ABb 13.39±3.58Ba 0.48±0.07Aa 注:不同大写字母表示林下植被不同管理措施同一土层酶活性差异显著(p<0.05);不同小写字母表示同一林下植被管理措施不同土层酶活性差异显著(p<0.05)。
Notes: Different capital letters indicated significant differences in enzyme activity in the same soil layer under different understory vegetation management measures (p<0.05). Different lowercase letters indicate significant differences in enzyme activity in different soil layers under the same understory vegetation management measure (p<0.05).表 4 林下植被不同管理措施培育杉木大径材林分土壤物理性质
Table 4. Soil physical properties of Cunninghamia lanceolata large-diameter timber cultivated forestunder different understory vegetation management measures
措施
Treatment土层
Soil layer/cm容重
Bulk density/(g·m−3)含水率
Moisture content/%总孔隙度
Total porosity/%毛管孔隙度
Capillary porosity/%非毛管孔隙度
Noncapillary porosity/%UP 0~20 1.21±0.05 25.4±2.78 50.2±2.35 39.5±5.91 10.7±2.32 20~40 1.24±0.05 24.4±1.17 45.8±3.12 39.3±5.36 6.5±0.03 40~60 1.36±0.02 23.2±0.67 45.0±5.72 36.3±3.79 5.4±0.17 UR 0~20 1.24±0.02 24.7±0.98 46.5±2.57 36.8±6.74 10.8±1.24 20~40 1.37±0.12 24.5±1.20 44.2±3.51 36.7±2.09 8.8±0.95 40~60 1.38±0.01 20.6±2.60 44.1±1.74 36.0±1.00 8.0±0.82 IP 0~20 1.02±0.27 22.7±0.54 53.2±2.35 41.6±4.90 11.6±2.44 20~40 1.23±0.01 20.4±0.62 50.7±7.28 41.5±10.96 9.2±1.06 40~60 1.26±0.02 20.1±0.34 44.5±1.83 35.4±0.58 9.1±2.17 表 5 林下植被不同管理措施培育杉木大径材林分土壤化学性质
Table 5. Soil chemical properties of Cunninghamia lanceolata large-diameter timber cultivated forest under different understory vegetation management measure
措施
Treatment土层
Soil layer/cm有机质
Organic content/
(g·kg−1)水解性氮
Hydrolysis nitrogen/
(mg·kg−1)有效磷
Available phosphate/
(mg·kg−1)速效钾
Rapidly availablepotassium/
(mg·kg−1)UP 0~20 32.72±2.57 7.68±0.33 0.57±0.27 62.99±2.61 20~40 28.93±2.47 5.86±0.39 0.47±0.03 52.44±3.44 40~60 19.07±0.22 5.70±0.44 0.42±0.04 49.82±1.99 UR 0~20 29.83±0.90 8.31±0.17 0.60±0.05 43.98±2.03 20~40 23.34±1.63 5.96±0.58 0.53±0.10 32.11±1.59 40~60 18.13±1.49 5.83±0.20 0.48±0.01 28.49±1.16 IP 0~20 31.77±0.40 9.69±0.86 0.79±0.03 52.58±3.82 20~40 27.31±1.28 6.47±0.73 0.65±0.01 44.52±2.90 40~60 19.74±0.92 6.35±0.36 0.60±0.03 35.15±1.73 表 6 土壤酶活性与土壤性质的相关性分析
Table 6. Pearson’s correlation coefficients between soil enzyme activities and properties
指标
Indicator有机质
Organiccontent水解性氮
Hydrolysis
nitrogen有效磷
Available
phosphate速效钾
Rapidly
available
potassium容重
Bulkdensity含水率
Moisture
content总孔隙度
Totalporosity毛管孔隙度
Capillary
porosity非毛管孔隙度
Noncapillary
porosity蔗糖酶Suarese 0.744** 0.703** 0.605** 0.532** −0.805** 0.224 0.482* 0.372 0.496** 脲酶Urease 0.599** 0.603** 0.528** 0.261 −0.463* 0.489** 0.352 0.376 0.609** 过氧化氢酶Catalase 0.586** 0.409* 0.209 0.605** −0.409* 0.374 0.398* 0.264 −0.027 酸性磷酸酶Acid phosphatase 0.395* 0.409* 0.744** 0.016 −0.358 0.011 0.652** 0.484* 0.560** 多酚氧化酶Polyphenol oxidase 0.679** 0.494** 0.342 0.828** −0.549** 0.390* 0.341 0.375 0.286 注Note:** P <0.01,* P <0.05.
表 7 土壤质量因子主成分的贡献率及因子载荷、权重
Table 7. Eigenvalue and contribution rate of principal factors
指标
Indicator主成分因子载荷
Principal component factor loadingPC1 PC2 PC3 蔗糖酶 Suarese 0.480 0.454 0.645 脲酶 Urease 0.710 0.232 0.395 过氧化氢酶 Catalase −0.030 0.725 0.277 酸性磷酸酶 Acid phosphatase 0.880 −0.069 0.205 多酚氧化酶 Polyphenol oxidase 0.188 0.796 0.342 有机质 Organic content 0.399 0.649 0.520 水解性氮 Hydrolysis nitrogen 0.309 0.212 0.805 有效磷 Available phosphate 0.643 −0.043 0.708 速效钾 Rapidly available potassium 0.106 0.882 0.258 容重 Bulk density −0.226 −0.287 −0.876 含水率 Moisture content 0.171 0.714 −0.184 总孔隙度 Total porosity 0.734 0.311 0.143 毛管孔隙度 Capillary porosity 0.626 0.359 0.085 非毛管孔隙度 Noncapillary porosity 0.742 0.018 0.392 特征根值 Eigenvalue 3.778 3.487 3.277 百分比 Percent/% 26.99 24.91 23.41 累积贡献率 Cumulative percent/% 26.99 51.90 75.31 表 8 土壤指标成分得分系数矩阵
Table 8. Component score coefficient matrix of soil index
指标
Indicator因子得分系数
Component score coefficient matrixPC1 PC2 PC3 蔗糖酶 Suarese 0.002 0.043 0.172 脲酶 Urease 0.207 −0.010 −0.025 过氧化氢酶 Catalase −0.140 0.238 0.059 酸性磷酸酶 Acid phosphatase 0.365 −0.102 −0.148 多酚氧化酶 Polyphenol oxidase −0.056 0.245 0.014 有机质 Organic content −0.005 0.146 0.085 水解性氮 Hydrolysis nitrogen −0.127 −0.074 0.377 有效磷 Available phosphate 0.081 −0.172 0.249 速效钾 Rapidly available potassium −0.072 0.298 −0.028 容重 Bulk density 0.195 0.054 −0.438 含水率 Moisture content 0.116 0.308 −0.305 总孔隙度 Total porosity 0.300 0.066 −0.210 毛管孔隙度 Capillary porosity 0.265 0.100 −0.220 非毛管孔隙度 Noncapillary porosity 0.233 −0.095 0.001 -
[1] 杜 忠, 蔡小虎, 包维楷, 等. 林下层植被对上层乔木的影响研究综述[J]. 应用生态学报, 2016,27(11):963-972. [2] Chen L M, Lei D, Man X L. Soil enzyme activities and plant diversity of undergrowth in spruce-fir forest[J]. Journal of Northeast Forestry University, 2009, 37(3): 58-61. [3] 何艺玲, 傅懋毅. 人工林林下植被的研究现状[J]. 林业科学研究, 2002, 15(6):727-733. doi: 10.3321/j.issn:1001-1498.2002.06.015 [4] Qiao Y, Miao S, Silva L C R, et al. Understory species regulate litter decomposition and accumulation of C and N in forest soils: A long-term dual-isotope experiment[J]. Forest Ecology and Management, 2014, 329: 318-327. doi: 10.1016/j.foreco.2014.04.025 [5] Lin G G, Mao R, Zhao L, et al. Litter decomposition of a pine plantation is affected by species evenness and soil nitrogen availability[J]. Plant and Soil, 2013, 373(1-2): 649-657. doi: 10.1007/s11104-013-1832-8 [6] Tripathi S K, Muttineni R, Singh S K. Extra precision docking, free energy calculation and molecular dynamics simulation studies of CDK2 inhibitors[J]. Journal of Theoretical Biology, 2013, 334(23): 87-100. [7] Tripathi S K, Sumida A, Ono K, et al. The effects of understorey dwarf bamboo (Sasa kurilensis) removal on soil fertility in a Betula ermanii forest of northern Japan[J]. Ecological Research, 2006, 21(2): 315-320. doi: 10.1007/s11284-005-0119-9 [8] Tripathi S K, Sumida A, Shibata H, et al. Growth and substrate quality of fine root and soil nitrogen availability in a young Betula ermanii forest of northern Japan: Effects of the removal of understory dwarf bamboo (Sasa kurilensis)[J]. Forest Ecology and Management, 2005, 212(1-3): 278-290. doi: 10.1016/j.foreco.2005.03.030 [9] Xiong Y M, Xia H P, Li Z A, et al. Impacts of litter and understory removal on soil properties in a subtropical Acacia mangium plantation in China[J]. Plant and Soil, 2008, 304: 179-188. doi: 10.1007/s11104-007-9536-6 [10] Zhao J, Wang X L, Shao Y H, et al. Effects of vegetation removal on soil properties and decomposer organisms[J]. Soil Biology and Biochemistry, 2011, 43(5): 954-960. doi: 10.1016/j.soilbio.2011.01.010 [11] Zornoza R, Guerrero C, Mataix-Solera J, et al. Assessing air-drying and rewetting pre-treatment effect on some soil enzyme activities under Mediterranean conditions[J]. Soil Biology and Biochemistry, 2006, 38(8): 2125-2134. doi: 10.1016/j.soilbio.2006.01.010 [12] Jha D K, Sharma G D, Mishra R R. Soil microbial population numbers and enzyme activities in relation to altitude and forest degradation[J]. Soil Biology and Biochemistry, 1992, 24(8): 761-767. doi: 10.1016/0038-0717(92)90250-2 [13] 杨万勤, 王开运. 森林土壤酶的研究进展[J]. 林业科学, 2004, 40(2):152-159. doi: 10.3321/j.issn:1001-7488.2004.02.027 [14] 颜路明, 郭祥泉. 盐碱胁迫对香樟幼苗根际土壤酶活性的影响[J]. 土壤, 2017, 49(4):733-737. [15] Guolei L I, Yong L, Bei G, et al. Seasonal response of soil enzyme activity to thinning intensity of aerial seeded, stands[J]. Frontiers of Forestry in China, 2008, 3(3): 286-292. doi: 10.1007/s11461-008-0061-4 [16] 陈春林, 周国英, 吴 毅, 等. 湖南黄丰桥林场杉木速生丰产林健康评价[J]. 生态学杂志, 2012, 31(11):2872-2876. [17] 国家林业局. 中国林业统计年鉴[M]. 北京: 中国林业出版社, 2013. [18] 谭秀凤. 中国木材供需预测模型及发展趋势研究[D]. 北京: 中国林业科学研究院, 2011. [19] 国家林业局. 中华人民共和国林业行业标准—森林土壤分析方法[M]. 北京: 中国标准出版社, 2000. [20] 关松荫. 土壤酶及其研究方法[M]. 北京: 农业出版社, 1986: 1-151. [21] 王长庭, 龙瑞军, 王根绪, 等. 高寒草甸群落地表植被特征与土壤理化性状和土壤微生物之间的相关性研究[J]. 草业学报, 2010, 19(6):25-34. doi: 10.11686/cyxb20100604 [22] Groffman P M, McDowell W H, Myers J C, et al. Soil microbial biomass and activity in tropical riparian forests[J]. Soil Biology and Biochemistry, 2001, 33(10): 1339-1348. doi: 10.1016/S0038-0717(01)00039-6 [23] Chapin Ⅲ F S. Nitrogen and phosphorus nutrition and nutrient cycling by evergreen and deciduous understory shrubs in an Alaskan black spruce forest[J]. Canadian Journal of Forest Research, 1983, 13(5): 773-781. doi: 10.1139/x83-107 [24] 傅静丹, 薛 立, 郑卫国, 等. 加勒比松凋落物对土壤性状的影响[J]. 林业科学研究, 2009, 22(2):303-307. doi: 10.3321/j.issn:1001-1498.2009.02.026 [25] 杨 洋, 王继富, 张心昱, 等. 凋落物和林下植被对杉木林土壤碳氮水解酶活性的影响机制[J]. 生态学报, 2016, 36(24):8102-8110. [26] 郑琳琳, 赵 琼, 曾德慧. 林下植被去除对樟子松人工林土壤酶活性的影响[J]. 生态学杂志, 2017, 36(11):3056-3063. [27] Kotroczó Z, Veres Z, István F, et al. Soil enzyme activity in response to long-term organic matter manipulation[J]. Soil Biology and Biochemistry, 2014, 70(2): 237-243. [28] Liu Z, Wu J, Zhou L, et al. Effect of understory fern (Dicranopteris dichotoma) removal on substrate utilization patterns of culturable soil bacterial communities in subtropical Eucalyptus plantations[J]. Pedobiologia, 2012, 55(1): 7-13. doi: 10.1016/j.pedobi.2011.07.014 [29] Zhao J, Wan S, Li Z A, et al. Dicranopteris-dominated understory as major driver of intensive forest ecosystem in humid subtropical and tropical region[J]. Soil Biology and Biochemistry, 2012, 49: 78-87. doi: 10.1016/j.soilbio.2012.02.020 [30] Wu J, Liu Z, Wang X, et al. Effects of understory removal and tree girdling on soil microbial community composition and litter decomposition in two Eucalyptus plantations in South China[J]. Functional Ecology, 2011, 25(4): 921-931. doi: 10.1111/j.1365-2435.2011.01845.x [31] 杨万勤, 钟章成, 陶建平, 等. 缙云山森林土壤酶活性与植物多样性的关系[J]. 林业科学, 2001, 37(4):124-128. doi: 10.3321/j.issn:1001-7488.2001.04.020 [32] 柴旭光. 油松不同林分组成对林地土壤酶活性的影响[J]. 防护林科技, 2016(4):32-33. [33] 陈爱玲, 陈青山, 蔡丽萍. 杉木拟赤杨混交林土壤肥力的研究[J]. 土壤与环境, 2000, 9(4):284-286. [34] Song Y, Song C, Yang G, et al. Changes in labile organic carbon fractions and soil enzyme activities after marshland reclamation and restoration in the Sanjiang plain in northeast China[J]. Environmental Management, 2012, 50(3): 418-426. doi: 10.1007/s00267-012-9890-x [35] Chen X, Chen H Y H, Chen X, et al. Soil labile organic carbon and carbon-cycle enzyme activities under different thinning intensities in Chinese fir plantations[J]. Applied Soil Ecology, 2016, 107: 162-169. doi: 10.1016/j.apsoil.2016.05.016 [36] 胡海波, 张金池, 高智慧, 等. 岩质海岸防护林土壤微生物数量及其与酶活性和理化性质的关系[J]. 林业科学研究, 2002, 15(1):83-87. doi: 10.3321/j.issn:1001-1498.2002.01.013 -




 下载:
下载: