-
中间锦鸡儿(Caragana intermedia Kuang et H.C.Fu),俗称柠条,为豆科、锦鸡儿属旱生落叶灌木,分布在我国西部荒漠草原、干旱草原地区,是这些地区的建群植物种[1]。中间锦鸡儿耐寒、耐热、抗干旱、耐贫瘠,对沙地环境有很强的适应能力,可防风固沙、保持水土、改善局部小环境[2]。近年来,对中间锦鸡儿的研究日益增多,特别是对其干旱适应性方面。
目前,对中间锦鸡儿干旱适应性的研究大致可分为生理机制研究和分子机制研究两类。生理研究发现,柠条气孔开度较小,全天开启进行不间断的蒸腾,这与一般植物中气孔白天开、晚间闭有明显的差异[3];当土壤、叶片间的水分关系紧张系数低于1.0以后,柠条蒸腾速率会自动调低,以避免大量失水[4],另有文章进一步证实柠条的抗旱性与蒸腾速率、小叶水势和叶含水率呈负相关[5];在受到干旱胁迫时,柠条胞间二氧化碳体积分数先轻微下降,而后上升,这与净光合速率的变化趋势相反[6];在不同CO2浓度下,干旱胁迫均会造成柠条生物量的减少,且根、茎、叶中生物量下降的程度不同[7-8]。分子机制方面,柠条锦鸡儿水分胁迫下叶片均一化全长cDNA文库及叶片抑制性差减杂交文库已被构建[9-10],且已经筛选出适合干旱胁迫下进行基因表达分析的内参基因[11]。综上,目前对柠条抗旱性的研究大多集中在生理方面,而对分子方面的研究相对较少。为探究中间锦鸡儿适应干旱的分子机理及进行抗性育种,寻找中间锦鸡儿抗旱相关基因并对其功能进行解析显得十分必要。
大豆miR2118被证实与干旱胁迫显著相关,推测miR2118通过对其靶基因TG01的调控参与到大豆的干旱胁迫响应[12]。通过分析中间锦鸡儿小RNA测序数据,我们获得了中间锦鸡儿miR2118全长[13],并预测了其靶基因cDNA序列片段。为了明确中间锦鸡儿miR2118靶基因是否参与干旱胁迫响应,我们克隆了其靶基因CiDR1的cDNA全长序列,并分析了它在中间锦鸡儿遭受干旱胁迫时的表达模式。本研究为了解中间锦鸡儿CiDR1基因表达与耐旱性的关系提供了借鉴,并为进一步进行中间锦鸡儿抗性育种提供了候选基因。
-
中间锦鸡儿种子于2013年7月中旬采集自中国林科院玉泉苗圃,用无菌水反复清洗后种植于含有蛭石、珍珠岩的营养土中于人工培养箱中培养(昼温28℃,夜温25℃,相对湿度60%)。生长3周后,将幼苗从土壤中取出,清水洗净后分根、茎、叶,分别保存于液氮中,用于基因克隆和幼年期基因表达分析。
从中国林科院玉泉苗圃5年生中间锦鸡儿植株上取根、茎、叶、花和种子,分别于液氮中保存,用于成年期营养器官和生殖器官中基因表达分析。
中间锦鸡儿种子在人工培养箱中培养3周后,选取长势一致的幼苗,用无菌水清洗干净后转入1/2 Hoagland ’s营养液中培养5天,期间每天更换培养液,以保证氧气充足。5天后将小苗取出,进行干旱胁迫处理。具体处理办法[12]:将小苗用清水洗净后,先用滤纸将表面残余水分吸干,而后用双层灭菌滤纸包裹根部放入干净锥形瓶中,于室温下放置。放置0、2、4、8、12、24、48 h后,分根、茎、叶取材,保存于液氮中,用于干旱胁迫下基因的表达分析。
-
将中间锦鸡儿根、茎、叶混合样于液氮中磨碎后,称取材料100 mg,按照TIANGEN公司的RNAprep Pure Plant Kit说明书进行总RNA的提取。样品总RNA的浓度和纯度用ND-1000分光光度计(Nanodrop Technologies,USA)进行检测;RNA的完整性用1.5%的凝胶电泳检测。cDNA第一链的合成采用RevertAid First Strand cDNA Synthesis Kit(Thermo,#K1621)进行,反应产物用于目的基因全长cDNA的扩增和基因表达分析。用于Rapid Amplification of cDNA Ends(RACE)的cDNA采用SMARTTM RACE cDNA Amplification Kit(clontech,USA)进行合成。
-
将已有中间锦鸡儿序列片段经PCR检测,确认真实表达后,用Primer 5.0设计引物,使用Advantage2 PCR Kit(Clontech,USA)进行RACE扩增。特异引物5’-GTCCCCGTTAACCTCCGCACATCAA-3’和5’-GGCAAAACAGATCGACCCTTGTCCT-3’分别为5’RACE的第一轮引物和第二轮引物。第一轮反应条件为94℃ 2 min预变性,循环1次;之后94℃ 30 s变性,65℃ 30 s退火;72℃ 2 min延伸,循环30次。第二轮反应程序不变,退火温度改为61℃。特异引物5’-GGATTCATGGAGTTGGTGGGA-3’和5’-TGGGTTGCCATATCTCCAAA-3’分别为3’ RACE的第一轮引物和第二轮引物。第一轮反应条件为94℃ 2 min预变性,循环1次;94℃ 30 s,65℃ 30 s,72℃ 3 min,循环30次。第二轮反应程序不变,退火温度改为66℃。电泳检测后,将扩增到的目的产物回收、连入pEASY-T1载体中送公司测序。
将3’RACE和5’RACE测序获得的片段与中间片段拼接后,设计引物5’-TTCATCCTTTTGCTTACAAGAAC-3’和5’-GACATGGACTCCGATTATCTCACA-3’,扩增cDNA全长进行验证。反应使用超保真DNA聚合酶(NEW ENGLAND Biolabs)进行,扩增条件为98℃初始变性30 s,98℃变性8 s,59℃复性20 s,72℃延伸30 s,循环35次;最后72℃终延伸8 min。电泳检测后,将扩增到的目的产物回收、连入pEASY-Blunt载体中送公司测序。
-
使用ORF Finder程序(http://www.ncbi.nlm.nih.gov/gorf/gorf.html)分析CiDR1基因开放阅读框,并用Pfam(http://pfam.xfam.org/)对预测到的CiDR1蛋白序列进行结构域分析。编码蛋白的理化性质和亲疏水性分别采用Protparam和ProtScale(http://web.expasy.org)程序进行分析,跨膜结构域和信号肽的查找及亚细胞定位分别使用TMHMM2.0、SignalP 4.1和TargetP 1.1(http://www.cbs.dtu.dk/services)进行,使用DNAMAN进行多序列比对及同源性分析,最后采用CLUSTAL X和Mega5.05软件进行进化树构建。
-
中间锦鸡儿CiDR1基因在生长发育各个时期、不同器官内的转录表达及干旱胁迫下的表达均以UNK2[11]为内参基因,采用SYBR Green荧光染料法,在BioRad CFX96TM实时荧光定量PCR仪上进行。定量PCR引物为5’-GAACGGGGATGGATATGAGC-3’和5’-CCAACTCCATGAATCCCTACCA-3’。根据SYBR Premix Ex TaqTM (TaKaRa)试剂盒说明书配制反应体系,每个反应做三次重复;反应程序为95℃预变性30 s,95℃变性5 s,59.5℃退火34 s,循环40次,之后95℃ 5 s,60℃ 30 s,反应结束后做溶解曲线分析。实验数据用BioRad实时荧光定量PCR系统软件和Excel 2010进行分析。
-
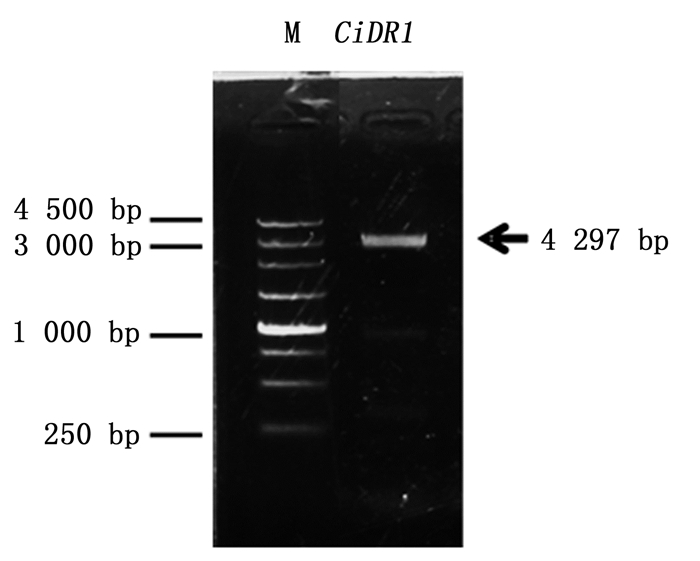
通过3’ RACE和5’RACE巢式扩增,分别获得3 383 bp和463 bp的序列片段,与中间片段拼接之后,得到了CiDR1基因的全长cDNA序列共计4 297 bp,全长引物验证结果如下(图 1)。将CiDR1基因的cDNA序列提交至GenBank,登录号为KP277100。分析表明,CiDR1基因的cDNA序列包含一个完整的开放阅读框,为3 732 bp,共编码1 243个氨基酸残基。经psRNATarget(http://plantgrn.noble.org/psRNATarget/)分析,这段序列中含有miR2118的结合位点(图 2),表明CiDR1可能是miR2118的靶基因。
ExPASy ProtParam分析表明,CiDR1蛋白的分子量约为141 069.6 Da,等电点为6.35;氨基酸组成中亮氨酸所占比例最高,达到14.1%,其余氨基酸含量均在10% 以下;CiDR1蛋白不稳定指数为42.91。ProtScale软件的分析结果表明CiDR1蛋白平均亲水性为-0.126,该蛋白既有亲水区又有疏水区,且亲水性较强而疏水性较弱。跨膜预测显示CiDR1无跨膜结构域。经SignalP4.1的神经网络算法预测CiDR1蛋白的信号肽,发现其信号肽分值为0.098,远低于0.5据此可以推测该蛋白不具备信号肽,可能为非分泌蛋白。亚细胞定位分析表明,该蛋白极有可能定位于细胞质中。
-
Pfam结构域预测结果显示,CiDR1蛋白具有TIR-NBS-LRR家族共有的TIR、NB-ARC、LRR等几个保守结构域[14-15],据此可进一步确认CiDR1为NBS型抗病基因。保守结构域搜索发现,其与GenBank中已登录的植物TIR-NBS-LRR类抗病基因具有一定同源性(图 3)。利用保守结构域在NCBI中BLAST其他植物中已知的TIR-NBS类抗病基因,剔除亲缘关系较远的物种后,最终选定14种植物,包括苜蓿(Medicago truncatula Gaertn.)、大豆(Glycine max (L.) Merr.)、芸豆(Phaseolus vulgaris L.)、鹰嘴豆(Cicer arietinum L.)、山荆子(Malus baccata (L.) Borkh.)、黄瓜(Cucumis sativus L.)、拟南芥(Arabidopsis thaliana (L.) Heynh.)等进行系统进化分析。

图 3 CiDR1基因保守结构域与其他已知物种抗病基因保守结构域的比较
Figure 3. Comparison of conserved domain of CiDR1 in C. intermedia with other species
采用CLUSTAL X进行多序列比对,将比对结果在Mega5.05软件中构建系统发育树。分析结果表明,中间锦鸡儿和苜蓿、鹰嘴豆亲缘关系最近,其次是大豆和芸豆(图 4),这五个物种同属于豆科蝶形花亚科,该结果与植物形态学分类结果相一致。与豆科植物在同一分支上的是黄瓜、可可(Theobroma cacao L.)和山荆子,说明它们中的DR1基因相似性较高。而在系统发育树中距离中间锦鸡儿CiDR1基因最远的是山白松(Pinus monticola Douglas ex D. Don)、火炬松(Pinus taeda L.)和毛果杨(Populus rihocarpa Torr. & A.Gray ex. Hook.),它们从起源处就处于不同分支,尤其是火炬松,其DR1基因与CiDR1基因相似性只有19.3%。
-
定量PCR分析表明,CiDR1基因在中间锦鸡儿幼年期及成年期的各个器官中均有表达,且在每个器官中,幼年期的表达量都低于成年期(图 5);无论是在幼年期还是在成年期,茎中的表达量相对较低,叶中的表达量相对较高(图 5)。干旱胁迫后,CiDR1表达量迅速下降,到第8小时左右表达量有小幅回升,到第12小时表达量又下降到比之前更低的水平;且在中间锦鸡儿根、茎和叶中,CiDR1基因响应干旱胁迫时的表达变化呈现相似的模式(图 6 (a),(b),(c))。
-
干旱严重影响到植物的生长和发育,有报道称干旱对作物造成的损失在所有非生物胁迫中占首位[16-17]。为最大限度地减轻干旱胁迫造成的伤害,植物中表达了一系列干旱适应相关基因,目前已克隆的干旱胁迫相关基因大致可分为两类[18-21]:一类基因编码的蛋白质在信号传导和基因表达中起调节作用,为调控基因,包括:转录因子,如DREB[22-23]等;蛋白激酶,如MAP激酶等[24]和其他一些信号分子如钙结合蛋白CDPK[25]等。另一类基因编码的蛋白质对干旱胁迫中的植物起直接的保护作用,为功能基因。这类基因包括脱水响应基因RD29A,RD29B等[26];水通道蛋白MIP等[27];渗透蛋白LEA蛋白等[28]及COR、KIN、LTI等晚期响应基因[29-30]。这些基因在植物遭受干旱胁迫时,其表达被快速诱导或抑制,以保障植物的生存。本研究发现,中间锦鸡儿在干旱胁迫后仅2 h,其根、茎和叶中CiDR1的表达均明显下降,表明CiDR1的表达受干旱抑制,可能与中间锦鸡儿适应干旱相关。值得关注的是,幼年期植物根、茎、叶中CiDR1的表达水平均低于成年期植物对应器官中的表达,表明CiDR1的转录受发育阶段调控。
CiDR1基因编码的产物为TIR-NBS-LRR类抗病相关蛋白。该蛋白的NBS结构域包含一些非常保守的基序,如p-loop(GGVGKTT)、Kinase-2a(VLDD)、Kinase-3a(GSRII)及跨膜区GLPL[31],因此常被用于植物抗病基因的识别和分类[32]。由生物信息学分析可以初步认定,CiDR1基因的编码产物结构不稳定,为亲水蛋白,无跨膜结构且属于非分泌蛋白,定位于细胞质中。编码氨基酸序列多重比对分析发现,CiDR1与其他植物有很高的同源性,特别是豆科植物苜蓿、鹰嘴豆、大豆等,相似性均达到60%以上,表明CiDR1可能与豆科植物苜蓿、大豆等有同一起源。有报道称大豆NBS抗病基因TG01受到干旱胁迫诱导表达[12],这更加为我们的实验结果提供了佐证。
植物抗旱基因的发掘及抗旱机制的研究一直是人们关注的焦点。中间锦鸡儿对干旱胁迫的响应涉及到体内各类物质代谢、能量代谢、信号转导和物质运输等多个方面[10]。本研究对CiDR1基因进行了初步研究,下一步对其功能的深入分析将有利于我们解析中间锦鸡儿干旱适应性的分子机理。
-
本研究中,中间锦鸡儿在干旱胁迫后其根、茎和叶中CiDR1的表达均明显下降,表明CiDR1的表达受干旱抑制,可能与中间锦鸡儿适应干旱相关,进一步研究发现CiDR1在根、茎、叶中的表达水平可能受发育阶段调控。相关研究为中间锦鸡儿抗旱基因工程和遗传改良提供了参考,并为进一步揭示其抗旱分子调控机制奠定了基础。
中间锦鸡儿CiDR1的克隆及干旱胁迫下的表达分析
Cloning of CiDR1 from Caragana intermedia and Its Expression under Drought Stress
-
摘要:
目的 研究并了解中间锦鸡儿CiDR1基因功能及其对干旱胁迫的响应,为抗性育种提供候选基因。 方法 通过RACE技术从中间锦鸡儿中克隆CiDR1基因的cDNA全长,利用生物信息学分析软件对其基因结构及功能进行分析和预测。再通过qRT-PCR技术对干旱胁迫后的幼苗中的CiDR1表达模式进行研究。 结果 从中间锦鸡儿中克隆到CiDR1基因的cDNA全长共计4 297 bp,GenBank登录号为KP277100。生物信息学分析表明,预测的CiDR1蛋白序列中含有1 243个氨基酸残基,具有抗病基因特征结构域TIR、NB-ARC、LRR等,其等电点为6.35,不稳定指数为42.91,不具备信号肽,为非分泌蛋白,定位于细胞质中。定量PCR检测发现,CiDR1基因在幼年期的茎中表达量较低,在成年期的叶片中表达量较高;在干旱胁迫处理后的幼苗中,CiDR1表达水平有明显下降,表明该基因的表达受干旱抑制,可能与中间锦鸡儿适应干旱相关。 结论 中间锦鸡儿在干旱胁迫后其根、茎和叶中CiDR1的表达均明显下降,表明CiDR1的表达受干旱抑制,可能与中间锦鸡儿适应干旱相关,进一步研究发现CiDR1在根、茎、叶中的表达水平可能受发育阶段调控。 -
关键词:
- 中间锦鸡儿
- / CiDR1
- / TIR-NBS-LRR
- / 干旱
Abstract:Objective To study the function of CiDR1 gene in Caragana intermedia and its response to drought stress. Method The full-length cDNA sequence of CiDR1 gene was cloned using RACE methods, and then the bioinformatics analysis were carried out to predict gene structure. Finally, CiDR1 expression patterns under drought stress was investigated by qRT-PCR. Result The full-length cDNA sequence of CiDR1 gene was 4 297 bp. The deduced amino-acid sequence for CiDR1 was 1 243 amino acids long and contained toll/interluekin receptor (TIR), nucleotide binding site (NBS) and leucine-rich repeats (LRR) motif, which are the characteristics of plant resistance genes. The theoretical isoelectric point and instability index of CiDR1 protein was 6.35 and 42.91, respectively. Further analysis showed that CiDR1 protein had no signal peptide, was a non-secretary protein and located in the cytoplasmic matrix. The qPCR analysis showed that the CiDR1 transcripts were expressed strongly in adult leaves and weakly in juvenile stems, and after drought treatments the levels of CiDR1 transcripts decreased. Conclusion The results indicate that CiDR1 might play an role in response of C. intermedia to drought and could be used for molecular breeding. -
Key words:
- Caragana intermedia
- / CiDR1
- / TIR-NBS-LRR
- / drought
-
-
[1] 牛勇. 中间锦鸡儿的特性及发展前景[J]. 内蒙古林业, 2013, 3: 22-23. [2] 时永杰, 常根柱. 中间锦鸡儿[J]. 中兽医医药杂志, (专辑), 2003, 148-149. [3] 李录章. 花棒、柠条蒸腾作用的研究[J]. 内蒙古林业, 1999, (6): 33. [4] 杨文斌, 任建民. 柠条锦鸡儿沙柳蒸腾速率与水分关系分析[J]. 内蒙古林业科技, 1995, (3): 1-6. [5] 王盂本, 李洪建, 柴宝峰. 柠条的水分生理生态学特性[J]. 植物生态学报, 1996, 20(6): 494-501. [6] 王志会, 夏新莉, 尹伟伦. 不同种源的柠条锦鸡儿的生理特性与抗旱性[J]. 东北林业大学学报, 2007, 35(9): 27-32. doi: 10.3969/j.issn.1000-5382.2007.09.010 [7] 高素华, 郭建平. 毛乌素沙地优势种在高CO2浓度条件下对土壤干旱胁迫的响应[J]. 草业学报, 2003, 12(2): 36-39. doi: 10.3321/j.issn:1004-5759.2003.02.007 [8] Xu Z, Zhou G, Wang Y. Combined effects of elevated CO2 and soil drought on carbon and nitrogen allocation of the desert shrub Caragana intermedia[J]. Plant Soil, 2007, 301: 87-97. doi: 10.1007/s11104-007-9424-0 [9] 郭九峰, 孙国琴, 乔慧蕾. 水分胁迫柠条锦鸡儿叶片均一化全长cDNA文库的构建及EST分析[J]. 华北农学报, 2012, 27(3): 67-71. doi: 10.3969/j.issn.1000-7091.2012.03.013 [10] 杨杞, 张涛, 王颖, 等. 干旱胁迫下柠条锦鸡儿叶片SSH文库构建及CkWRKY1基因克隆[J]. 林业科学, 2013, 49(7): 62-68. [11] Zhu J, Zhang L, Li W, et al. Reference gene selection for quantitative real-time PCR normalization in Caragana intermedia under different abiotic stress conditions[J]. Plos One, 2013, 8, 1-10. [12] 罗中钦. 大豆逆境胁迫相关microRNA的发掘与验证[D]. 北京, 中国农科院作物研究所, 2011. [13] Zhu J, Li W, Yang W, et al. Identification of microRNAs in Caragana intermedia by highthroughput sequencing and expression analysis of 12 microRNAs and their targets under salt stress[J]. Plant Cell Reports, 2013, 32(9): 1339-1349. doi: 10.1007/s00299-013-1446-x [14] Andrew F B. Plant disease resistance genes: Function meets Structure[J]. The Plant Cell, 1996, 8, 1757-1771. doi: 10.2307/3870228 [15] Blake C M, Allan W D, Richard W M, et al. Plant disease resistance genes encode members of an ancient and diverse protein family within the nucleotide-binding superfamily[J]. The Plant Journal, 1999, 20(3): 317-332. doi: 10.1046/j.1365-313X.1999.t01-1-00606.x [16] Boyer J S. Plant productivity and environment[J]. Science, 1982, 218: 443-448. doi: 10.1126/science.218.4571.443 [17] 陈善福, 舒庆尧. 植物耐干旱胁迫的生物学机理及其基因工程研究进展[J]. 植物学通报, 1999, 16(5): 555-560. [18] Cattivelli L, Rizza F, Badeck F W, et al. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics[J]. Field Crops Research, 2008, 105: 1-14. doi: 10.1016/j.fcr.2007.07.004 [19] Holmberg N, Bulow L. Improving stress tolerance in plants by gene transfer[J]. Trends in Plant Science, 1998, 3(2): 61-66. doi: 10.1016/S1360-1385(97)01163-1 [20] Shinozaki K, Yamaguchi-Shinozaki K. Gene networks involved in drought stress response and tolerance[J]. Journal of Experimental Botany, 2007, 58(2): 221-227. [21] 康宗利, 杨玉红, 张立军. 植物响应干旱胁迫的分子机制[J]. 玉米科学, 2006, 14(2): 96-100. doi: 10.3969/j.issn.1005-0906.2006.02.033 [22] Liu Q, Kasuga M, Sakuma Y, et al. Two transcription factors, DREB1 and DREB2, with an EREBP/AP2 DNA-binding domain separate two cellular signal transduction pathways in drought- and low temperature-responsive gene expression, respectively, in Arabidopsis[J]. Plant Cell, 1998, 10: 1391-1406. doi: 10.1105/tpc.10.8.1391 [23] Nakashima K, Shinwari Z K, Sakuma Y, et al. Organization and expression of two Arabidopsis DREB2 genes encoding DRE-binding proteins involved in dehydration- and high-salinity-responsive gene expression[J]. Plant Molecular Biology Reporter, 2000, 42: 657-665. doi: 10.1023/A:1006321900483 [24] Jonak C, Pay A, Bogre L, et al. The plant homologue of MAP kinase is expressed in a cell cycle-dependent and organ-specific manner[J]. The Plant Journal, 1993, 3(4): 611-617. doi: 10.1046/j.1365-313X.1993.03040611.x [25] Saijo Y, Hata S, Kyozuka J, et al. Over-expression of a single Ca2+-dependent protein kinase confers both cold and salt/drought tolerance on rice plants[J]. The Plant Journal, 2000, 23(3): 319-327. doi: 10.1046/j.1365-313x.2000.00787.x [26] Kim J M, To T K, Ishida J, et al. Alterations of Lysine modifications on the Histone H3 N-Tail under drought stress conditions in Arabidopsis thaliana[J]. Plant Cell Physiology, 2008, 49(10): 1580-1588. doi: 10.1093/pcp/pcn133 [27] Yamada S, Katsuhara M, Kelly W B, et al. A family of transcripts encoding water channel proteins: Tissue-Specific expression in the common ice plant[J]. The Plant Cell, 1995, 7(8): 1129-1142. [28] Bahieldin A, Mahfouz H T, Eissa H F, et al. Field evaluation of transgenic wheat plants stably expressing the HVA1 gene for drought tolerance[J]. Plant Physiology, 2005, 123, 421-427. doi: 10.1111/j.1399-3054.2005.00470.x [29] Jaglo-Ottosen K R, Gilmour S J, Zarka D G, et al. Arabidopsis CBF1 overexpression induces COR genes and enhances freezing tolerance[J]. Science, 1998, 280: 104-106. doi: 10.1126/science.280.5360.104 [30] Kasuga M, Liu Q, Miura S, et al. Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor[J]. Nature Biotechnology, 1999, 17: 287-291. doi: 10.1038/7036 [31] Naomi O, Yuval E, llan P, et al. The I2C family from the wilt disease resistance locus I2 belongs to the nucleotide binding, leucine rich repeat superfamily of plant resistance genes[J]. The Plant Cell, 1997, 9, 521-532. [32] 王友红, 张鹏飞, 陈建群. 植物抗病基因及其作用机理[J]. 植物高技术通讯, 2005, l5(2): 71-78. -





 下载:
下载: