-
全球气候变化改变降雨量、降雨频率(天、周、月模式)和降雨强度,未来严重干旱发生的可能性增大[1-3]。近年,突然的和大范围的森林死亡、衰败被认为与干旱和温度升高有关[4];而森林和林地植物死亡致快速地和大尺度地生态系统结构和功能的改变[5]。水力失衡和碳饥饿假说是目前解释干旱导致树木死亡的主要机制[6]。水力失衡假说是指减少的土壤水分供应和高的蒸发需求导致木质部导管和根系产生空穴化,植物长距离水分运输受限,使植物发生不可逆的干化现象[7]。非结构性碳(NSC)是植物的光合产物,主要是可溶性糖和淀粉,其累加值为总非结构性碳(TNC)。TNC为植物生长繁殖和新陈代谢提供能量,对抵抗干扰和干扰后的恢复也有重要作用[8]。碳饥饿假说认为,长期的干旱胁迫会打破植物碳摄取与碳支出的平衡,当包括光合作用、可动员的TNC储备和水解供应的NSC不能抵消包括呼吸、生长和防御所需的NSC的时候,NSC的浓度会降低,当NSC降低到一定程度时,植物会因碳饥饿而死亡[8-10]。很多植物为了阻止水力失衡的发生关闭了气孔,但气孔关闭的代价是光合碳固定减少[7]。碳饥饿和水力失衡之间可能存在交互作用,因为栓塞修复可能需要利用木质部薄壁细胞内的糖降低栓塞导管的渗透势,进而对栓塞进行修复[11];而水分的有效性也影响NSC的生产和运输[8],并且在抵抗干旱过程中,NSC对渗透调节和维持细胞膨压等方面也有作用[12]。水力失衡与碳饥饿在树木死亡过程中哪一个起主要作用还不清楚,并且缺乏实例证据,特别是国内还未见这方面的报道,且国外已经报道的研究也大多采用盆栽控水方法[12-13],缺少森林原位的研究。
2013年4月笔者在河南省西南部的宝天曼锐齿栎(Quercus aliena var. acuteserrata Maxim.)天然次生林内建立了截雨样地(减少的是林内穿透雨),以伴生种三桠乌药(Lindera obtusiloba Bl. Mus. Bot.)为研究对象,探讨降雨减少对三桠乌药生理生态的影响,从水和碳的角度,探讨三桠乌药顶端枯死的原因,确定截雨处理后三桠乌药是否出现适应性的变化。
-
研究地点在河南省西南部的宝天曼国家级自然保护区内的宝天曼森林生态定位研究站,属伏牛山南坡(111°47′~112°04′E,33°20′~33°36′N),大陆性季风气候。宝天曼的最高海拔为1 830 m,研究地海拔约1 400 m;成土母质主要是花岗岩,低海拔为山地黄棕壤和褐土,高海拔为山地棕壤。气象数据是由距研究地点200 m以内的气象站采集,年均气温15.1℃,年均降水量900 mm,年蒸发量991.6 mm,极端高温41.2℃,极端低温-14. 8℃,相对湿度68%,年积温4 200~4 900℃。保护区植物种类丰富,共有种子植物150科783属2 147种。植被区划属暖温带落叶阔叶林,锐齿栎是研究地主要优势冠层树种,三桠乌药是伴生的一个亚冠层小乔木种。
-
2013年4月在森林原位建立3块20 m×20 m的降雨减少(截雨)样地,3块20 m×20 m的对照样地。截雨样地通过拦截近地位置的穿透雨实现样地降雨减少,拦截率约为50%。每块截雨样地均由约160块(0.5 m×3 m)透明减水膜组成,薄膜安装在距地面约1.5~2.5 m高的不锈钢架上,装置所截雨水由导水槽导出样地之外。每块样地四周均设有约70 cm深的隔水板,防止样地内外的土壤水分交换。截雨样地距离宝天曼生态站约4 km。
截雨样地共有三桠乌药21棵(样地内树木大部分为锐齿栎),胸径为4.11±0.46 cm,对照样地共有三桠乌药38棵,胸径为4.3±0.33 cm,2个样地三桠乌药的胸径差异不显著,且2个样地的三桠乌药树高一般都不超过5 m。每次测定时每个样地随机采样6棵。
-
降雨数据用临近的一个气象站采集,样地土壤湿度用Em50 Series Data Collection System (Decagon Devices, Inc., USA)进行测定,测定值为土壤体积含水量,每个样地2个探头,随机布设,频率为30 min,测定深度为5 cm。
-
2014年8月2日用压力室(model 1000; PMS Instruments Co., USA)测定截雨样地和对照样地的枝条凌晨水势和中午水势,凌晨水势测定时间为5:30—6:30,中午水势测定时间为12:30—13:00。为防止测定栓塞(导水损失率)时产生人为栓塞和了解三桠乌药木质部导管特性,在进行栓塞测定之前,用注入空气法[14]测定三桠乌药的最大导管长度,约为60 cm。采样时保证测定凌晨栓塞和中午栓塞的枝条长度大于60 cm,约1 m,采样时间与水势测定时间相同,采样后用湿浴巾小心包裹,马上带到宝天曼生态站实验室,用低压液流系统[14]测定导水损失率和枝条导水率。所测枝条的所有叶片都用叶面积仪(Li-3000C; Li-Cor Inc., USA)测定叶面积,枝条末端的边材面积与枝条总叶面积的比值为Huber值,并选取成熟展开叶片计算平均叶面积。冲洗栓塞后的枝条最大导水率与所测茎段的边材面积的比值为边材比导率,最大导水率与茎段以上所有叶片总面积的比值为叶片比导率。测定栓塞的茎段用FAA固定液(乙醇∶乙酸∶甲醛∶水=10∶1∶2∶7)带回实验室,制作半薄切片,用Axio Imager A1光学显微镜(Carl Zeiss Inc., Oberkochen, Germany)观察导管并拍照,当茎段大于2年生,只测定最外面2圈年轮内的导管,截雨样地与对照样地的三桠乌药均选取20个以上的观察视野统计导管直径和导管密度。
为方便三桠乌药栓塞脆弱性曲线测定,均是雨后凌晨在生态站周围取样,采用传统的干燥法[14]进行测定。曲线用如下公式拟合:
$ PLC = \frac{{100}}{{1 + \exp (a(\psi - b))}} $
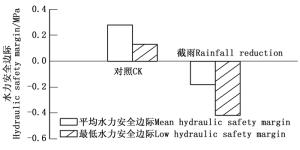
式中:PLC是导水损失率(%),ψ为对应的水势(MPa)。导水损失率50%所对应的水势值为P50,实测的中午水势平均值与P50的差值为平均水力安全边际,而实测到的水势最低值与P50的差值为最低水力安全边际。
-
用LI-6400XT型便携式光合仪(Li-Cor, Lincoln, USA)测定光合生理参数,包括净光合速率(Pn)、蒸腾速率(Tr)、气孔导度(Gs)。设定CO2浓度为400 μmol·mol-1,叶室温度25℃,光合有效辐射(PAR)为1 500 μmol·m-2·s-1,选择晴朗天气测定。2014年8月3日上午9:00—11:00,用高枝剪剪下枝条后马上测定,待数据稳定后再记录。
-
测定凌晨水势时采样,带到实验室并把枝条的韧皮部和木质部分离,105℃杀青30 min,75℃烘干72 h,烘干样品研磨过100目筛。用蒽酮-硫酸法[15]测定叶片、韧皮部、木质部3个器官的可溶性糖和淀粉浓度,可溶性糖和淀粉浓度的和即为总非结构性碳(TNC)浓度。
-
数据用SPSS 17.0软件进行统计分析。各指标均用独立样本t检验分析截雨样地与对照样地的差异显著性,当P<0.05时认为差异显著。文中数据均为平均值±标准误差,且全部为6个重复。采用Sigmaplot 12.5软件作图。
-
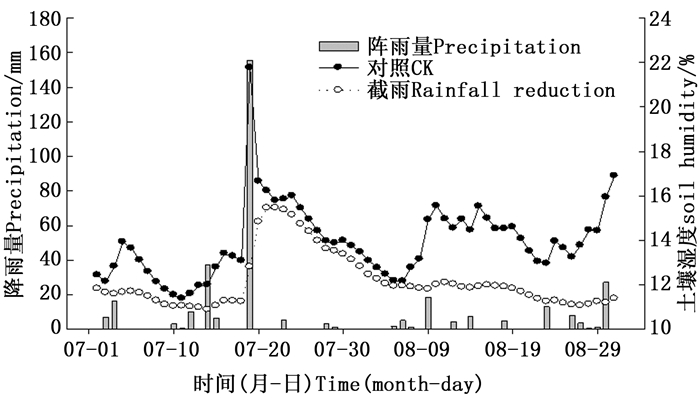
由图 1可知:2014年7月19日有一场强降雨,但在7月20日至8月8日间,降雨量非常小,单日最大降雨量只有5.6 mm,20 d的总降雨量也只有19.6 mm;之后一直到8月末,虽然有几场小降雨,但是截雨样地的土壤湿度一直降低且低于对照样地,而对照样地波动较大,即在远离大的降雨事件后,截雨样地的土壤水分明显低于对照。
-
本试验在2014年8月初进行测定,此时有几天连续的晴朗天气。8月末样地调查时发现,截雨样地的21棵三桠乌药有不同程度的顶梢枯死,而对照样地的38棵三桠乌药只有5棵发生顶梢枯死。
-
从表 1可知:截雨样地三桠乌药的凌晨水势和中午水势显著低于对照,且中午水势平均值为-1.61 MPa,最小值为-1.85 MPa;三桠乌药凌晨的栓塞与对照差异不显著,但中午的栓塞显著高于对照,且均值高达81.3%,实测最大值达88.3%。
表 1 对照样地和截雨样地三桠乌药的凌晨和中午水势、栓塞
Table 1. Pre-dawn and midday water potential, and pre-dawnand midday PLC of Lindera obtusiloba in control plots and rainfall reduced plots
处理Treatment 水势Water potential/MPa 栓塞Embolism/% 凌晨Pre-dawn 中午Midday 凌晨Pre-dawn 中午Midday 对照CK -0.39±0.029a -1.15±0.089a 36.6±5.91a 57.8±1.24b 截雨Rainfall reduction -0.51±0.034b -1.61±0.083b 49.6±5.24a 81.3±2.33a 注:表中同列不同小写字母表示差异显著(P<0.05),下同。
Note: Different lowercases in the same column denote significant differences at p<0.05, the same below. -
由三桠乌药的栓塞脆弱性曲线得到其栓塞50%的水势值(P50)为-1.43 MPa。由图 2可知:截雨样地三桠乌药的平均水力安全边际和最低水力安全边际都低于对照,且都是负值,而对照都是正值。
-
截雨样地三桠乌药的净光合速率(Pn)、气孔导度(Gs)和蒸腾速率(Tr)均显著低于对照(表 2),而截雨样地三桠乌药叶片、韧皮部、木质部的可溶性糖、淀粉和总非结构性碳浓度均与对照差异不显著(图 3)。
表 2 对照样地和截雨样地三桠乌药的气体交换参数
Table 2. The gas exchange parameters of Lindera obtusiloba in control plots and rainfall reduced plots
处理
Treatment净光合速率(Pn)
Net photosynthetic rate/(μmol·m-2·s-1)气孔导度(Gs)
Stomatal conductance/(mol·m-2·s-1)蒸腾速率(Tr)
Transpiration rate/(mmol·m-2·s-1)对照CK 6.73±0.29a 0.187±0.009a 1.22±0.053a 截雨Rainfall reduction 5.03±0.43b 0.118±0.017b 0.72±0.079b -
由表 3可知:截雨样地三桠乌药的单叶叶面积显著低于对照,而Huber值显著大于对照;截雨样地三桠乌药的边材比导率、叶片比导率和导管直径均显著低于对照,而导管密度显著大于对照。
表 3 对照样地和截雨样地三桠乌药的水力结构参数
Table 3. The hydraulic parameters of Lindera obtusiloba in control plots and rainfall reduced plots
处理Treatment 对照CK 截雨Rainfall reduction 单叶叶面积Single leaf size/cm2 34.02±1.32a 29.43±1.09b Huber值Huber value/×10-4(mm2·cm-2) 86.76±3.79b 102.78±6.15a 边材比导率Sapwood specific conductivity/(kg·m-1·s-1·MPa-1) 2.31±0.203a 1.07±0.396b 叶片比导率Leaf specific conductivity/×10-4(kg·m-1·s-1·MPa-1) 1.99±0.18a 1.00±0.37b 导管密度Vessel density/(N·mm-1) 133.96±6.14b 202.83±10.78a 导管直径Vessel diameter/μm 30.59±0.32a 25.77±0.29b -
在截雨处理和对照样地中,三桠乌药叶片、韧皮部、木质部的可溶性糖、淀粉和总非结构性碳的差异均不显著,而截雨样地枝条木质部栓塞严重,中午栓塞最大值达88.3%,水力安全边际为负值,同时从三桠乌药的栓塞脆弱性可看出,三桠乌药的栓塞脆弱性很大,容易产生栓塞,所以水力失衡是三桠乌药顶梢枯死的主要原因。
栓塞因空气进入木质部导管而形成[16],木质部栓塞的脆弱性与导管长度相关,因为长导管具有衔接其他相邻导管更多的面积,因此,可能拥有更多的纹孔,提高了周围栓塞导管中的气泡进入未栓塞导管的可能性,所以,长导管易导致更高的栓塞[17]。三桠乌药的最大导管长度达60 cm,且一般情况下导管直径越大,越易产生栓塞[18]。三桠乌药与样地中另外2个树种四照花(Dendrobenthamia japonica (DC.) Fang var. chinensis (Osborn.) Fang)、水榆花楸(Sorbus alnifolia (Sieb. et Zucc.) K. Koch)相比,最大导管长度更长,导管直径更大,并且P50更高(未发表数据),即三桠乌药的栓塞脆弱性相对更大。当水势低于P50,木质部的水分运输会显著地受到损害,且植物存在加速栓塞的危险,将导致水分输导能力降低、组织死亡和最终整个植株死亡[19]。目前,世界上发现P50最高的树种在热带湿润森林,P50为-0.18 MPa,而最低的树种在澳大利亚荒漠中,P50为-18.8 MPa[17]。本研究中,截雨样地三桠乌药实测的水势最低值比三桠乌药的P50值低,说明干旱时三桠乌药的长距离水分运输易受到严重损害。
在Dickman等[20]的截雨研究中,干旱处理确实导致了树木非结构性碳显著下降,但本研究结果与之不同。在干旱胁迫时,植物生长的降低发生在光合作用降低之前,即植物生长对干旱的敏感性远大于光合。这是因为细胞扩张和分裂由膨压驱动[21],干旱胁迫时膨压会迅速降低,而光合作用的响应更晚一些[8],所以,轻度干旱胁迫容易出现非结构性碳的盈余,而长时间干旱会导致非结构性碳的降低[8]。在本研究中,截雨样地三桠乌药的净光合速率显著降低,即非结构性碳的源减少,而作为非结构性碳的汇(非结构性碳的利用)的生长,如叶面积生长,也降低了,且有20 d未明显降雨,土壤水势可能快速降低,对非结构性碳储备可能还未产生影响,但枝条已极端脱水[12]。总之,本研究中叶片、韧皮部、木质部非结构性碳没有发生显著差异可能是多方面共同作用的结果。
植物通过气孔调节适应环境的水分变化,气孔关闭,水分散失随之减少。本研究中,截雨样地三桠乌药的气孔导度、蒸腾速率和单叶叶面积均显著低于对照,且Huber值显著大于对照。Huber值是枝条末端的边材面积与枝条总叶面积的比值,Huber值大,说明截雨样地三桠乌药枝条的总叶面积小于对照,叶片面积减少可以减轻植物蒸腾的压力,抵御水分亏缺。边材比导率和叶片比导率是没有栓塞时枝条的最大导水率分别与边材面积和枝条总叶面积的比值。截雨样地三桠乌药的边材比导率、叶片比导率和导管直径均显著低于对照,导管密度显著高于对照,说明截雨处理后,三桠乌药新生枝条的木质部导水组织发生了适应性变化。Hagen-Poiseuille方程表明:枝条导水率与导管直径的四次方成正比,与导管数量为累加关系,即导水率主要由导管直径决定,大的导管传输水分效率高[22]。截雨处理后,三桠乌药边材比导率、叶片比导率和导管直径降低,表明三桠乌药在一定程度上由运输水分的高效性向安全性转变。2013年4月建立截雨样地,截雨处理1年多后,三桠乌药气孔导度降低、叶面积变小、枝条导水率及导管直径降低都是适应环境的变化,但2014年8月初截雨样地三桠乌药的水分状态依然很差,水力安全边际为负值,且在8月末部分枝条出现顶端枯死,说明三桠乌药水分传输系统的适应不足以抵消环境变化对它的损害。
-
(1) 水力失衡是降雨减少后三桠乌药顶端枯死的主要原因。
(2) 三桠乌药的水分传输系统在降雨减少后产生了一定的适应性调节,但这些调节不足以使三桠乌药在缺水情况下避免水力失衡而导致的顶端枯死。
宝天曼三桠乌药对降雨减少后的生理生态响应
Ecophysiological Responses of Lindera obtusiloba to Rainfall Reduction in Baotianman Nature Reserve
-
摘要:
目的 在宝天曼森林原位建立降水减少(截雨)样地后,样地内的三桠乌药出现了顶端枯死,本研究从水和碳的角度,探讨三桠乌药顶端枯死的原因。 方法 2013年4月在宝天曼锐齿栎林原位建立了3块截雨样地,在2014年生长季干旱时,采用压力室、低压液流系统和蒽酮硫酸法测定了三桠乌药的水力系统特征和非结构性碳等指标。 结果 显示:(1)三桠乌药在截雨处理后出现了顶端枯死;三桠乌药最大导管长度约为60 cm,栓塞50%时的水势(P50)为-1.43 MPa,其木质部栓塞脆弱性较大。在截雨处理1年后生长季较干旱时,净光合速率、凌晨水势、中午水势显著低于对照,中午栓塞显著高于对照,且水力安全边际为负值。(2)三桠乌药气孔导度、蒸腾速率、叶面积、导管直径、边材比导率、叶片比导率显著下降,Huber值、导管密度显著升高;而叶片、韧皮部、木质部3个器官的可溶性糖、淀粉、总非结构性碳与对照差异不显著。 结论 水力失衡是三桠乌药顶端枯死的主要原因,而蒸腾面积、水分输导系统等的变化表明三桠乌药在缺水环境下进行了一定的适应性调节,但这些调节不足以使三桠乌药在缺水情况下避免水力失衡而导致的顶端枯死。 Abstract:Objective The objective of this study is to investigate the reason why Lindera obtusiloba Bl.Mus.Bot.in Baotianman Mountain showed dieback based on the perspective of water and carbon. Method Three rainfall-intercepted plots were established at the forest in Baotianman Mountain in April 2013. The hydraulic characteristics and nonstructural carbohydrate of L. obtusiloba were measured during the drought period of August, 2014. Result (1) The rainfall reduction caused a lot of shoot dieback of L. obtusiloba. The maximum vessel length of L. obtusiloba was about 60 cm and the water potential causing 50% loss of hydraulic conductivity (P50) was-1.43 MPa, and thus, the xylem vulnerability to cavitation was very large. After being treated with the interception of rainfall for more than one year, in comparison with the control, the net photosynthetic rate, pre-dawn water potential and midday water potential significantly decreased, the percentage loss of hydraulic conductivity significantly increased in the dry period, and terribly its hydraulic safety margin was negative. (2) In addition, the stomatal conductance, transpiration rate, leaf area, vessel diameter, sapwood specific conductivity and leaf specific hydraulic conductivity significantly decreased, the Huber value and the vessel density significantly increased compared with the control. However, there were no significant differences in the soluble sugar, starch and total nonstructural carbohydrate of leaf, phloem and xylem between the rainfall-intercepted trees and the control. Conclusion The results indicate that L. obtusiloba dieback in the rainfall-intercepted plots is attributed to hydraulic failure rather than carbon starvation, and the variation of transpiration area and hydraulic conduction system after treated with rainfall interception indicate that L. obtusiloba has to some degree drought acclimation performances in drought circumstance, but the adaptive adjustment is not enough to prevent L. obtusiloba from dieback. -
表 1 对照样地和截雨样地三桠乌药的凌晨和中午水势、栓塞
Table 1. Pre-dawn and midday water potential, and pre-dawnand midday PLC of Lindera obtusiloba in control plots and rainfall reduced plots
处理Treatment 水势Water potential/MPa 栓塞Embolism/% 凌晨Pre-dawn 中午Midday 凌晨Pre-dawn 中午Midday 对照CK -0.39±0.029a -1.15±0.089a 36.6±5.91a 57.8±1.24b 截雨Rainfall reduction -0.51±0.034b -1.61±0.083b 49.6±5.24a 81.3±2.33a 注:表中同列不同小写字母表示差异显著(P<0.05),下同。
Note: Different lowercases in the same column denote significant differences at p<0.05, the same below.表 2 对照样地和截雨样地三桠乌药的气体交换参数
Table 2. The gas exchange parameters of Lindera obtusiloba in control plots and rainfall reduced plots
处理
Treatment净光合速率(Pn)
Net photosynthetic rate/(μmol·m-2·s-1)气孔导度(Gs)
Stomatal conductance/(mol·m-2·s-1)蒸腾速率(Tr)
Transpiration rate/(mmol·m-2·s-1)对照CK 6.73±0.29a 0.187±0.009a 1.22±0.053a 截雨Rainfall reduction 5.03±0.43b 0.118±0.017b 0.72±0.079b 表 3 对照样地和截雨样地三桠乌药的水力结构参数
Table 3. The hydraulic parameters of Lindera obtusiloba in control plots and rainfall reduced plots
处理Treatment 对照CK 截雨Rainfall reduction 单叶叶面积Single leaf size/cm2 34.02±1.32a 29.43±1.09b Huber值Huber value/×10-4(mm2·cm-2) 86.76±3.79b 102.78±6.15a 边材比导率Sapwood specific conductivity/(kg·m-1·s-1·MPa-1) 2.31±0.203a 1.07±0.396b 叶片比导率Leaf specific conductivity/×10-4(kg·m-1·s-1·MPa-1) 1.99±0.18a 1.00±0.37b 导管密度Vessel density/(N·mm-1) 133.96±6.14b 202.83±10.78a 导管直径Vessel diameter/μm 30.59±0.32a 25.77±0.29b -
[1] IPCC. Climate change 2013: the physical basis. In: Summary for Policymakers. Contribution of Working Group I to the fifth assessment report of the Intergovernmental Panel on Climate Change[M]. Cambridge, UK & New York, NY, USA: Cambridge University Press, 2013. [2] Beier C, Beierkuhnlein C, Wohlgemuth T, et al. Precipitation manipulation experiments-challenges and recommendations for the future[J]. Ecology Letters, 2012, 15: 899-911. doi: 10.1111/j.1461-0248.2012.01793.x [3] 朱雅娟, 吴波, 卢琦. 干旱区对降水变化响应的研究进展[J]. 林业科学研究, 2012, 25(1): 100-106. doi: 10.3969/j.issn.1001-1498.2012.01.017 [4] Allen C D, Macalady A K, Chenchouni H, et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests[J]. Forest Ecology and Management, 2010, 259: 60-684. [5] Breshears D D, Cobb N S, Rich P M, et al. Regional vegetation die-off in response to global-change type drought[J]. Proceedings of the National Academy of Sciences, USA, 2005, 102: 15144-15148. doi: 10.1073/pnas.0505734102 [6] 陈志成, 万贤崇. 虫害叶损失造成的树木非结构性碳减少与树木生长、死亡的关系研究进展[J]. 植物生态学报, 2016, 40(9): 958-968. [7] McDowell N, Pockman W T, Allen C D, et al. Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought?[J]. New phytologist, 2008, 178: 719-739. doi: 10.1111/j.1469-8137.2008.02436.x [8] McDowell N. Mechanisms Linking Drought, Hydraulics, Carbon Metabolism, and Vegetation Mortality[J]. Plant physiology, 2011, 155: 1051-1059. doi: 10.1104/pp.110.170704 [9] Bossel H. Dynamics of forest dieback: systems analysis and simulation[J]. Ecological Modeling, 1986, 34: 259-288. doi: 10.1016/0304-3800(86)90008-6 [10] Mueller-Dumbois D. Natural dieback in forests[J]. Bioscience, 1987, 37: 575-583. doi: 10.2307/1310668 [11] Canny M J. Vessel content during transpiration-embolisms and refilling[J]. American Journal of Botany, 1997, 84: 1223-1230. doi: 10.2307/2446046 [12] Sevanto S, McDowell N G, Dickman L T, et al. How do trees die? A test of the hydraulic failure and carbon starvation hypotheses[J]. Plant, Cell & Environment, 2014, 37: 153-161. [13] O'brien M J, David F R, Burslem P, et al. Contrasting nonstructural carbohydrate dynamics of tropical tree seedlings under water deficit and variability[J]. New Phytologist, 2015, 205: 1083-1094. doi: 10.1111/nph.13134 [14] Wang R Q, Zhang L L, Zhang S X, et al. Water relations of Robinia pseudoacacia L. : do vessels cavitate and refill diurnally or are R-shaped curves invalid in Robinia?[J]. Plant, Cell & Environment, 2014, 37(12): 2667-2678. [15] Hanson J, Moller I. Percolation of starch and soluble carbohydrates from plant tissue for quantitative determination with anthrone[J]. Annals of Biochemistry, 1975, 68: 87-94. doi: 10.1016/0003-2697(75)90682-X [16] Tyree M T, Sperry J S. Vulnerability of xylem to cavitation and embolism[J]. Annual Review of Plant Physiology and Molecular Biology, 1989, 40: 19-38. doi: 10.1146/annurev.pp.40.060189.000315 [17] Markesteijn L, Poorter L. Ecological differentiation in xylem cavitation resistance is associated with stem and leaf structural traitspc[J]. Plant, Cell & Environment, 2011, 34: 137-148. [18] Cai J, Tyree M T. The impact of vessel size on vulnerability curves: data and models for within-species variability in saplings of aspen, Populus tremuloides Michx[J]. Plant, Cell & Environment, 2010, 33: 1059-1069. [19] Choat B, Jansen S, Brodribb T J. Global convergence in the vulnerability of forests to drought[J]. Nature, 2012, 491: 752-756. doi: 10.1038/nature11688 [20] Dickman L T, McDowell N, Sevanto S. Carbohydrate dynamics and mortality in a piñon-juniper woodland under three future precipitation scenarios[J]. Plant, Cell & Environment, 2015, 38, 729-739. [21] Kirkham M B, Gardner W R, Gerloff G C. Regulation of cell division and cell enlargement by turgor pressure[J]. Plant Physiology, 1972, 49: 961-962. doi: 10.1104/pp.49.6.961 [22] Tyree M T, Zimmermann M H. Xylem Structure and the Ascent of Sap, 2nd Edn[M]. Berlin, Germany: Springer Verlag, 2002. -




 下载:
下载: