-
云南切梢小蠹(Tomicus yunnanensis Kirkendall & Faccoli)是一种以危害云南松为特征的针叶树毁灭性害虫[1],该虫自20世纪80年代在云南中部大发生以来,已扩散至四川和贵州等地,累积因云南切梢小蠹危害致死的活立木蓄积量超过20万hm2,严重影响了云南松林的可持续发展和生态环境安全[2-7]。
云南切梢小蠹具有独特的危害特点:新羽化的成虫喜食新鲜嫩梢,一般仅选择健康松树补充营养,致树势衰弱;待性成熟后则只选择衰弱的云南松树干作为繁殖场所,阻碍树木水分和营养物质输送,最终导致树木死亡[2, 5, 7-8]。可见,云南切梢小蠹存在一个明显的蛀梢期和蛀干期,而转移期是相当短暂的,其绝大部分的生活史,如成虫取食、内生殖器发育、交配、产卵,幼虫生长、羽化等过程均在枝梢或树干内完成,这种隐蔽的生活方式为研究其产卵习性带来诸多不便。而另一方面,枝梢内补充营养和树干内繁殖的生活方式,与已知的黄杉大小蠹(Dendroctonus pseudotsugae)[9]、华山松大小蠹(D. armandi)[10]、十二齿小蠹(Ips sexdentatus)[11]、赤材小蠹(Xyleborus ferrugineus)[12]、咖啡果小蠹(Hypothenemus hampei)[13]等在树干或果内补充营养和繁殖的小蠹虫有着本质的区别,加之卵巢发育受环境温度的调控[14-15],意味着枝梢内补充营养的切梢小蠹的卵巢发育更易受到环境温度的作用,进而影响切梢小蠹生殖和种群繁衍。因此,本研究于2014年12月至2016年1月连续对在云南松枝梢内取食的云南切梢小蠹雌成虫卵巢进行解剖观察,并结合当地的环境温度和林间蛀干调查,借此分析云南切梢小蠹卵巢发育程度与温度间的关联性,以便了解云南切梢小蠹的种群数量动态和成灾机制,为后续开展预测预报和综合防治提供理论依据。
-
试验样地设在云南省曲靖市沾益区九龙山林场(103°47′15″E, 25°40′35″N),该地区属低纬度高原季风气候,年降雨量1 000 mm,平均气温16.3~18.6℃。样地内有云南松(Pinus yunnanensis Franch.)、华山松(P. armandii Franch.)、滇朴(Celtis tetrandra Roxb.)、川滇桤木(Alnus ferdinandi-coburgii Schneid.)等人工营造云南松纯林和混交林,自2000年发现有云南切梢小蠹虫(Tomicus yunnanensis)、横坑切梢小蠹(T. minor)、短毛切梢小蠹(T. brevipilosus)、纵坑切梢小蠹(T. piniperda)等危害以来,一直是切梢小蠹持续危害严重的区域。
-
于2014年12月至2016年1月,每月中旬随机选择正在云南松松梢内取食的切梢小蠹带回实验室,剥取成虫,依据鞘翅斜面第二沟间部颗瘤有无、刻点排列、颗瘤毛长短等特征鉴定云南切梢小蠹[1, 16],并依据小蠹虫腹部背板是否可见第8节及其背板形态区分雌雄[17]。鉴定出的雌成虫暂时保存在4℃冰箱内,保存时间不超过3天。
-
将鉴定出的云南切梢小蠹雌成虫固定于蜡盘内,内滴生理盐水,置于蔡氏体视镜下(Zeiss/Stereo Discovery V20)。解剖时,用镊子从腹部后端向胸部前端纵向、缓慢挑开,接着逐步撕去体壁,再用昆虫针挑动卵巢管,切断气管和微气管,将卵巢从腹部拉出,并清除残存的脂肪体和油脂。清洁干净后移至载玻片上,用体视摄影镜观察卵巢发育程度,拍照,分级,并记录每一级别个体数,卵巢管长(不包括端丝长)和宽。以树干内初羽化成虫的卵巢作为对照或称为第Ⅰ级,每月每次解剖20~30头。
-
每次采集云南切梢小蠹时,将重新激活的自供电系统纽扣温度计(Maxim DS1922L/T,±0.5℃)悬挂于树冠处,记录每个采样周期内的环境温度。同时,根据云南切梢小蠹生物学特征[2, 7],蛀干期时在样地设置一个50 m×50 m样方,随机选择30株树势衰弱或有切梢小蠹蛀梢危害的云南松作为观察对象,记录胸径处30 cm范围内是否有云南切梢小蠹蛀干,统计蛀干数量。
-
由于卵巢管发育级别形态变化,Ⅰ级和Ⅱ级卵巢体积用V=πr2L计算,Ⅲ级~Ⅴ级用公式V=2πLW2/3计算[18](L卵巢管长,W为宽,r管柄半径)。采用开源统计分析平台R studio(R Version 3.3.2, https://cran.r-project.org/)进行均值Kruskal-Wallis Test非参检验,Steel-Dwass test进行多重比较,Kendal’s tau-b进行相关性检验。
-
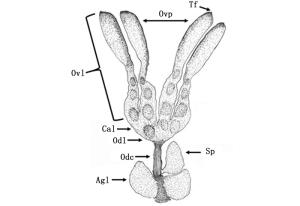
云南切梢小蠹卵巢位于第4腹板消化道下方,由1对卵巢(Ovarioles)、2根短侧输卵管(Lateral oviduc)、1根中输卵管(Common oviduc)、储精囊(Spermatheca)、1对附腺(Accessory gland)和悬带(Terminal filaments)等组成,其中左右卵巢各有2条卵巢管,彼此平行排列,且表面附着大量的气管和微气管,而侧输卵的前端与卵巢小管交界处膨大成卵巢萼(Calyx)(图 1)。
-
根据腹部外部特征,卵巢小管、侧输卵管、中输卵管、附腺、脂肪体形态和颜色,卵黄沉积状况,卵巢萼内卵粒有无、颜色及排列形式等将云南切梢小蠹卵巢发育分为透明期(Ⅰ级)、延伸期(Ⅱ级)、卵黄沉积前期(Ⅲ级)、卵黄沉积期(Ⅳ级)、产卵期(Ⅴ级)共5个等级,各等级的核心特征见表 1。
表 1 云南切梢小蠹卵巢发育分级标准
Table 1. Grading criteria of ovarian development in Tomicus yunnanensis
级别 腹部特征 卵巢 卵巢管 侧输卵管 储精囊 附腺 脂肪体 Ι级 棕黄色、略软 无色透明 纤细管状 略膨大、皱褶不明显 梨形、无色透明 肾形、无色透明 较少 Ⅱ级 渐棕褐色、较软 乳白色 梭形 膨大、皱褶不明显 梨形、乳白色 肾形、乳白色 少量 Ⅲ级 棕褐色、坚硬 乳白色 圆柱形 卵巢萼,具未成熟卵粒 梨形、淡褐色 肾形、淡黄色 较多 Ⅳ级 棕褐色、坚硬、略膨胀 乳白色 圆柱形 卵巢萼、皱褶明显,具成熟卵粒 梨形、淡褐色 肾形、杏黄色 大量 Ⅴ级 棕褐色、坚硬、明显膨胀 亮乳白色 圆柱形 卵巢萼、皱褶明显,大量成熟卵粒 梨形、淡褐色 肾形、杏黄色 大量 透明期(Ⅰ级):从迁飞蛀梢开始,持续15~26天,该时期腹部呈棕黄或渐棕褐色,略软;整个卵巢无色透明,卵巢管纤细,管状,管柄与侧输卵管黏连;侧输卵管略微膨大,包裹于管柄,皱褶不明显;中输卵管无色透明,末端与储精囊和附腺交界处略膨大,储精囊梨形,附腺肾形;卵巢表面布满气管和微气管,背面和腹面未见脂肪附着,但腹腔内已有少量脂肪体存在(图 2 A,A1)。
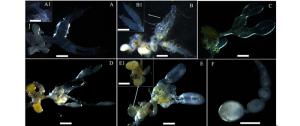
图 2 云南切梢小蠹雌成虫卵巢发育过程的形态变化
Figure 2. Processing-morphology Changes of ovarian development in Tomicus yunnanensis
延伸期(Ⅱ级):卵巢管延伸膨大阶段,持续45~62天,该时期小蠹腹部呈渐棕褐色,较硬;卵巢乳白色,卵巢管延伸明显,梭形,管柄与侧输卵管渐分离;侧输卵膨大,皱褶不明显,中输卵管略显淡黄褐色,短粗;附腺和储精囊增大,附腺乳白色;微气管与卵巢附着力强,剥离难度大,腹腔内脂肪体数量增加明显(图 2B,B1)。
卵黄沉积前期(Ⅲ级):卵巢管膨胀阶段,持续45~60天,该时期小蠹腹部坚硬,卵巢乳白色,卵巢管膨大,圆柱形,管柄分离明显;侧输卵管与卵巢管交界处开始膨大为卵巢萼,但没有成熟卵粒;中输卵管黄褐色,短粗,储精囊淡褐色,附腺淡黄色;卵巢表面附着气管和微气管,可剥离;有少量黄色脂肪附着,腹腔内也出现有大量的黄色脂肪体(图 2C)。
卵黄沉积期(Ⅳ级):该时期持续120~135天,此时切梢小蠹腹部略微膨胀,坚硬;卵巢小管内的卵黄形成乳白色未成熟的卵粒排列于卵巢萼内,侧输卵管和附腺继续膨大且皱褶明显,中输卵管短粗,黄褐色,附腺杏黄色,光滑饱满。卵巢表面附有黄色脂肪,腹腔内黄色脂肪体数量众多(图 2D)。
产卵期(Ⅴ级):持续时间20~45天,腹部明显膨胀,坚硬;卵巢管亮乳白色,端部到末端依次可见到未成熟卵粒和成熟卵粒,呈念珠状排列,而卵巢萼内充满大量的成熟卵粒;储精囊发育成熟,梨形,附腺杏黄色;卵巢萼、附腺、储精囊均附着大量的黄色脂肪,腹腔内黄色脂肪体略少于卵粒形成期(图 2E,E1,F)。
-
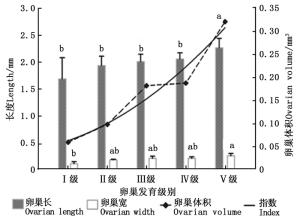
云南切梢小蠹卵巢管长度在Ⅰ~Ⅳ级时增长较为缓慢,Ⅳ~Ⅴ级增长快速(χ2=23.103, P=0.000 12;图 3),如由Ⅳ级的2.05±0.12 mm显著增长到Ⅴ级的2.26±0.17 mm(t=3.120, P=0.016)。卵巢宽度有两个快速增加期(χ2=32.549, P < 0.000 1),一是由Ⅰ级的0.105 ± 0.038 mm显著增加到Ⅲ级的0.179 ± 0.011 mm(t=2.825, P=0.040),二是由Ⅳ级的0.206±0.027 mm显著增加到Ⅴ级的0.256±0.041 mm(t=2.791, P=0.042)。卵巢体积呈幂指数级增长(y=0.05e0.4x, R2=0.948)。
-
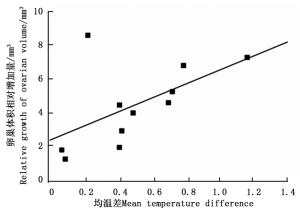
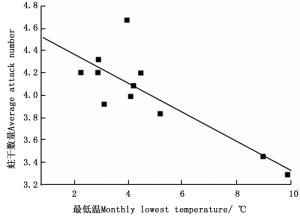
环境温度对云南切梢小蠹卵巢发育有明显的影响(图 4),当环境均温大于10℃时,羽化的云南切梢小蠹从树干进入枝梢开始补充营养,此时卵巢为透明期(Ⅰ级),占比为100%,随后比例逐步降低,而延伸期(Ⅱ级)和卵黄沉积前期(Ⅲ级)开始增加,至10月温度开始降低才逐渐进入卵黄沉积期(Ⅳ级)和产卵期(Ⅴ级)。当环境最低温低于5℃时,即从12月份至翌年1月份,Ⅴ级卵巢比例逐步增加到100%,蛀干产卵开始。相关性分析表明卵巢体积相对增加量与环境均温差呈正相关(r=0.556, P=0.016;图 5),而蛀干产卵数量与月最低温为负相关(r=-0.828, P=0.022;图 6)。
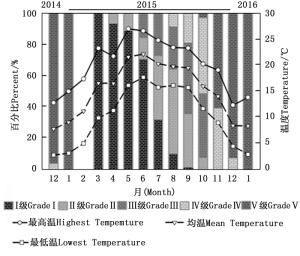
图 4 不同月份云南切梢小蠹不同时间卵巢发育级别与温度的关系
Figure 4. Relationship between ovarian development and environment temperature on the grading ovary of T. yunnanensis in each month
-
卵巢是昆虫非常保守的生殖器官[19]。对于鞘翅目小蠹科昆虫来说,卵巢均有4根卵巢管(左右各2根),但其储精囊和附腺的形态、位置和发育程度等在不同属间存在变化,而同属不同种之间变化并不明显[9-13, 19-21]。例如,咖啡果小蠹附腺为乳白色丝瓜状,储精囊呈管状[13];黄杉大小蠹[9]和华山松大小蠹[10]初羽化时附腺刚形成,受精囊还未出现,待卵巢发育至卵黄沉积期时两者才开始发育,至产卵期时发育成乳白色的球状;而本研究中,云南切梢小蠹即使在蛹期时已形成完整形态的卵巢,只是这时卵巢管、附腺和储精囊更加纤细透明,形态与透明期相似,随着发育附腺呈现杏黄色,储精囊为乳白色(图 2 E,E1)。小蠹虫为卵育型(Synovigenic)昆虫,羽化后必须补充营养物质,根据解剖结构、羽化天数及体内其它部分的特征,卵巢发育常划分为透明期(Ⅰ级)、延伸期(Ⅱ级)、卵黄沉积前期(Ⅲ级)、卵黄沉积期(Ⅵ级)、产卵期(Ⅴ级)5个级别[9-13],一些研究人员认为卵巢发育级别间存在过渡和临界状态[19, 22-23]。但是,小蠹虫很少有临界状态的报道[9-13],推测这可能与小蠹虫解剖难度大,样本数量不足有关。为此,作者通过一年内连续解剖350多只不同发育阶段的云南切梢小蠹,发现存在一些临界状态的卵巢,如延伸期末期卵巢管柄有极少量黄色脂肪沉积(图 2B),这是卵黄沉积前期才有的特征,而卵巢延伸期与卵黄沉积前期的核心差别主要集中在卵巢萼是否膨胀,加之该阶段持续时间短,作者仍将这一阶段的卵巢归入到延伸期,这可能从另一方面解释了为何小蠹虫卵巢分级研究很少有过渡和临界状态的划分。
-
云南切梢小蠹卵巢管长度Ⅰ~Ⅳ级时发育较为缓慢,Ⅴ级时发育快速,而卵巢管宽度Ⅱ级和Ⅴ级发育快,意味云南切梢小蠹卵巢管的发育初期主要是横向的膨胀,Ⅴ级转为纵向的延伸,卵巢管体积呈现幂指数增长。切梢小蠹有干转梢和梢转干两个截然不同生活方式[2],干转梢时虫体体壁相对较柔软,对卵巢的膨胀延伸限制较小,营养物质的补充使其卵巢宽度明显增加(图 3),卵巢面积增加主要为宽度增加所致;待发育至产卵期时,坚硬的体壁限制力增强,利于卵巢长度的增加,而隆起的腹部有卵巢管宽度增加所产生的一部分结果,此时卵巢面积的显著增加是卵巢长与宽的共同作用[17]。云南切梢小蠹雌雄交尾发生在产卵期,雌成虫一生孕卵量60~80粒(每根卵巢管有15~20粒),雄成虫每个精囊内精细胞有200~10 000多个,意味雌成虫一次交尾可满足使所有卵原子受精,交尾也可促进卵巢发育,缩短卵巢发育历期,这与咖啡果小蠹和华山松大小蠹的研究结论是一致的[10, 13]。
-
云南切梢小蠹生活习性与环境温度密切相关。每年3月至11月,沾益地区平均温度14.4℃,温差7.3~13.4℃,温度高波动较小利于云南切梢小蠹营养物质的补充,促进卵巢发育和卵子发生,且随着梢内取食时间的延长,产卵量会随之增加[24-26]。从12月至翌年2月,这一时期温度波动较大,最低温度常常会低于5℃,且伴随霜降、冻雨、降雪等,一旦出现这样的气温骤降,云南切梢小蠹会在很短的时间内由梢内转入干内(图 4)。而作者在不同地区林间蛀干调查也表明:沾益地区的云南切梢小蠹蛀干时间常在12月中下旬,昆明在1月上中旬,玉溪地区出现在1月中下旬至2月初,3个地区的共同特征是温度骤降的时间不同(见中国天气网历史天气)。此外,云南、贵州、四川等地的云南切梢小蠹的蛀梢和蛀干时间是不同的,低温出现的越早,蛀干产卵时间越提前[2, 4, 7-8, 24-25]。另一方面,李丽莎等[2]报道云南切梢小蠹有3月份蛀入树干产卵繁殖的习性,但根据作者对卵巢发育程度和环境温度的分析,此时云南切梢小蠹卵巢处于透明期,卵巢萼未膨大,且没有发育成熟的卵粒,蛀入树干是无法产卵的,推测可能是因当地温度骤降致使云南切梢小蠹躲避低温的一种适应性机制,与繁殖产卵无关。云南切梢小蠹蛀梢补充营养和蛀干产卵均在“隐蔽性”方式下进行,较低的后代数量及亲代抚育使其生态对策更偏向于K对策[27-28],雌成虫作为“先锋个体种”先蛀入树干,释放性信息素,吸引雄虫,雄虫再通过特异性声音识别交配[29-31]。但是,在侵入云南松枝梢和树干过程中,雌虫首先需面对云南松产生的原生性树脂抗性和诱导性抗性,如松树释放的萜烯类和树脂酸类化合物会降低小蠹虫营养物质获取转化和产卵几率[5, 32-33]。同时,蛀梢和蛀干期间,甚至扩散迁飞时,云南切梢小蠹势必受到天敌直接和间接的胁迫,或者出现饥饿,进而影响其生长发育和繁殖[34-35]。因此,云南切梢小蠹在梢内补充营养、迁飞扩散、存活和繁殖投资上必须做出权衡,在温度合适时,必须尽可能多取食,加速卵巢的发育;在气温骤降时,尽快寻找合适的产卵寄主,引诱雄虫交配,完成后代繁衍的任务。当然,繁殖力的实现与昆虫所孕育的卵子数量、出现时间、活力、胚后发育及干扰媒介等关系密切[34, 36-37],但形态解剖对解决这些问题存在明显的局限性,下一步结合荧光染色和共聚焦显微技术将有利于精确判断云南切梢小蠹卵巢发育级别与其繁殖力间所蕴含的深层次关系,可为揭示云南切梢小蠹种群数量动态和成灾机制提供理论依据。
-
(1) 云南切梢小蠹卵巢组成与小蠹科其它种相似,均由卵巢、侧输卵管、中输卵管、储精囊和附腺等组成,但其形态结构、附腺及储精囊的着生位置与不同种存在差异性。
(2) 云南切梢小蠹卵巢发育分为透明期(Ⅰ级)、延伸期(Ⅱ级)、卵黄沉积前期(Ⅲ级)、卵黄沉积期(Ⅳ级)和产卵期(Ⅴ级),其卵巢发育主要集中在Ⅲ、Ⅴ级,其中卵巢管宽度增长主要集中在Ⅱ级和Ⅴ级,长度增长集中在Ⅴ级。
(3) 温度对云南切梢小蠹卵巢发育有明显影响,卵巢体积相对增加量与环境均温差呈正相关,低温会促使发育成熟的云南切梢小蠹蛀干产卵。
云南切梢小蠹雌成虫卵巢发育程度的分级研究
Processing-morphology Change and Grading Criteria for Ovarian Development in Tomicus yunnanensis (Coleoptera, Scolytidae)
-
摘要:
目的 探究卵巢发育与环境温度在云南切梢小蠹生殖和种群繁衍中的作用,进而加深对其种群数量动态和成灾机制的认识。 方法 采用随机抽样法,于2014年12月-2016年1月解剖正在云南松枝梢内取食的云南切梢小蠹雌成虫。 结果 云南切梢小蠹雌成虫卵巢位于第4腹节消化道侧下方,由1对卵巢、2根短侧输卵管、1根中输卵管、受精囊和附腺构成;卵巢包括四根管状的卵巢管,每个卵巢管前端延伸出端丝并集合成悬带,附着于体壁或脂肪体上。根据卵巢发育形态特征的变化,分为透明期(Ⅰ级)、延伸期(Ⅱ级)、卵黄沉积前期(Ⅲ级)、卵黄沉积期(Ⅳ级)、产卵期(Ⅴ级)共5个等级,卵巢发育过程与蛀梢补充营养密切相关,每一级别可持续1~4月不等;Ⅰ-Ⅳ级卵巢管长度增长较慢,Ⅴ级增长快速,而卵巢管宽度Ⅱ级和Ⅴ级增加快,卵巢面积呈幂指数级增长。结合环境温度和林间蛀干调查,发现云南切梢小蠹卵巢发育与温度呈正相关,而蛀干产卵与温度负相关。 结论 云南切梢小蠹雌成虫卵巢发育主要集中在Ⅲ~Ⅴ级,且温度影响该蠹虫的蛀梢和蛀干产卵,研究可为云南切梢小蠹产卵期的预测预报提供理论依据 Abstract:Objective To investigate the role of ovarian development associated with environmental temperature in reproduction and population growth of Yunnan shoot borer Tomicus yunnanensis for further understanding the basic mechanism of its population fluctuation. Method The females of T. yunnanensis were randomly obtained from the shoot of Pinus yunnanensis, and dissected on a paraffin plate. Result The female reproductive system of Yunnan shoot borer is located in the inferior toward the lateral part of alimentary canal at the fourth abdominal segment. It consists of a pair of ovaries and two short lateral oviducts, a common oviduct, a spermatheca and two accessory glands. The ovaries have four tubular ovarioles. The terminal filaments extended from the anterior end of each ovariole can adhere to the internal tegument or fat body. According the morphological changes, the ovarian development was divided into five grades, including transparent period (grade Ⅰ), terminal elongation period (grade Ⅱ), previtellogenic (Ⅲ grade), vitellogenic eggs (grade Ⅵ), and ovipositing period (grade Ⅴ). Each grade of ovarian development can last less than 1 to 4 month, and show a closely relation to the nutrient acquisition. The length of ovariole grows slowly in the previous and middle and rapidly in late grades, but the width of ovariole grow rapidly in the previous and late grades. The whole size of ovary increase rapidly with a power-law dependence. Using Kendall's tau-b correlation analysis, it was found that the correlation between ovarian development and temperature was positive, and a negative relationship was found between oviposition and temperature. Conclusion The ovarian devel-opment of female adults of T. yunnanensis focuses mainly on the middle and late grades. The environment temperature has a significant effect on the life style of shoot and stem, respectively. The results can help to further deepening the theoretical basis for the prediction of oviposition of Yunnan shoot borers. -
Key words:
- Tomicus yunnanensis
- / ovarian morphology
- / ovarian development
- / grading criteria
-
表 1 云南切梢小蠹卵巢发育分级标准
Table 1. Grading criteria of ovarian development in Tomicus yunnanensis
级别 腹部特征 卵巢 卵巢管 侧输卵管 储精囊 附腺 脂肪体 Ι级 棕黄色、略软 无色透明 纤细管状 略膨大、皱褶不明显 梨形、无色透明 肾形、无色透明 较少 Ⅱ级 渐棕褐色、较软 乳白色 梭形 膨大、皱褶不明显 梨形、乳白色 肾形、乳白色 少量 Ⅲ级 棕褐色、坚硬 乳白色 圆柱形 卵巢萼,具未成熟卵粒 梨形、淡褐色 肾形、淡黄色 较多 Ⅳ级 棕褐色、坚硬、略膨胀 乳白色 圆柱形 卵巢萼、皱褶明显,具成熟卵粒 梨形、淡褐色 肾形、杏黄色 大量 Ⅴ级 棕褐色、坚硬、明显膨胀 亮乳白色 圆柱形 卵巢萼、皱褶明显,大量成熟卵粒 梨形、淡褐色 肾形、杏黄色 大量 -
[1] Kirkendall L R, Faccoli M, Ye H. Description of the Yunnan shoot borer, Tomicus yunnanensis Kirkendall & Faccoli sp. n. (Curculionidae, Scolytinae), an unusually aggressive pine shoot beetle from southern China, with a key to the species of Tomicus[J]. Zootaxa, 2008, 1819: 25-39. [2] 李丽莎, 王海林, 柴秀山, 等.云南松纵坑切梢小蠹危害及生物学特性研究[J].云南林业科技, 1997, 79(2): 1-7. [3] 周建华, 肖育贵, 肖银波, 等.四川地区云南松切梢小蠹监测技术研究[J].四川林业科技, 2004, 25(4): 1-6. doi: 10.3969/j.issn.1003-5508.2004.04.001 [4] 朱增明.盘县云南切梢小蠹的发生规律及防治研究[J].现代农村科技, 2016, 6(9): 27-27. doi: 10.3969/j.issn.1674-5329.2016.09.021 [5] Lieutier F, Ye H, Yart A. Shoot damage by Tomicus sp.(Coleoptera: Scolytidae) and effect on Pinus yunnanensis resistance to subsequent reproductive attacks in the stem[J]. Agricultural and Forest Entomology, 2003, 5(3): 227-233. doi: 10.1046/j.1461-9563.2003.00182.x [6] Liu H, Zhang Z, Ye H, et al. Response of Tomicus yunnanensis (Coleoptera: Scolytinae) to infested and uninfested Pinus yunnanensis Bolts [J]. Journal of Economic Entomology, 2010, 103(1): 95-100. doi: 10.1603/EC09080 [7] 叶辉.纵坑梢小蠹对云南松蛀害研究[J].昆虫学报, 1999, 42(4): 394-400. [8] Långström B, Lisha L, Hongpin L, et al. Shoot feeding ecology of Tomicus piniperda and T. minor (Col., Scolytidae) in southern China[J]. Journal of Applied Entomology, 2002, 126(7-8): 333-342. doi: 10.1046/j.1439-0418.2002.00651.x [9] Sahota T, Chapman J, Nijholt W. Ovary development in a scolytid beetle Dendroctonus pseudotsugae (Coleoptera: Scolytidae): Effect of farnesyl methyl ether[J]. The Canadian Entomologist, 1970, 102(11): 1424-1428. doi: 10.4039/Ent1021424-11 [10] 王静静.华山松大小蠹雌性生殖器结构及卵巢发育的研究[D]扬凌: 西北农林科技大学, 2010. [11] Lieutier F. Ovarian and fat body protein concentrations in Ips sexdentatus (Coleoptera: Scolytidae) parasitized by nematodes[J]. Journal of Invertebrate Pathology, 1984, 43(1): 21-31. doi: 10.1016/0022-2011(84)90060-0 [12] Norris D M, Chu H M, Rao K D P. Changes in ovarian ultrastructure and ecdysteroid titer during the aging process of female Xyleborus ferrugineus (Coleoptera: Scolytidae) [J]. Journal of Morphology, 1983, 177(3): 245-254. doi: 10.1002/(ISSN)1097-4687 [13] Rubio G J D, Bustillo P. A E, Vallejo E. L F, et al. Alimentary canal and reproductive tract of Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae, Scolytinae)[J]. Neotropical Entomology, 2008, 37(2): 143-151. doi: 10.1590/S1519-566X2008000200006 [14] 刘永华, 叶恭银, 李鲜花.温度对野蚕黑卵蜂卵巢发育和卵子发生的影响[J].昆虫天敌, 2006, 28(3): 126-131. doi: 10.3969/j.issn.1674-0858.2006.03.006 [15] Phoofolo M W, Obrycki J J, Krafsur E S. Temperature-dependent ovarian development in Coccinella septempunctata (Coleoptera: Coccinellidae)[J]. Annals of the Entomological Society of America, 1995, 88(1): 72-79. doi: 10.1093/aesa/88.1.72 [16] 李霞, 张真, 曹鹏, 等.切梢小蠹属昆虫分类鉴定方法[J].林业科学, 2012, 48(2): 110-116. doi: 10.3969/j.issn.1006-1126.2012.02.007 [17] 王平彦, 张真, 袁素蓉, 等.一种区分三种切梢小蠹性别的新方法[J].中国森林病虫, 2015, 34(6): 17-20. doi: 10.3969/j.issn.1671-0886.2015.06.005 [18] Giron D, Casas J. Mothers reduce egg provisioning with age [J]. Ecology Letters, 2003, 6(4): 273-277. doi: 10.1046/j.1461-0248.2003.00429.x [19] Büning J. The insect ovary: Ultrastructure, previtellogenic growth and evolution [M]. Netherlands Springer, 1994. [20] 殷蕙芬, 黄复生, 李兆麟.中国经济昆虫志.第二十九册.鞘翅目.小蠹科[M].北京:科学出版社, 1984. [21] 李汝铎, 王金其, 苏德明.昆虫卵巢发育与害虫预测预报[M].上海:复旦大学出版社, 1987. [22] 任金龙, 赵莉, 葛婧.意大利蝗Calliptamus italicus (L.)卵巢发育的研究[J].应用昆虫学报, 2014, 51(5): 1280-1288. [23] 欧阳倩, 莫如江, 吴伟坚.瓜实蝇卵巢的发育阶段及分级[J].生物安全学报, 2014, 23(1): 24-29. doi: 10.3969/j.issn.2095-1787.2014.01.005 [24] 叶辉.纵坑切梢小蠹蛀梢期生物学研究[J].昆虫学报, 1996, 39(1): 58-62. [25] 赵涛, 周楠, 李丽莎.纵坑切梢小蠹在云南松上的繁殖特性[J].西北林学院学报, 2003, 18(3): 47-49. doi: 10.3969/j.issn.1001-7461.2003.03.014 [26] Miller W E. Spruce budworm (Lepidoptera: Tortricidae): Role of adult imbibing in reproduction[J]. Environmental Entomology, 1987, 16(6): 1291-1295. doi: 10.1093/ee/16.6.1291 [27] Lissemore F M. Frass clearing by male pine engraver beetles (Ips pini; Scolytidae): Paternal care or paternity assurance[J]. Behavioral Ecology, 1997, 8(3): 318-325. doi: 10.1093/beheco/8.3.318 [28] 南开大学等合编.昆虫学(下) [M].北京: 高等教育出版社, 1980. [29] Rudinsky J A, Michael R R. Sound production in scolytidae: 'Rivalry' behaviour of male Dendroctonus beetles[J]. Journal of Insect Physiology, 1974, 20(7): 1219-1230. doi: 10.1016/0022-1910(74)90228-5 [30] Lindeman A A, Yack J E. What is the password? Female bark beetles (Scolytinae) grant males access to their galleries based on courtship song[J]. Behavioural Processes, 2015, 115: 123-131. doi: 10.1016/j.beproc.2015.03.009 [31] 王鸿斌, 赵丽稳, 罗茜, 等.红脂大小蠹声音信号记录, 分析与发声器官电镜观察[J].林业科学研究, 2012, 25(1): 17-22. doi: 10.3969/j.issn.1001-1498.2012.01.004 [32] Nebeker T E, Hodges J D, Blanche C A, et al. Variation in the constitutive defensive system of loblolly pine in relation to bark beetle attack[J]. Forest Science, 1992, 38(2): 457-466. [33] Lieutier F. Mechanisms of resistance in conifers and bark beetle attack strategies [M]. Nethrlands, Kluwer Academic, 2002. [34] 李姣, 金晨钟, 龙大彬, 等.天敌昆虫对害虫的非直接致死效应[J].应用昆虫学报, 2014, 51(4): 863-870. [35] Elkin C M, Reid M L. Low energy reserves and energy allocation decisions affect reproduction by mountain pine beetles, Dendroctonus ponderosae[J]. Functional Ecology, 2005, 19(1): 102-109. doi: 10.1111/fec.2005.19.issue-1 [36] Srivastava R K, Gurusubramanian G, Krishna S S. Postembryonic development and reproduction in Dysdercus koenigii (F.) (Heteroptera: Pyrrhocoridae)on exposure to Eucalyptus oil volatiles [J]. Biological Agriculture & Horticulture, 1995, 12(1): 81-88. [37] Shuker, D M, Burdfield-Steel E R. Reproductive interference in insects[J]. Ecological Entomology, 2017, 42(S1): 65-75. -




 下载:
下载: