-
母代效应(Maternal effects)在自然生态系统中是普遍存在的[1-2]。亲代的取食和生活经历等因素可影响生物母体对资源的分配从而影响其后代的发育、表型分化、繁殖等生活史特征[3-5],这种由于母体介导所带来的表观遗传信息跨代传递往往提高种群适合度[6-7]。昆虫天敌亦会受到母代效应的影响[4, 8-10],林间寄生性天敌昆虫母代在成虫寿命期内完成首次寄生后可再次搜索寄生从而产生多个姊妹窝子代[11-14]。这些母代调控下的各个姊妹窝子代发育策略及其在林间的活动将影响其种群的定殖和扩散,有关寄生蜂不同姊妹窝子代生长发育情况以及表型如何分化还未见报道。
白蜡吉丁肿腿蜂(Sclerodermus pupariae Yang et Yao)是近年来在白蜡窄吉丁(Agrilus planipennis)幼虫和蛹上发现的一种卵育型抑性外寄生蜂(synovigenic idiobiont ectoprasitoid)[15],该蜂对寄主有较强的搜索和主动攻击能力,现已成为我国天牛类和吉丁甲类幼虫的优势天敌[16-17]。对白蜡吉丁肿腿蜂生态学和生物学特性的研究显示,该蜂主要营两性生殖,雄蜂先羽化,咬破雌蜂茧壳后钻入交尾。未经交配的雌蜂也能孤雌生殖,但后代全部为雄性。雌雄蜂均可多次交配,仅交配一次的雌蜂补充营养后仍继续产出雌性后代[18];多寄生,1头寄主幼虫常常繁育出数十头,有时高达100多头子代蜂[19-20]。此外,武辉等[18]在室内变温条件下观察到该蜂每代能产出3~5个姊妹窝子代,但是母代产生的各姊妹窝子代发育历期和表型分化等生物学参数尚无详细记载。因此,本研究以白蜡吉丁肿腿蜂为对象,在室内恒温条件下母蜂连续依次寄生多头寄主幼虫,观察其在寿命期内所产生的姊妹窝个数,并比较随着姊妹窝子代产出次数的增加,母蜂对寄主幼虫的寄生能力以及各姊妹窝子代的发育历期、翅型分化和性比情况,研究母蜂连续寄生所产各个姊妹窝子代的生物学参数变化,为阐明该蜂在林间定殖和种群延续机制提供理论依据。
-
白蜡吉丁肿腿蜂建群蜂采集于天津市大港区官港森林公园(38°56′N,117°29′E)内绒毛白蜡(Fraxinus velutina)林受害树上的白蜡窄吉丁蛀道内。寄主麻竖毛天牛(Thyestilla gebleri)幼虫购自市售。野外肿腿蜂种群用麻竖毛天牛幼虫作为替代寄主于室内人工气候箱中(宁波海曙赛福PRX-450C)进行传代饲养建立实验种群,饲养条件为30±1℃,光周期14L:10D,相对湿度为65%±5%。室内种群F32中挑选羽化10 d且体型中等的无翅型母蜂为供试肿腿蜂,选取质量230.0±10.0 mg麻竖毛天牛幼虫作为供试寄主。
-
待白蜡吉丁肿腿蜂雌雄蜂交配后,从羽化10 d的寄生蜂种群中随机选取50头,无需补充营养,直接用毛笔单头挑出并置入有1头麻竖毛天牛的指形管(1.0 cm×5.0 cm)内,用脱脂棉塞紧管口,所有供试昆虫均置于30±1℃,14L︰10D,65%±5%RH的人工气候箱培育。当母蜂首次产下的子代开始吐丝结茧时将母蜂移出,不经过交配直接将母蜂单头接入有1头麻竖毛天牛幼虫的指形管内让其寄生。依次类推,在母蜂整个寿命期内观察母蜂姊妹窝子代的产出情况,直到所有母蜂死亡。所有观察均在Zeiss体视显微镜(Zeiss, Stemi 2000-c)下完成。
-
接蜂后,每天定时观察并记录白蜡吉丁肿腿蜂在连续产出姊妹窝子代后在麻竖毛天牛幼虫体上的产卵和羽化情况。以母蜂在寄主幼虫体上产出卵粒记为寄生,以母蜂子代在寄主体上成功羽化出成蜂记为寄生成功。统计母蜂对寄主幼虫的寄生率和寄生成功率,以及各姊妹窝子代中的雌蜂数量。寄生率(bi)和寄生成功率(ri)是衡量寄主适合度的重要指标,计算方法参考田军等[21]。
$ {{b}_{i~}}=\text{ }{{R}_{i}}/{{S}_{i}}\text{, }{{r}_{i}}~={{A}_{i}}/{{S}_{i}} $
式中Ri为被寄生(以麻竖毛天牛幼虫被白蜡吉丁肿腿蜂蛰刺、麻痹、停止发育并在寄主体上产卵为标志)的寄主数量,Ai为白蜡吉丁肿腿蜂在麻竖毛天牛幼虫上寄生并完成生长发育、羽化为成蜂的寄主数目,Si为供试的麻竖毛天牛幼虫总数。
b∈[0, 1], r∈[0, 1], b, r越接近1,则说明寄主越适合被白蜡吉丁肿腿蜂寄生,同时也反映出该蜂对麻竖毛天牛幼虫寄生作用。白蜡吉丁肿腿蜂雌蜂作为卵育型寄生蜂,在首次产生子代后,如果能搜索到新的寄主幼虫,会再次取食寄主补充营养从而繁育出新的姊妹窝子代。母蜂连续寄生所产的后代均为姊妹窝子代,分别记为第1个姊妹窝子代(sister broodⅠ),第2个姊妹窝子代(sister broodⅡ),第3个姊妹窝子代(sister brood Ⅲ),…,依此类推。
-
记录母蜂接入寄主幼虫的时间(a′)、第1粒卵粒的产出时间(a)、卵粒开始孵化时间(b)、寄生蜂幼虫吐丝时间(c)、第1头雄蜂羽化时间(d)以及第1头雌蜂羽化时间(e),将a-a′、b-a、c-b、d-c、e-c、d-a、e-a分别记作产卵前期、卵期、幼虫期、雄蜂茧蛹期、雌蜂茧蛹期、雄蜂发育历期、雌蜂发育历期;统计各姊妹窝子代雌蜂有翅率、雄蜂数量和性比。以有翅雌蜂个体占子代雌蜂总量的百分比作为雌蜂有翅率,以雄性个体占子代数量的百分比作为寄生蜂性比。
-
采用单因素方差分析比较产出不同姊妹窝次数的母蜂的产卵前期、不同姊妹窝子代卵期、幼虫期、茧蛹期、雄蜂发育历期和雌蜂发育历期的差异,用LSD法检验各参数均值之间的差异显著性。采用卡方检验评价不同姊妹窝子代有翅率、性比的差异,组内比较的显著水平经过布氏矫正后为0.008 3。所有数据使用SPSS 20.0软件分析完成。
-
研究发现,白蜡吉丁肿腿蜂在麻竖毛天牛寄主幼虫上可以产生4个姊妹窝子代,产出第2个姊妹窝后母蜂存活个体数开始降低。第3个姊妹窝产出少量雌蜂,产生第4个姊妹窝后的母蜂继续接入寄主幼虫则不能成功寄生。母蜂连续3次的寄生行为均对寄主幼虫有较高的寄生率和寄生成功率(>75%),随着产出姊妹窝次数的增加,母蜂对寄主幼虫的寄生能力和子代雌蜂数量逐渐降低(表 1)。
表 1 连续产出姊妹窝子代的白蜡吉丁肿腿蜂寄生能力和子代雌蜂数量
Table 1. Parasitic ability and number of female progeny of S. pupariae produced successive sister broods
母蜂所产姊妹窝次数
Number of sister brood produced by female重复数
Replicates寄生率
Parasitism rate/%寄生成功
Successful parasitism/%平均雌蜂产出量
Average female progeny1 50 98.0 a 92.0 a 70.54±16.03A 2 50 82.0 a 76.0 a 52.65±14.07A 3 37 83.8 a 81.1 a 21.97±17.31B 4 20 10.0 b 10.0 b 4.00±2.83B 5 1 0 0 0 表中数据为平均值±标准差。同列数据后不同的小写字母表示在0.008 3水平上差异显著(卡方检验);同列数据后不同大写字母表示在0.05水平上差异显著(单因素方差分析,LSD检验)。Data in the table are presented as mean ± SD. Those in the same column followed by different small letters are significantly different at the 0.008 3 level, by Chi-square test. Those in the same column followed by different capital letters are significantly different at the 0.05 level, by one-way ANOVA, LSD test. -
随着白蜡吉丁肿腿蜂产出姊妹窝次数的增多,母蜂产卵前期逐渐延长,当寄生蜂已经繁育出3个姊妹窝子代后,再次寄生新的寄主幼虫时产卵前期显著延长(df = 3, 112, F = 3.816, P = 0.012)。各个虫态的发育历期虽然在各个姊妹窝之间存在差异,但其历期并无逐代延长或者缩短的规律(表 2)。
表 2 连续产出姊妹窝子代的白蜡吉丁肿腿蜂产卵前期和子代发育历期
Table 2. Pre-oviposition duration and progeny developmental duration of S. pupariae produced successive sister brood
母蜂所产姊妹窝次数
Number of sister broodproduced by female产卵前期
Pre-ovipositionperiod卵期
Egg duration幼虫期
Larval duration雄蜂茧蛹期
Male pupalduration雌蜂茧蛹期
Female pupalduration雄蜂发育历期
Developmental duration of male雌蜂发育历期
Developmental duration of female1 5.37±0.57 b 2.65±0.85c 6.89±0.71a 12.53±0.72 a 12.74±0.68a 19.41±1.05 ab 19.63±1.14 b 2 5.45±0.67 b 3.63±0.54a 7.34±2.64a 11.58±1.08 b 11.89±1.01b 18.92±1.94 b 19.24±1.92 b 3 5.60±1.40 b 3.03±0.81b 5.73±1.17b 11.27±1.02 b 11.77±0.77 b 20.03±1.59 a 20.53±1.43 a 4 7.50±0.70 a 3.00±0.00abc 6.00±0.00ab 11.00±0.00 b 11.00±0.00b 20.00±0.00 ab 20.00±0.00ab 表中数据为平均值±标准差,同列数据后不同小写字母表示在0.05水平上差异显著(单因素方差分析,LSD检验)。Data in the table are presented as means followed ± SD, those in the same column, by different small letters are significantly different at the 0.05 level, by one-way ANOVA, LSD test. -
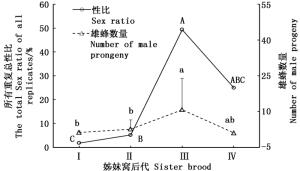
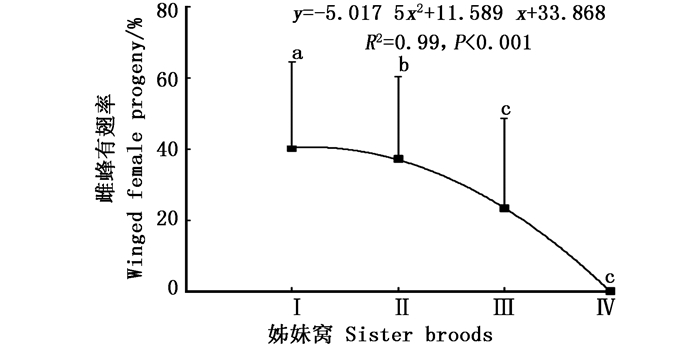
白蜡吉丁肿腿蜂产出的4个姊妹窝子代之间雌蜂有翅率差异显著(df = 3, χ2 = 76.104, P<0.001),在最先产出的两个姊妹窝子代中,寄生蜂种群均保持着一定比例的有翅型和无翅型个体,随着姊妹窝数量的增加,雌蜂有翅率递减;第1个和第2个姊妹窝子代的有翅率在40%左右,第3个姊妹窝子代开始降至23.3%,第4个姊妹窝子代所产少量子代雌蜂均为无翅型(图 1)。
-
白蜡吉丁肿腿蜂各姊妹窝子代雄蜂产出量有显著差异(df=3, 112, F=12.2, P<0.001)(图 2),随着母蜂所产姊妹窝数增加,雄蜂数量逐渐增加,第3个姊妹窝子代产出后有所回落;各姊妹窝子代之间性比也有显著差异(df=3, χ2 =1 091.142, P<0.001),第3个姊妹窝子代性比接近1:1。
-
母代的能量状态会影响其投入生殖的总能量以及对后代个体所投入的能量[22-25],子代生活史能量分配取决于母代繁殖初始时的能量储备[26-27],开始繁殖时母代的低能量储备会对子代个体的生殖投资产生生理制约[28-30]。本研究中,白蜡吉丁肿腿蜂母蜂最多产出了4个姊妹窝子代,其对麻竖毛天牛寄主幼虫的寄生力和母蜂产雌力随着母蜂产出姊妹窝子代次数增加而降低直至为零。这可能是由于母体对寄主的搜索、攻击以及子代产出的过程中大量耗能影响了后代的资源分配[31],母蜂能量损耗可以通过再次取食寄主来达到能量的“收支平衡”,但是随着产出姊妹窝次数增多,营养补给能量已经不能抵消掉母蜂生殖耗能,最终的结果即为寄生蜂虽可成功麻痹寄主,但是已不能成功繁殖子代。
产卵前期是母蜂适合度评估标准之一,可以直观的判定寄生蜂的生理和营养状况[32-33]。白蜡吉丁肿腿蜂的产卵前期随着姊妹窝子代次数的增加而增长,说明经过多轮寄生后母蜂适合度显著降低, 也表明母蜂能量储备因生殖而不断减少。母蜂所产姊妹窝子代之间不同虫态的发育历期以及总发育历期虽然存在显著差异,但并没有规律性。在昆虫母代效应的研究中发现母代年龄对子代发育历期产生一定的影响,如斑腹刺益蝽(Podisus maculiventris)7周大的母代所产子代发育历期长于2周大的母亲所产子代[9];同样,四纹豆象(Callosobruchus maculatus)母代在0.5~7.5 d日龄内,随着日龄的增长,子代发育历期逐渐延长[34]。总之在寄主密度和食物质量一致的前提下,母代年龄均会对子代发育历期和繁殖力产生影响[3],本研究中,各姊妹窝子代发育历期的差异和白蜡吉丁肿腿蜂母蜂生理状况是否有关联,差异来自于母体产出姊妹窝子代导致自身能量的减少还是来自产出不同姊妹窝子代的母蜂日龄的增加,还需要进一步试验验证。
母代状况和经历包括母代翅型、母代个体对寄主的选择行为、性选择、产卵行为等,都会对子代的翅型分化产生重要影响[35-36]。翅型分化是昆虫应对环境变化中在种群飞行扩散和繁殖能力之间权衡的一种生态策略[37],有翅型可以远距离扩散至合适栖境,无翅型主要将能量用于繁殖。白蜡吉丁肿腿蜂母蜂进行多轮寄生产生不同姊妹窝子代的过程中子代翅型明显分化,总体来看种群从两种翅型并存逐渐过渡到全为无翅型个体的寄生蜂种群。从母蜂寿命期内产出的所有姊妹窝子代翅型表现来看,在早期产出的姊妹窝子代中,母蜂能量一定程度投入到子代有翅型的产出来帮助种群扩散至新栖境,从而提高搜索到寄主的成功率,随后才将更多的能量投入到种群的繁殖上,提高其生态适应性。推测这种情况下的翅型分化是寄生蜂母代效应下该蜂对环境异质性的翅型应答机制,体现出该蜂高度的环境适应能力。
随着姊妹窝子代的产出,子代性比和雄蜂数量逐代增加,到第3个姊妹窝子代性比已经接近1:1,且有的雌蜂所产子代全为雄性。说明经过受精的卵已经全部产完,以后产的是未受精的卵。这和武辉的研究结果一致[18]。寄生蜂总输卵管背方着生的受精囊可以贮存精子,且可以分泌营养物质使精子以活跃的状态存活相当长的时间[38],从而使得寄生蜂在早期姊妹窝子代大量雌卵产出后,之后取食寄主仍产出一定数量的雄蜂。理论上,白蜡吉丁肿腿蜂随着产卵次数的增加,姊妹窝性比增大,到某一姊妹窝子代则全部产出雄蜂,在本研究中,该蜂产出的第4个姊妹窝子代性比和雄蜂数量有所回落,仍有少量雌蜂产出,这可能是因为寄生蜂寿命受限因而难于全部产出雄蜂的姊妹窝以及产出第4个姊妹窝子代的母蜂重复数量较少导致的数据偏差。另外,还有一种可能,寄生蜂通常会携带病毒或类病毒颗粒,某些共生性微生物对寄生蜂和寄主的协同进化具有重大影响[39],雄蜂在后期姊妹窝子代中大量产出可能是寄生蜂携带了可以延长其寿命的某种病毒,由于雄性可以与多个雌性交配,更高比例的雄性可以使病毒更广泛地扩散。为了更有利于病毒传播,寄生蜂会选择产出更高比例的雄性从而调控性比[40]。但这点还有待于进一步验证。
值得注意的是,本研究在对该蜂产出的4个姊妹窝子代的观察中发现,子代雌蜂有翅率和性比皆在第3个姊妹窝子代产出时出现较大差异,分别表现为子代蜂种群接近无翅型和偏雌性比的寄生蜂产出性比接近1:1的子代。白蜡吉丁肿腿蜂母蜂的寿命期内最多可以经历4个姊妹窝子代的产出,所以第3个姊妹窝子代的产出很可能是寿命期内调控翅型和性比来达到种群适应度最大化的关键节点。天敌昆虫在林间多次寄生,母代势必会因为搜索、寄生和生殖消耗大量能量,明确母代效应下子代发育策略对林间异质环境的应答机制有助于为阐明寄生蜂在林间种群繁殖成功提供理论基础。今后有必要进一步开展用不同种类的寄主幼虫,在不同或者变温条件下,观察母代寿命期内对寄主幼虫的寄生作用及其自身的繁殖情况,以及母代连续产出的姊妹窝子代生活史特征、表型和性比等变化规律。此外,能量消耗产生的相关化学信号通过母代如何传导给子代,其翅型分化中分子水平上的跨代信号传递途径还有待进一步研究。
-
随着白蜡吉丁肿腿蜂母代雌蜂所产姊妹窝子代次数的增多,其对寄主幼虫的寄生能力逐渐降低;雌性有翅率在各姊妹窝子代间递减,从而完成子代从扩散-定殖型到定殖型的过渡,显示出该蜂在应对林间异质环境的一种适应性进化。白蜡吉丁肿腿蜂第3姊妹窝子代的产出可能是其成虫寿命期内调控翅型和性比达到种群适合度最大化的关键节点。
白蜡吉丁肿腿蜂不同姊妹窝子代翅型分化和性比变化
Wing Dimorphism and Sex Ratio Changes in Progeny of Various Sister Broods in Parasitoid Sclerodermus pupariae (Hymenoptera: Bethylidae)
-
摘要:
目的 探究具有多次产卵习性的寄生蜂不同姊妹窝子代种群参数的变化,从母代效应角度明确寄生蜂在自然栖境中增殖和延续种群的机制。 方法 室内30℃±1℃,光周期14L︰10D,相对湿度65%±5%的条件下测定了白蜡吉丁肿腿蜂在其成虫寿命期内所有姊妹窝子代产出过程中对麻竖毛天牛幼虫的寄生作用,比较各姊妹窝子代中雌蜂数量、发育历期、有翅型雌蜂比例和性比等参数的变化。 结果 白蜡吉丁肿腿蜂雌蜂对寄主幼虫的寄生能力和自身的产雌数量逐渐减低;各姊妹窝子代中雌性有翅率逐次递减,其中第3姊妹窝子代降幅最大,直至为零;各姊妹窝子代的雌雄性比呈抛物线趋势,第3姊妹窝子代雌雄性比接近1:1。 结论 白蜡吉丁肿腿蜂姊妹窝子代间翅的非遗传多型性可能是该蜂在应对林间异质环境的一种适应性进化。白蜡吉丁肿腿蜂第3姊妹窝子代的产出可能是其成虫寿命期内调控翅型和性比达到种群适合度最大化的关键节点。 Abstract:Objective This study aims to explore the changes of population parameters among different sister broods in parasitoids with multiple host attacking habit, so as to explain the mechanisms of expanding and sustaining population from the perspective of maternal effects. Method The parasitism on Thyestilla gebleri larva of Sclerodermus pupariae was studied during the production of successive sister broods in its lifetime. The number of female progeny, developmental duration, percentage of winged female, sex ratio and the other parameters of all the sister broods were recorded in the laboratory under the conditions of 30℃, photoperiod of 14L:0D, and RH 65% ±5%. Result The parasitism ability on host larvae and brood sizes reduced gradually. The percentage of winged females decreased with the number of produced sister broods till to zero, with the most dramatic decrease in the third sister brood. The sex ratio of progeny showed a parabolic trend at various successive sister broods and it was close to 1:1 for the third sister brood. Conclusion Wing polyphenism in various sister broods of S. pupariae may reflect adaptive evolution of this parasitoid coping with heterogeneous environment of forest. The production of the third sister brood might be a key point for regulating wing morph and sex ratio of progeny to maximize the fitness of the population in the lifetime in S. pupariae. -
Key words:
- Sclerodermus pupariae
- / maternal effect
- / sister brood
- / parasitic effect
- / percentage of winged female
- / sex ratio
- / fitness
-
表 1 连续产出姊妹窝子代的白蜡吉丁肿腿蜂寄生能力和子代雌蜂数量
Table 1. Parasitic ability and number of female progeny of S. pupariae produced successive sister broods
母蜂所产姊妹窝次数
Number of sister brood produced by female重复数
Replicates寄生率
Parasitism rate/%寄生成功
Successful parasitism/%平均雌蜂产出量
Average female progeny1 50 98.0 a 92.0 a 70.54±16.03A 2 50 82.0 a 76.0 a 52.65±14.07A 3 37 83.8 a 81.1 a 21.97±17.31B 4 20 10.0 b 10.0 b 4.00±2.83B 5 1 0 0 0 表中数据为平均值±标准差。同列数据后不同的小写字母表示在0.008 3水平上差异显著(卡方检验);同列数据后不同大写字母表示在0.05水平上差异显著(单因素方差分析,LSD检验)。Data in the table are presented as mean ± SD. Those in the same column followed by different small letters are significantly different at the 0.008 3 level, by Chi-square test. Those in the same column followed by different capital letters are significantly different at the 0.05 level, by one-way ANOVA, LSD test. 表 2 连续产出姊妹窝子代的白蜡吉丁肿腿蜂产卵前期和子代发育历期
Table 2. Pre-oviposition duration and progeny developmental duration of S. pupariae produced successive sister brood
母蜂所产姊妹窝次数
Number of sister broodproduced by female产卵前期
Pre-ovipositionperiod卵期
Egg duration幼虫期
Larval duration雄蜂茧蛹期
Male pupalduration雌蜂茧蛹期
Female pupalduration雄蜂发育历期
Developmental duration of male雌蜂发育历期
Developmental duration of female1 5.37±0.57 b 2.65±0.85c 6.89±0.71a 12.53±0.72 a 12.74±0.68a 19.41±1.05 ab 19.63±1.14 b 2 5.45±0.67 b 3.63±0.54a 7.34±2.64a 11.58±1.08 b 11.89±1.01b 18.92±1.94 b 19.24±1.92 b 3 5.60±1.40 b 3.03±0.81b 5.73±1.17b 11.27±1.02 b 11.77±0.77 b 20.03±1.59 a 20.53±1.43 a 4 7.50±0.70 a 3.00±0.00abc 6.00±0.00ab 11.00±0.00 b 11.00±0.00b 20.00±0.00 ab 20.00±0.00ab 表中数据为平均值±标准差,同列数据后不同小写字母表示在0.05水平上差异显著(单因素方差分析,LSD检验)。Data in the table are presented as means followed ± SD, those in the same column, by different small letters are significantly different at the 0.05 level, by one-way ANOVA, LSD test. -
[1] Bernardo J. The particular maternal effect of propagule size, especially egg size:patterns, models, quality of evidence and interpretations[J]. American Zoologist, 1996, 36(2):216-236. doi: 10.1093/icb/36.2.216 [2] Mousseau T A, Fox C W. Maternal Effects as Adaptions[M]. Oxford University Press, New York. 1998. [3] Mousseau T A, Dingle H. Maternal effects in insect life histories[J]. Annual Review of Entomology, 1991, 36(1):511-534. doi: 10.1146/annurev.en.36.010191.002455 [4] Hunter M D. Maternal effects and the population dynamics of insects on plants[J]. Agricultural and Forest Entomology, 2002, 4(1):1-9. [5] Raveh S, Vogt D, Kö lliker M. Maternal programming of offspring in relation to food availability in an insect (Forficula auricularia)[J]. Proceedings of the Royal Society B-Biological Sciences, 2016, 283:20152936. doi: 10.1098/rspb.2015.2936 [6] Asch M V, Julkunen-Tiito R, Visser M E. Maternal effects in an insect herbivore as a mechanism to adapt to host plant phenology[J]. Functional Ecology, 2010, 24(5):1103-1109. doi: 10.1111/j.1365-2435.2010.01734.x [7] Galloway L F. Maternal effects provide phenotypic adaptation to local environmental conditions[J]. New Phytologist, 2005, 166(1):93-100. doi: 10.1111/j.1469-8137.2004.01314.x [8] Legaspi J C, O'Neil R J. Developmental response of nymphs of Podisus maculiventris (Heteropera:Pentatomidae) reared with low numbers of prey[J]. Environmental Entomology, 1994, 23(2):374-380. doi: 10.1093/ee/23.2.374 [9] Mohaghegh J, De Clercq P, Tirry L. Effects of maternal age and egg weight on developmental time and body weight of offspring of Podisus maculiventris (Heteroptera:Pentatomidae)[J]. Annals of the Entomological Society of America, 1998, 91(3):315-322. doi: 10.1093/aesa/91.3.315 [10] Mohaghegh J, De Clercq P, Tirry L. Maternal age and egg weight affect offspring performance in the predatory stink bug Podisus maculiventris[J]. Biocontrol, 1998, 43(2):163-174. doi: 10.1023/A:1009902710191 [11] 雷琼, 李孟楼, 杨忠岐.花绒坚甲的生物学特性研究[J].西北农林科技大学学报:自然科学版, 2003, 31(2):62-66. [12] Sauvard D. Reproductive capacity of Tomicus piniperda L. (Col., Scolytidae):2. Analysis of the various sister broods[J]. Journal of Applied Entomology, 1993, 116:25-38. doi: 10.1111/j.1439-0418.1993.tb01165.x [13] Lentini A, Coinu M, Luciano P. Biological studies on Tomicus destuens (Wollaston) (Cleoptera Curculionidae Scolytinae):Phenology, voltinism and sister broods[J]. Journal of Zoology, 2015, 98:37-47. [14] Davídková M, Dolež al P. Sister broods in the spruce bark beetle, Ips typographus (L.)[J]. Forest Ecology and Management, 2017, 405:13-21. doi: 10.1016/j.foreco.2017.08.040 [15] Yang Z Q, Wang X Y, Yao Y X, et al. A new species of Sclerodermus (Hymenoptera:Bethylidae) parasitizing Agrilus planipennis (Coleoptera:Buprestidae) from China, with a key to Chinese species in the genus[J]. Annals of the Entomological Society of America, 2012, 105(5):619-627. doi: 10.1603/AN12017 [16] 王小艺, 杨忠岐, 唐艳龙, 等.白蜡吉丁肿腿蜂对栗山天牛低龄幼虫的寄生作用[J].昆虫学报, 2010, 53(6):675-682. [17] 杨忠岐, 王小艺, 张翌楠, 等.以生物防治为主的综合控制我国重大林木病虫害研究进展[J].中国生物防治学报, 2018, 34(2):163-183. [18] 武辉, 王小艺, 李孟楼, 等.白蜡吉丁肿腿蜂的生物学和生态学特性及繁殖技术研究[J].昆虫学报, 2008, 51(1):46-54. [19] Gao S K, Wei K, Tang Y L, et al. Effect of parasitoid density on the timing of parasitism and development duration of progeny in Sclerodermus pupariae (Hymenoptera:Bethylidae)[J]. Biological Control, 2016, 97:57-62. doi: 10.1016/j.biocontrol.2016.03.003 [20] Wei K, Tang Y L, Wang X Y, et al. The developmental strategies and related profitability of an idiobiont ectoparasitoid Sclerodermus pupariae vary with host size[J]. Ecological Entomology, 2014, 39:101-108. doi: 10.1111/een.12074 [21] 田军, 王小艺, 杨忠岐, 等.温度对白蜡吉丁柄腹茧蜂发育和繁殖的影响[J].昆虫学报, 2009, 52(11):1223-1228 doi: 10.3321/j.issn:0454-6296.2009.11.007 [22] Pollock D E. Egg production and life-history strategies in some clawed and spiny lobster populations[J]. Bulletin of Marine Science, 1997, 61(1):97-109. [23] Bommarco R. Stage sensitivity to food limitation for a generalist arthropod predator, Pterostichus cupreus (Coleoptera:Carabidae)[J]. Environmental Entomology, 1998, 27(4):863-869. doi: 10.1093/ee/27.4.863 [24] Stelzer C P. Resource limitation and reproductive effort in a planktonic rotifer[J]. Ecology, 2001, 82(9):2521-2533. [25] Koivula M, Koskela E, Mappes T, et al. Cost of reproduction in the wild:manipulation of reproductive effort in the bank vole[J]. Ecology, 2003, 84(2):398-405. doi: 10.1890/0012-9658(2003)084[0398:CORITW]2.0.CO;2 [26] Elkin C K, Reid M L. Low energy reserves and energy allocation decisions affect reproduction by Mountain Pine Beetles, Dendroctonus ponderosae[J]. Functional Ecology, 2005, 19(1):102-109. doi: 10.1111/j.0269-8463.2005.00935.x [27] Brough, C N, Dixon A F D. The effects of starvation on development and reproductive potential of apterous virginoparae of vetech aphid, Megoura viciae[J]. Entomologia Experimentalis et Applicata, 1990, 55(1):41-45. doi: 10.1111/j.1570-7458.1990.tb01346.x [28] Jönsson K I. Capital and income breeding as alternative tactics of resource use in reproduction[J]. Oikos, 1997, 78(1):57-66. doi: 10.2307/3545800 [29] Glazier D S. Trade-offs between reproductive and somatic (storage) investments in animals:a comparative test of the Van Noordwijk and De Jong model[J]. Evolutionary Ecology, 1999, 13:539-555. doi: 10.1023/A:1006793600600 [30] Fox C W, Czesak M E. Evolutionary ecology of progeny size in arthropods[J]. Annual Review of Entomology, 2000, 45(1):341-369. doi: 10.1146/annurev.ento.45.1.341 [31] 吕楠楠, 石棋, 仵均祥, 等.麦长管蚜有翅成蚜母代饥饿对其后代发育和繁殖的影响[J].昆虫学报, 2018, 61(10):1177-1183. [32] Barrette M, Wu G M, Brodeur J, et al. Testing competing measures of profitability for mobile resources[J]. Oecologia, 2009, 158(4):757-764. doi: 10.1007/s00442-008-1175-y [33] 魏可, 王小艺, 杨忠岐.补充营养对白蜡吉丁肿腿蜂寄生效率和发育进程的影响[J].林业科学研究, 2016, 29(3):369-376. doi: 10.3969/j.issn.1001-1498.2016.03.009 [34] Fox C W, Dingle H. Dietary mediation of maternal age effects on offspring performance in a seed beetle (Coleoptera:Bruchidae)[J]. Functional Ecology, 1994, 8:600-606. doi: 10.2307/2389921 [35] Braendle C, Davis G K, Brisson J A, et al. Wing dimorphism in aphids[J]. Heredity, 2006, 97:192-199. doi: 10.1038/sj.hdy.6800863 [36] 张方梅, 李祥瑞, 张云慧, 等.母代效应对麦长管蚜翅型分化的影响[C]. 2014年中国植物保护学会学术年会论文集福建厦门. 442. [37] Zera A J, Denno R F. Physiology and ecology of dispersal polymorphism in insects[J]. Annual Review of Entomology, 1997, 42(1):207-230. doi: 10.1146/annurev.ento.42.1.207 [38] 杨忠岐(译), 膜翅目[M].香港: 香港天泽出版社, 1992, 84-85. [39] 王小艺, 杨忠岐.多寄主型寄生性天敌昆虫的寄主适应性及其影响因素[J].生态学报, 2010, 30(6):1615-1627. [40] Wang F, Fang Q, Wang B B, et al. A novel negative-stranded RNA virus mediates sex ratio in its parasitoid host[J]. Plos Pathogens, 2017, 13(3):e100620 -




 下载:
下载: