-
舞毒蛾(Lymantria dispar)属鳞翅目毒蛾科,是一种杂食性林木叶部害虫[1],可以取食杨树、榆树、松树等500多种植物[2],具有分布广、危害重、幼虫顺风迁徙等特点[3]。重金属是指锰、钴、铜、铅、镍等比重大于4.5 g·cm−3的一类很难被自然降解的重金属元素[4]。重金属广泛存在于自然环境中,采矿、化石燃料燃烧、工业废弃物排放等活动可将重金属带入环境,形成一种具有长期性、不可逆性和隐蔽性的累积污染,对整个生态系统造成潜在的破坏[5]。重金属离子能和生物体内蛋白质及酶等发生作用,使它们失去活性,并对生物造成很大的危害。而且通过食物链的传递作用,重金属可以不断在人体内富集,对人类的健康造成很大威胁[6-7]。
随着我国经济的高速发展,重工业对环境所造成的污染也在不断加剧,同时重金属污染已成为全球严重的生态问题[8]。在电池制造厂附近,大量的重金属锰、钴被排入河水以及周围环境中对农业也造成了巨大的危害[7]。本研究采用加有Mn2+和Co2+的人工饲料饲喂舞毒蛾幼虫,测定了舞毒蛾幼虫营养取食指标、营养成分含量以及总抗氧化力。拟为锰和钴污染对生物的影响研究及重金属污染地区的舞毒蛾防治提供理论依据。
-
舞毒蛾虫卵及饲料购自中国林业科学研究院。总抗氧化力试剂盒购于南京建成生物研究所。六水氯化钴(分析纯),购自天津市巴斯夫化工有限公司;四水氯化锰(分析纯),购自天津市东丽;牛血清白蛋白(生物纯),购自Biotopped Science &Technology Co.Ltd;总抗氧化力试剂盒,购自南京建成生物工程研究所。
-
舞毒蛾虫卵用10%甲醛溶液浸泡1 h消毒,流水冲洗30 min,晾干后置于(25±1)℃、光照周期16 L:8 D、相对湿度75±1%条件下孵化,用人工饲料饲养,每两天更换饲料,清理养虫盒[9]。
-
先进行毒力测定,取浓度为0.2 、0.4、0.8 mmol·g−1Mn2+的四水氯化锰混合饲料和0.42、0.83、1.66 mmol·g−1 Co2+的氯化钴溶液分为6组,每组10只虫,观察舞毒蛾4龄幼虫的死亡率,发现0.40 mmol·g−1 Mn2+的四水氯化锰混合饲料和0.83 mmol·g−1 Co2+的六水氯化钴混合饲料处理的舞毒蛾4龄幼虫84 h时死亡率同为30%。因此取配制好的四水氯化锰溶液和六水氯化钴溶液各40 mL煮沸,然后分别加入干饲料11 g、琼脂0.8 g煮沸后倒入饲料盒中配制成两种混合饲料。选取大小一致的舞毒蛾4龄幼虫,采用两种混合饲料分别进行饲喂处理。以蒸馏水配制饲料作为对照。观察84 h后测定舞毒蛾幼虫的死亡率、取食量、虫粪量、体质量增长量,然后取存活幼虫液氮研磨后,放置于-80℃冰箱保存。每组处理幼虫30头,水平重复为3次。
-
取研磨样品100 mg,加入0.9%的NaCl溶液900 μL,漩涡振荡抽提2 min,4 ℃、2 500 r ·min−1离心10 min,取上清液作为蛋白质、碳水化合物及海藻糖含量测定的样品溶液[10]。蛋白质含量采用Bradford考马斯亮蓝G-250染色法进行测定[11],碳水化合物含量测定参考J.H. Roe[12]的方法进行测定,海藻糖含量参照雷芳等蒽酮法[13]测定,脂质含量参照P. Simek等的方法[14]测定。
-
总抗氧化力采用南京建成生物工程研究所总抗氧化力试剂盒,按说明书进行测定计算,测定时用双蒸水调零,波长520 nm,每个测定管设一个对照管。
-
采用SPSS 21.0软件和Excel软件进行数据统计分析。数据以“平均值±标准误”表示。采用配对样本T检验分析处理组与对照组之间的差异显著性。柱形图绘制均采用Origin Pro 9. 0软件。
-
重金属处理的舞毒蛾幼虫死亡率均为30%(表1)。
表 1 Mn2+和Co2+对舞毒蛾幼虫死亡情况的影响
Table 1. Effects of Mn2+ and Co2+ on the death of gypsy moth larvae
处理 死亡率/% Mn2+ 30.0±0.5 a Co2+ 30.0±0 c CK 0.0±0 c 注:不同小写字母表示不同处理之间的显著性差异(P<0.05)
Note: different lowercase letters mean significant difference between different treatments (P < 0.05) -
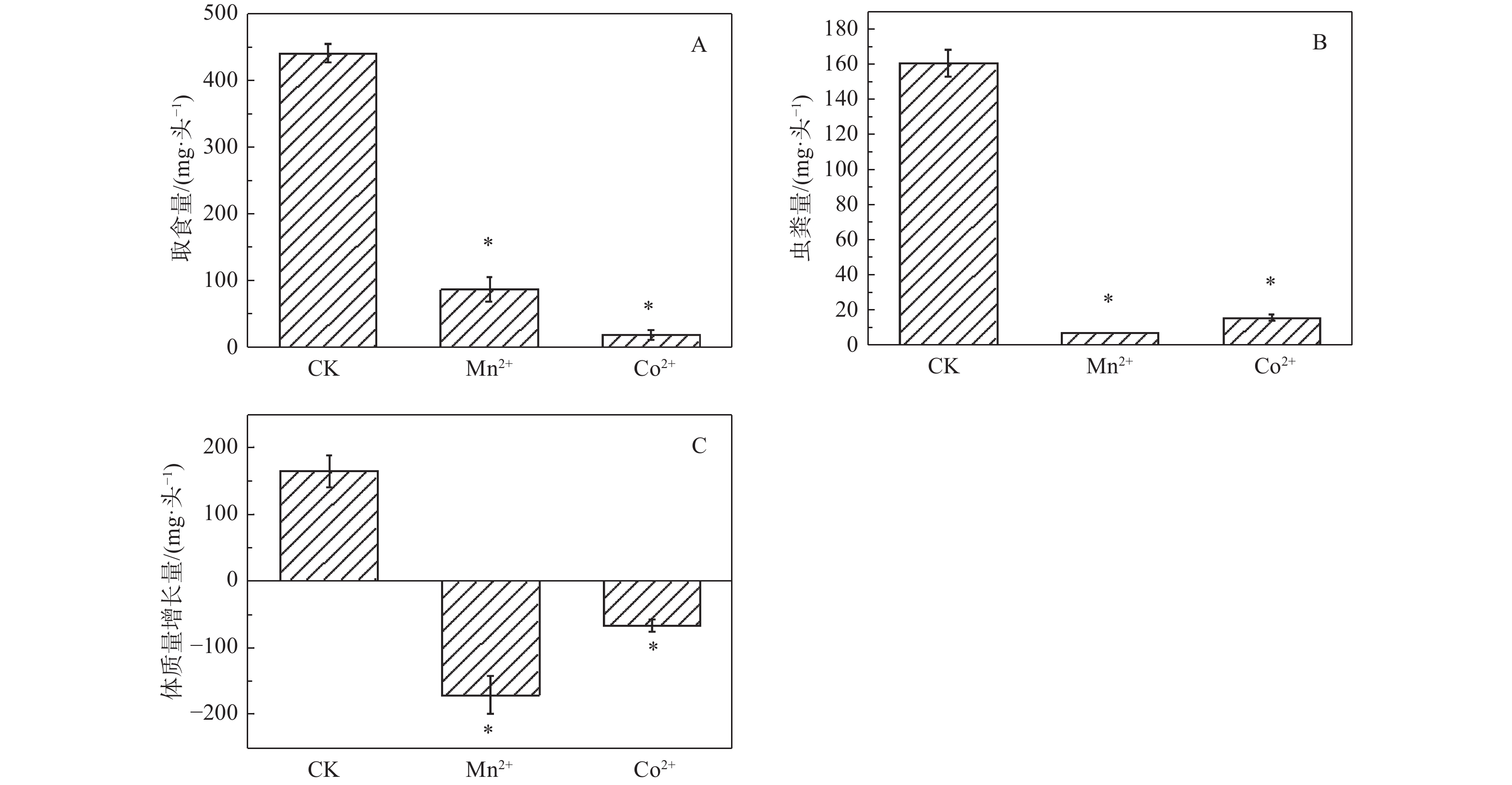
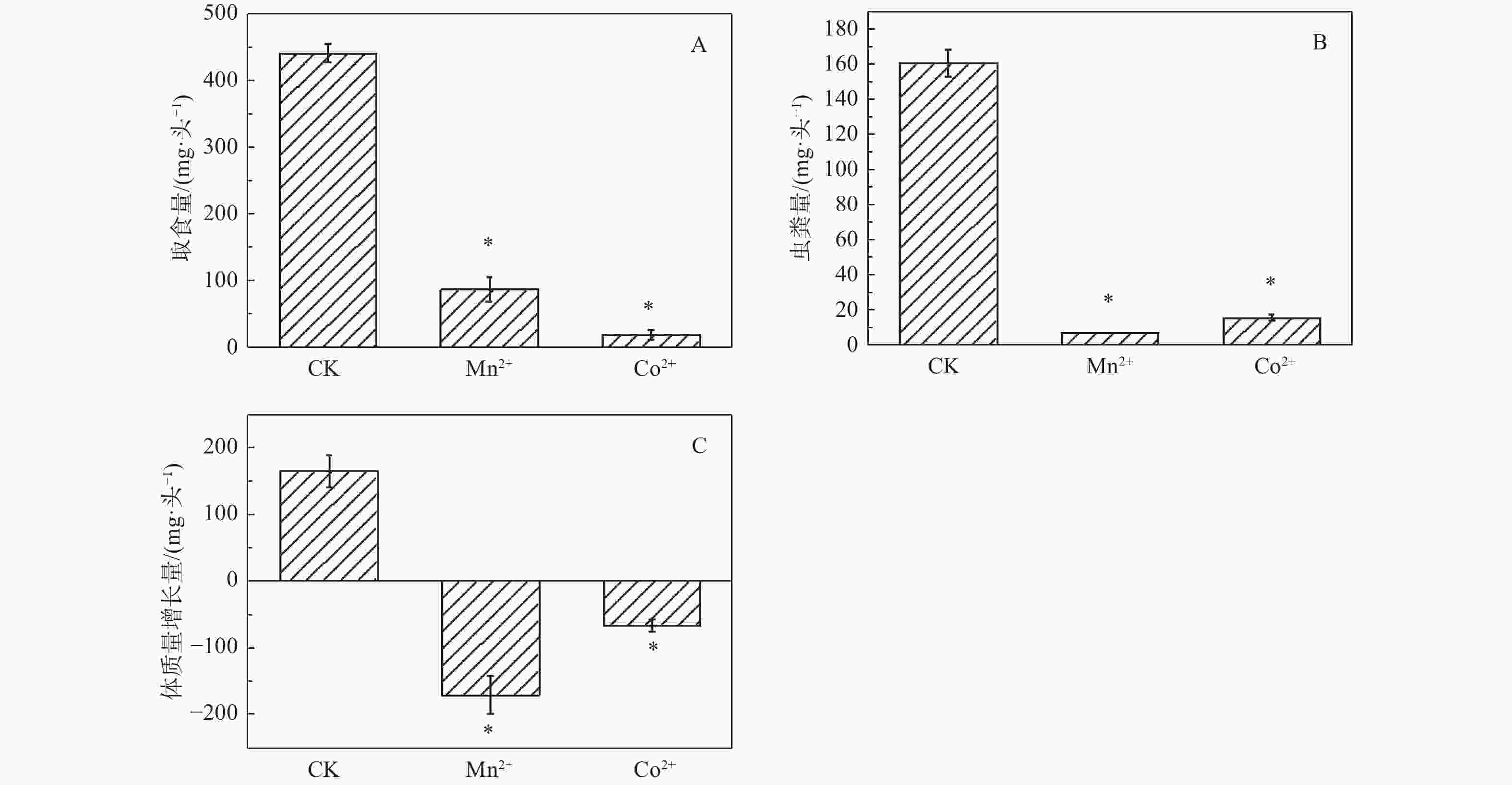
重金属处理的舞毒蛾幼虫取食量、虫粪量、体质量增长量均显著低于对照组。相比对照组,Mn2+处理组取食量降低80.06%,虫粪量降低95.64%,体质量增长量降低203.94%;Co2+处理组取食量降低95.29%,虫粪量降低90.26%,体质量增长量降低140.86%。表明Mn2+和Co2+对舞毒蛾幼虫取食量、虫粪量、体质量增长量均有显著的抑制作用(图1)。
-
重金属处理的舞毒蛾幼虫可溶性蛋白含量、碳水化合物含量、海藻糖含量、脂质含量均显著低于对照组。与对照组相比,Mn2+处理组可溶性蛋白含量降低33.76%,碳水化合物含量降低57.56%,海藻糖含量降低64.05%,脂质含量降低27.77%;Co2+处理组可溶性蛋白含量降低47.46%,碳水化合物含量降低59.45%,海藻糖含量降低59.16%,脂质含量降低33.71%。表明Mn2+和Co2+对舞毒蛾幼虫可溶性蛋白含量、碳水化合物含量、海藻糖含量、脂质含量均有显著的抑制作用(图2)。
-
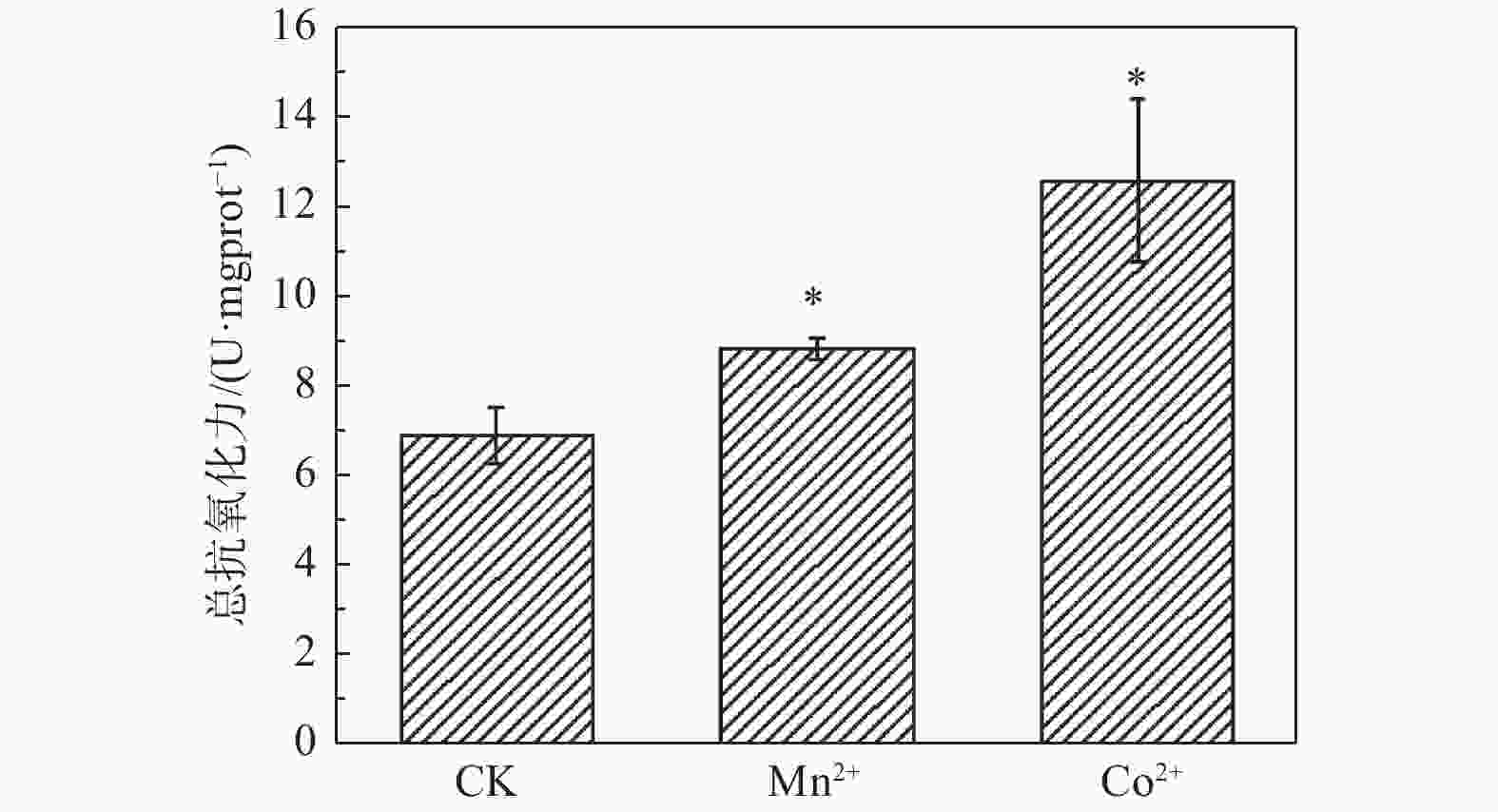
重金属处理的舞毒蛾幼虫总抗氧化力均显著高于对照组。与对照组相比,Mn2+处理组总抗氧化力增加28.25%,Co2+处理组总抗氧化力增加82.54%。表明Mn2+和Co2+对舞毒蛾幼虫总抗氧化力有显著的促进作用(图3)。
-
环境中重金属会对植物的生长发育产生不同程度的影响,可直接影响植物生长,也可通过影响土壤的生理特性进一步影响植物的生长发育[15]。舞毒蛾幼虫作为杂食性昆虫,主要以植物的叶片为食,但由于不同树种叶片营养成分、吸收存储重金属离子能力存在差异[16],同时舞毒蛾幼虫对不同树叶存在取食偏好性等原因[17],采取不同树叶进行研究容易摄入过多变量,导致难以分析食物中重金属的含量对舞毒蛾幼虫的影响。因此本实验采用重金属离子溶液配置的混合饲料饲喂舞毒蛾4龄幼虫进行实验。有研究表明在饲料中添加Cd2+显著延长了舞毒蛾幼虫的发育时间,并且显著降低了其取食量和体质量增长量[18],可能是因为舞毒蛾幼虫的取食偏好性,对含重金属的食物不喜欢所致。本研究发现舞毒蛾幼虫的肠道受到严重的破坏,甚至溶解,说明很大的可能性是由于舞毒蛾幼虫的肠道遭到了重金属的破坏。Malakar C等人的研究也发现在Cd2+的胁迫作用下,短角稻蝗(Dnopherula brevicornis)幼虫的日增重、生长速率和寿命会明显降低[19];与本实验结果相吻合;表明很可能是因为重金属本身对舞毒蛾幼虫具有刺激作用。可溶性蛋白是重要的渗透调节物质和营养物质,其变化可以反映细胞内蛋白质的合成、变性和降解等重要信息[20]。有研究表明,重金属如Pb等可通过与蛋白质的-SH或其它基团的相互作用,改变其结构或功能,进而影响其合成代谢[21],这结果与本实验结果相吻合,这可能是因为重金属胁迫对蛋白质的合成具有一定的抑制作用,抑制了蛋白合成酶的活性。碳水化合物是昆虫重要的能源物质,是维持其生命活动所需能量的主要来源[22],研究表明,当舞毒蛾受到阿维·杀铃脲复合剂胁迫时,体内碳水化合物含量会有所下降[23];与本研究结果一致,说明重金属胁迫可以抑制碳水化合物的转化,使昆虫正常的生命活动受到影响。海藻糖是昆虫体内主要的糖类,脂质是其主要的储能物质,研究表明,用添加了Cd2+的人工饲料饲养舞毒蛾幼虫后,其组织和淋巴中的储能物质海藻糖、糖原和脂质含量显著下降[24];与本实验结果相一致,因此我们可以推测出舞毒蛾幼虫为了缓解重金属胁迫带来的不良影响,可能会激发海藻糖酶和脂肪酶的活性,促使海藻糖分解为葡萄糖,脂质转化为脂肪酸和甘油,为维持其正常的生长发育提供更多能量。总抗氧化力是用于评价机体抗氧化系统功能状况的综合性指标。它的大小可以从总体上反映机体防御体系抗氧化能力的高低[25]。研究表明,高浓度的Cd2+和Po2+胁迫会导致果蝇体内的总抗氧化力T-AOC,以及SOD和CAT的活性表现为抑制现象,且与胁迫的重金属浓度相关[26]。本研究中发现舞毒蛾幼虫在重金属Mn2+和Co2+的胁迫作用下总抗氧化力显著提高。其可能的原因是试验浓度下Mn2+和Co2+毒力并未超过舞毒蛾4龄幼虫抗氧化系统的承受能力,促使舞毒蛾幼虫分泌各种抗氧化物质和抗氧化酶,从而提高它总抗氧力,以维持其正常生长发育。而高浓度的Cd2+和Po2+会导致果蝇的抗氧化系统破坏。说明昆虫抗氧化系统抵御氧化胁迫的能力是有限的,低剂量的重金属可以诱导虫体抗氧化基因的表达,高剂量重金属的持续胁迫则会造成抗氧化系统的破坏[27]。
-
重金属Mn2+和Co2+对舞毒蛾幼虫体质量、营养取食量、排便量、可溶性蛋白含量、碳水化合物含量、海藻糖含量、脂质含量均有显著的抑制作用,但对总抗氧化力有显著的促进作用。说明舞毒蛾幼虫主要通过提高自身抗氧化力的来应对重金属的胁迫,同时也提高自身对重金属的抗性。
Mn2+和Co2+胁迫对舞毒蛾幼虫营养和抗氧化力的影响
Effects of Manganese Ion and Cobalt Ion Stress on Nutrition and Antioxidant Capacity of Gypsy Moth Larvae
-
摘要:
目的 为了明确舞毒蛾幼虫对Mn2+和Co2+胁迫的响应机制。 方法 采用0.40 mmol·g−1 Mn2+和0.83 mmol·g−1 Co2+的两种混合饲料分别喂食舞毒蛾4龄幼虫,测定了Mn2+和Co2+(LC30)对舞毒蛾4龄幼虫体质量、营养取食情况、营养成分含量及总抗氧化力。 结果 与对照组相比较,其取食量、虫粪量、体质量增长量均显著降低;Mn2+处理组可溶性蛋白含量降低33.76%,碳水化合物含量降低57.56%,海藻糖含量降低64.05%,脂质含量降低27.77%;Co2+处理组可溶性蛋白含量降低47.46%,碳水化合物含量降低59.45%,海藻糖含量降低59.16%,脂质含量降低33.71%;Mn2+处理组总抗氧化力增加28.25%,Co2+处理组总抗氧化力增加82.54%。表明Mn2+和Co2+对舞毒蛾幼虫取食量、虫粪量、体质量增长量、可溶性蛋白含量、碳水化合物含量、海藻糖含量、脂质含量均有显著的抑制作用,对其总抗氧化力有显著的促进作用。 结论 舞毒蛾幼虫是主要通过提高自身抗氧化力来应对重金属的胁迫,从而提高自身对重金属的抗性。 Abstract:Objective To determine the response mechanism of gypsy moth (Lymantria dispar) larvae to Mn2+ and Co2+ stress. Method The effects of 0.40 mmol Mn2+·g−1 and 0.83 mmol Co2+·g−1 on body weight, nutrient intake, nutrient composition and total antioxidant capacity of the fourth instar larvae of gypsy moth were measured. Result Compared with the control group, the amount of food intake, feces and weight gain were significantly reduced. In the Mn2+ treatment group, the soluble protein content reduced by 33.76%, carbohydrate content by 57.56%, trehalose content by 64.05% and lipid content by 27.77%. In Co2+ treatment group, the soluble protein content reduced by 47.46%, carbohydrate content by 59.45%, trehalose content by 59.16% and lipid content by 33.71%. The total antioxidant capacity of the Mn2+ treatment group increased by 28.25%, and that of the Co2+ treatment group increased by 82.54%. These results showed that Mn2+ and Co2+ have significant inhibitory effects on the food intake, fecal content, weight growth, soluble protein content, carbohydrate content, trehalose content and lipid content of the larvae, and significantly promoted their total antioxidant capacity. Conclusion The larvae of gypsy moth respond to the stress of heavy metals mainly by improving the activity of their own antioxidant capacity, reducing sensitivity and improving resistance to heavy metals. -
Key words:
- Lymantria dispar
- / larvae
- / manganese ion
- / cobalt ions
- / nutrition feeding
- / nutritional composition
- / total oxidation resistance
-
表 1 Mn2+和Co2+对舞毒蛾幼虫死亡情况的影响
Table 1. Effects of Mn2+ and Co2+ on the death of gypsy moth larvae
处理 死亡率/% Mn2+ 30.0±0.5 a Co2+ 30.0±0 c CK 0.0±0 c 注:不同小写字母表示不同处理之间的显著性差异(P<0.05)
Note: different lowercase letters mean significant difference between different treatments (P < 0.05) -
[1] Enayati AA, Ranson H, Hemingway J. Insect glutathione transferases and insecticide resistance[J]. Insect Molecular Biology, 2005, 14(1): 3-8. doi: 10.1111/j.1365-2583.2004.00529.x [2] Jiang D, Yan S. Effects of Cd, Zn or Pb stress in Populus albaberolinensis on the development and reproduction of Lymantriadispar[J]. Ecotoxicology, 2017, 26(10): 1305-1313. doi: 10.1007/s10646-017-1855-7 [3] 矫丽曼, 杨志岩, 冯连荣, 等. 舞毒蛾生防菌Entomophagamaimaiga研究进展[J]. 江苏农业学报, 2018, 34(5):1183-1190. doi: 10.3969/j.issn.1000-4440.2018.05.030 [4] 王菊英. 海洋沉积物的环境质量评价研究[D]. 青岛, 中国海洋大学, 2004. [5] 贾 丽, 刘建华, 张 璐, 等. 吉林省红旗岭和漂河川镍矿区耕地土壤重金属污染特征[J]. 吉林农业大学学报, 2015, 37(3):338-345. [6] Cui B, Zhang Q, Zhang K, et al. Analyzing trophic transfer of heavy metals for food webs in the newly-formed wetlands of the Yellow River Delta, China[J]. Environmental Pollution, 2011, 159(5): 1297-1306. doi: 10.1016/j.envpol.2011.01.024 [7] Yi Y, Yang Z, Zhang S. Ecological risk assessment of heavy metals in sediment and human health risk assessment of heavy metals in fishes in the middle and lower reaches of the Yangtze River basin[J]. Environmental Pollution, 2011, 159(10): 2575-2585. doi: 10.1016/j.envpol.2011.06.011 [8] 俞华齐, 王 娟, 姜忍忍, 等. 重金属铬单克隆抗体特异性ELISA免疫检测方法的建立[J]. 江苏农业学报, 2016, 32(2):460-466. doi: 10.3969/j.issn.1000-4440.2016.02.034 [9] 梁洪柱, 陈 倩, 田会鹏, 等. 舞毒蛾核型多角体病毒室内增殖的研究[J]. 林业科学研究, 2013, 26(5):593-597. [10] 曾健勇, 张方明, 吴 玥, 等. 阿维菌素与杀铃脲对舞毒蛾幼虫的联合作用机制[J]. 林业科学, 2018, 54(12):110-115. doi: 10.11707/j.1001-7488.20181212 [11] 张雄帅, 周国娜, 高宝嘉. 油松毛虫体内酶系对油松诱导抗性的响应机制[J]. 林业科学, 2014, 50(10):181-187. [12] Roe J H. The determination of sugar in blood and spinal fluid with an throne reagent[J]. J Biol Chem, 1955, 212(1): 335. [13] 雷 芳, 张桂芬, 万方浩, 等. 寄主转换对B型烟粉虱和温室粉虱海藻糖含量和海藻糖酶活性的影响[J]. 中国农业科学, 2006, 39(7):1387-1394. doi: 10.3321/j.issn:0578-1752.2006.07.013 [14] SIMEK P, SULA J, KOSTAL V. Physiology of drought tolerance andcold hardiness of the mediterranean tiger moth Cymbalophorapudica during summer diapause[J]. Journal of Insect Physiology, 1998, 44(2): 165-173. doi: 10.1016/S0022-1910(97)00047-4 [15] 郑志林, 罗有发, 周佳佳, 等. 铅锌废渣堆场4种先锋修复植物根际微域磷素赋存形态特征[J]. 水土保持研究, 2019, 26(03):269-278. [16] 徐婷婷. 一株耐重金属镉、铅、锰菌株的筛选及其复合型吸附菌剂的制备[D]. 桂林, 广西师范大学, 2018. [17] 吴明峰, 高 尚, 杨 耀, 等. 棉铃虫和烟青虫初孵幼虫对植物顶尖嫩叶的偏好性差异[J]. 植物保护, 2016, 42(3):63-69. doi: 10.3969/j.issn.0529-1542.2016.03.010 [18] Matić D, Vlahović M, Kolarević S, et al. Genotoxic effects of cadmium and influence on fitness components of Lymantriadispar caterpillars[J]. Environmental Pollution, 2016, 218: 1270-1277. doi: 10.1016/j.envpol.2016.08.085 [19] Malakar C, Ganguly A, Sarkar A, et al. Effects of mercury on development of Oxyafuscovittata (Marschall) (Orthoptera: Acrididae)[J]. Journal of Orthoptera Research, 2009, 18(2): 159-164. doi: 10.1665/034.018.0204 [20] Berntssen M H G, Lundebye A K. Energetics in Atlantic salmon (Salmosalar L.) parr fed elevated dietary cadmium[J]. Camp Biochem Physiol, 2001, 128(3): 311-323. [21] 杨丽娜. 舞毒蛾对环境因子及Pb胁迫的响应机制[D]. 哈尔滨, 东北林业大学, 2018. [22] 陈 龙, 周晓榕, 高利军, 等. 沙葱萤叶甲成虫越夏期间糖类、蛋白及脂肪含量的变化[J]. 昆虫学报, 2018, 61(7):808-814. [23] 张方明, 曾健勇, 吴 玥, 等. 舞毒蛾幼虫对阿维·杀铃脲复合剂胁迫的响应机制[J]. 黑龙江畜牧兽医, 2019(1):106-110. [24] Bischof C. Effects of heavy metal stress on carbohydrate and lipidconcentrations in the haemolymph and total body tissue of parasitized Lymantria dispar.L larvae(Lepidoptera)[J]. Comp Biochem Physiol, 1995, 112(1): 87-92. [25] 周桂莲, 杨慧霞. 小麦抗旱型鉴定的生理生化指标及其分析评[J]. 干旱地区农业研究, 1996, 14(2):65-70. doi: 10.3321/j.issn:1000-7601.1996.02.013 [26] 李长春, 王 永, 姚国新, 等. 铅、镉胁迫对果蝇生长发育和抗氧化能力的影响[J]. 西南农业学报, 2019, 32(2):429-433. [27] Augustyniak M, Migula P, Mesjasz-Przyby1owicz J, et al. Short-term effects of dimethoate on metabolic responses in Chrysolinapardalina (Chrysomelidae) feeding on Berkheya coddii(Asteraceae), a hyper-accumulator of nickel[J]. Environmental Pollution, 2007, 150(2): 0-224. -




 下载:
下载: