-
焦枯病是近年来树木或植物常见的一类叶部病害。其病因复杂,多由不适环境因素引起,如营养元素过量和钾元素缺乏、干旱胁迫、高温日晒等,造成植物水分生理失调,导致叶组织大面积产生枯斑,引发叶焦枯[1–2]。随着气候变暖和环境变化,病原菌导致植物叶焦枯病的种类增多,如由侵染性病原菌真菌拟茎点霉Phomopsis mollissim与其共生菌板栗蛇孢日规壳Ophiognomonia castaneae引起的板栗褐缘叶枯病[3–4];通过昆虫媒介传播的植物病原细菌木质部难养菌Xylella fastidiosa导致的叶焦枯病也日益频发。该细菌病原菌营养需求复杂、寄主范围很广,在多种重要的林木、观赏性植物及农业经济作物形成严重危害,已经成为全球性日益关注的新发流行病害[5–6]。
中国板栗(Castanea mollissima Bl.)属于壳斗科栗属的植物,原产于中国,有“干果之王”之称[7–8],河北板栗主要产于河北省北部的燕山山区,以颗粒饱满、香甜、皮薄、适于糖炒等特点著称于世,久负盛誉,是河北省出口农副产品中具有较大优势的土特产品之一,其产量、出口量和质量,均居全国第一位[8–9]。并且,板栗栽种面积占到全省有林地的10%,成为河北省重要的经济和生态树种[10]。近些年,青龙、迁西等板栗主产地相继发生一种新的叶部病害,板栗发病后,叶片上产生黄褐色的斑点,多个黄褐斑可渗透连片,以致使整个叶片上呈不规则的大面积焦枯状,引起果实重量减少,产量下降,造成栗农经济损失,该病害发病原因亟待明确,尽早进行有效的防控。
-
标本于2019年7月至9月,采于河北青龙县和迁西县(40.40° N, 118.95° E)5个病害发生严重的板栗果园,果园相距5~70 km。在栗园,观察症状的同时,放大镜镜检病症;在实验室,解剖镜镜检病组织表面病症,及显微镜镜检病组织真菌分生孢子器和子实体或细菌雾;实验室新鲜离体病叶组织湿培25℃,每天观察病症至第7天。
-
病原菌的分离和纯化采用组织分离法[11],将新鲜的病叶用清水冲洗干净,用75% 乙醇表面消毒,从其病健交界处用灭菌手术刀切取3~5 mm, 置于PDA培养基、MEA培养基和细菌蛋白胨培养基上25℃恒温培养,待菌丝或菌落长出后,进行转接纯化,并在PDA培养基上于25℃恒温培养,菌丝长满后,置于4℃冰箱中保存备用。每户叶片组织接种12个平板,每个PDA平板接3块组织。
-
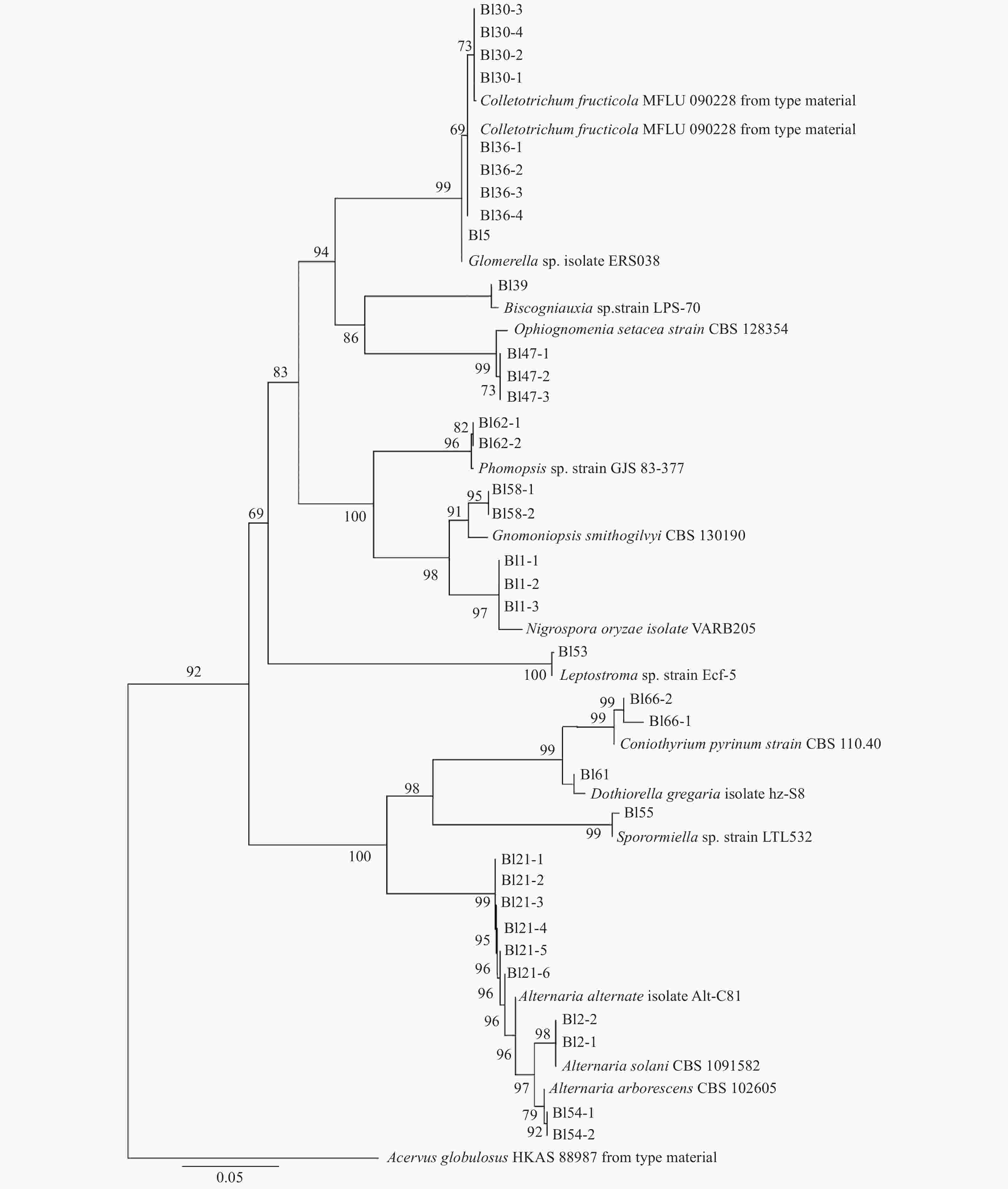
分离菌株按形态初步归类后,用CTAB法提取和纯化菌株的基因组DNA[12],用通用引物ITS1和ITS4进行ITS rDNA基因片段的扩增,PCR产物经回收纯化后,由北京六合华大基因有限公司进行测序。所得序列经NCBI进行碱基Blast同源性分析,确定菌株分类地位。利用Mega X构建系统发育树,建树方法最大简约法(Maximum parsimony,MP),可靠性检验bootstrap法,初始值1000次,外群为Acervus globulosus HKAS 88987,系统发育树中本研究测得的序列号为MT373425-MT373459,Genbank中下载用于比对分析做系统发育树的序列见表1。
表 1 Genbank中下载用于比对分析做系统发育树的参考序列
Table 1. Sequences from Genbank for alignment and phylogenetic tree
序列来源
Source菌株号
Strain No.ITS序列号
ITS No.Alternaria alternata isolate Alt-C81 MN044802 Colletotrichum gloeosporioides IMI 356878 from type material NR_160754 Colletotrichum fructicola MFLU 090228 from type material NR_144783 Nigrospora oryzae isolate VARB205 KT966519 Ophiognomonia setacea CBS 128354 MH864895 Alternaria solani CBS 109158 KJ718239 Alternaria arborescens CBS 102605 NR_135927 Phomopsis sp. GJS 83-377 AF102999 Gnomoniopsis smithogilvyi CBS 130190 NR_166040 Coniothyrium pyrinum CBS 110.40 MH856053 Biscogniauxia sp. strain LPS-70 MF379340 Glomerella sp. isolate ERS038 KY413720 Leptostroma sp. strain Ecf-5 MK211262 Sporormiella sp. strain LTL532 MF663586 Dothiorella gregaria isolate hz-S8 FJ517548 Acervus globulosus HKAS 88987 NR_159565 -
依据1.3的结果,结合已报道致病性的菌株进行接种试验。室内离体接种选取健康无病的新鲜叶片,叶片采自温室1年健康苗的成熟叶片(停止伸展发育后20~30天),用清水冲洗干净,75% 乙醇消毒5 min,再用无菌水冲洗多次,置于事先灭菌的培养皿(直径150 mm,底部垫2~3层无菌滤纸,并加入适量无菌水保湿)中。因为发现在培养观察期间,分离菌株培养有产孢和不产孢,因此,接种体前者用分生孢子,后者用菌丝(菌饼)。将PDA平板上培养5~7天的分离物,无菌水冲洗孢子,制成106孢子·mL−1接种液;或用直径5 mm的打孔器在菌落边缘取菌饼。预实验发现无伤接种不成功,因此,用刺伤进行接种(用消毒昆虫针在健康叶片表面针刺后,将菌饼贴接于刺伤部位)。每处理接种5个叶片,每片叶接3个点,另以叶片相同部位接种无菌水处理为对照。接种封好后置于25℃培养箱黑暗培养。接种48 h后移去菌丝块,隔天观察发病情况并记录两周。
根据离体叶片接种结果,选取病原菌进行1年生盆栽苗接种实验,将菌丝块接种盆栽苗板栗叶片上,每种致病菌株接种3盆苗木,每苗木处理5个叶片,并用无菌水同时接种等数量的盆栽板栗苗作为对照,保鲜袋保湿24~48 h后去掉,定期观察发病情况。发病后取具有症状的组织再次进行病原菌分离。
-
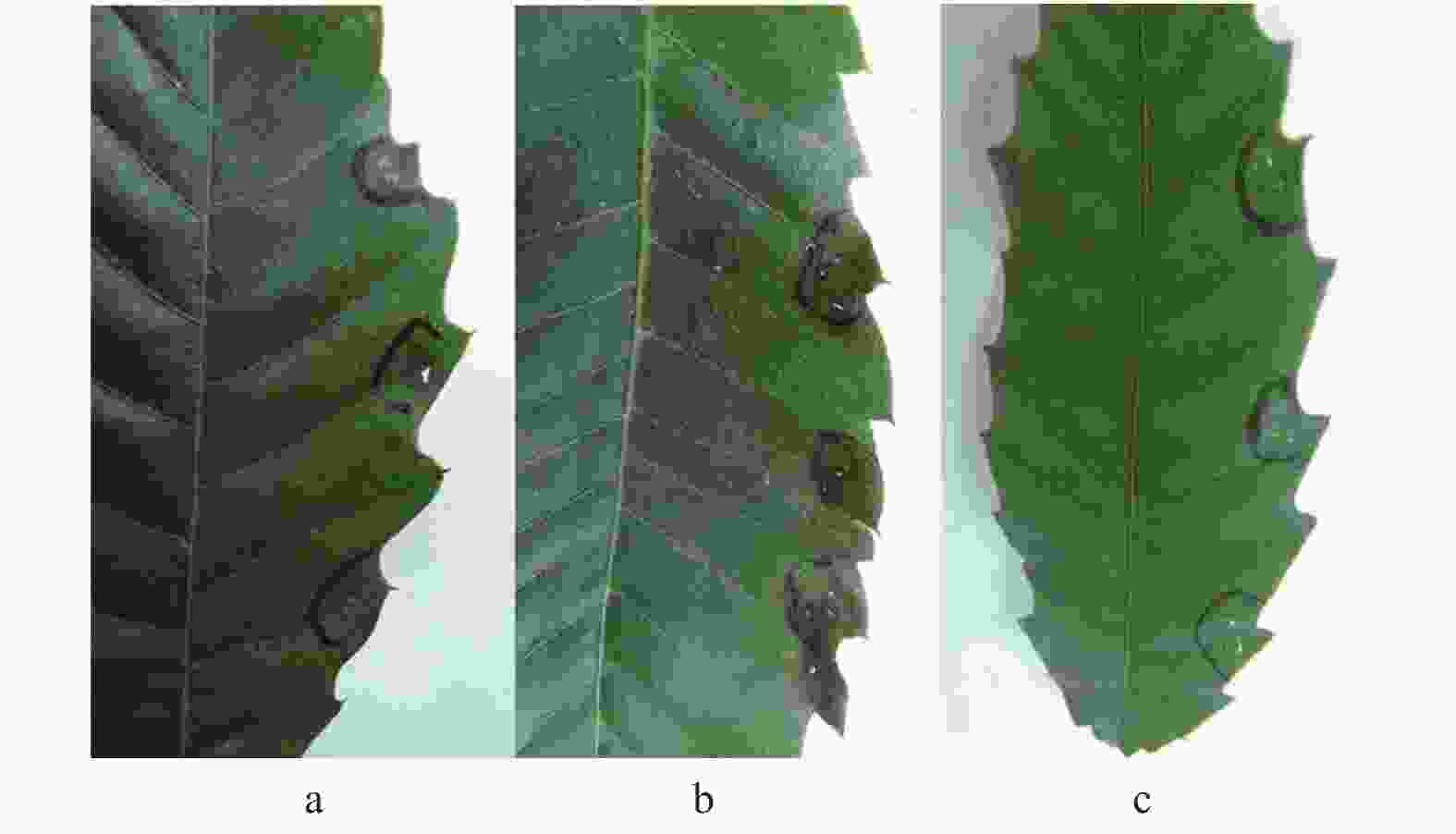
病害在河北板栗园6月开始发病,病树整树叶片均出现症状,发病树木相对集中(图1a),果园树病率10%~50%,单株病叶率可至100%。该病害主要症状是,初始在叶片锐齿尖现褐色小点坏死(图1b),随着时间,顺沿叶缘至两侧脉之间的叶间组织呈现黄褐色至黑褐色病斑,病斑形状不规则,边缘不整齐,大小为2~3 cm,每叶片上能产生多个病斑(图1c),多个黄褐斑可渗透连片,以致使整个叶片上呈不规则的大面积焦枯状,均出现在叶脉间的叶组织上,病斑病健部有不规则宽度的亮黄色边缘线(图1d)。但病害不造成早期落叶。林间病叶未见病症。室内显微镜观察,也未见显著真菌分生孢子器和子实体。室内病组织保湿培养3天后,可镜检到链隔孢属Alternaria的分生孢子。栗园发病症状如图1。
-
栗园及室内在新鲜症状组织上均没有观察或镜检到病症,湿培3天后的病组织表面上可见有分散的较暗的细点,镜检确认是链隔孢属Alternaria的产孢梗及其分生孢子,极易被从病组织表面抹去。显微镜镜检也没有发现病健交界组织中的细菌菌溢现象。
病叶组织分离纯化共获得有效菌株102株,均为真菌。在菌落形态聚类的基础上,选取菌株扩增ITS。结果这些真菌归属于10科,12属,15种,丰度最高为Alternaria alternata(表2)。分离菌株的系统发育树见图2。分离菌株的主要培养物形态见图3。
表 2 河北板栗焦枯叶真菌菌株分离结果
Table 2. Fungi isolation from leaf scorch samples
分离菌号
Codes分离物鉴定
Isolates分离物数
Number of
isolates分离频率
Isolated
frequencies /%Bl21 Alternaria alternata 53 51.96 Bl36 Colletotrichum gloeosporioides 21 20.58 Bl30 Colletotrichum fructicola 7 6.86 Bl1 Nigrospora oryzae 3 2.94 Bl47 Ophiognomonia setacea 3 2.94 Bl12 Alternaria solani 2 1.96 Bl54 Alternaria arborescens 2 1.96 Bl62 Phomopsis sp. 2 1.96 Bl58 Gnomoniopsis smithogilvyi 2 1.96 Bl66 Coniothyrium pyrinum 2 1.96 Bl39 Biscogniauxia sp. 1 0.98 Bl5 Glomerella sp. 1 0.98 Bl53 Leptostroma sp. 1 0.98 Bl55 Sporormiella sp. 1 0.98 Bl61 Dothiorella gregaria 1 0.98 -
依据文献报道,重点挑选分离菌株中潜在的致病菌进行板栗离体叶片接种实验。选出8个菌:半壳霉属Leptostroma sp.,蛇孢日规壳Ophiognomonia setacea(Pers.) Sogonov,梨盾壳霉Coniothyrium pyrinum(Sacc.) J. Sheld.,稻黑孢菌Nigrospora oryzae (Berk. & Broome) Petch,拟茎点霉属Phomopsis sp.,胶孢炭疽菌Colletotrichum gloeosporioides (Penz.) Penz. & Sacc.,Gnomoniopsis smithogilvyi L.A. Shuttlew., E.C.Y. Liew & D.I. Guest,交链链格孢Alternaria alternata (Fr.) Keissl.。只有其中O. setacea和C. pyrinum两种菌接种的实验叶片出现了病斑,4~5天出现症状,一周后病斑基本稳定,不再变化,病斑位置局限于接种点附近(图4)。记号笔标出了接种部位,而其他几组菌种实验连续观察后均未出现明显病斑症状。另外我们还尝试混合了8种菌丝液,并混合Ophiognomonia setacea和Phomopsis sp.菌丝液进行板栗离体叶片接种实验,但并未出现明显症状。
在O. setacea和C. pyrinum离体叶片致病的基础上,进一步进行了两种菌株的1年生板栗盆栽苗致病实验。结果O. setacea和C. pyrinum两种菌均引起盆栽苗成熟叶片的病变,出现褐色坏死病斑。病斑接种后一周左右出现,大小不定,形状不规则,10~14天后病斑停止扩展趋于稳定(图5)。从发病部位可再次分离到这两种病原菌。
-
河北地区发生的栗叶焦枯病害主要症状是,初始叶缘刺突现褐色小点坏死,随后,顺沿叶缘至两侧脉之间的叶间组织呈现黄褐色至黑褐色不规则病斑,可扩展连片,呈不规则的大面积焦枯,叶脉附近组织无症状,病健部有不规则宽度的亮黄色边缘线。病树在栗园可集中成片发生,病树之间受害程度较为一致,单株病树的病叶之间及栗园病树间症状不呈现点状扩散分布趋势特征,病树发病叶片几乎遍布整株,病叶之间症状极为一致。同一栗园同一树木可连年发病,程度年间呈现变化。不同立地或地块栗园和不同栗树间均可发病,程度上可有差异。初步比较观察,病树的根部与健康树的根部没有明显病变和异常气味;病叶枝条(2~3年生)横切面观察,未见到维管束褐变。我国板栗病害,姜淑霞等[13]曾报道了山东地区板栗褐缘叶枯病,称病害突出特点为病斑坏死组织扩散十分快,整叶出现焦枯,叶片脱落严重。叶片脱落后正面产生少量散生子实体,显微镜下分生孢子器散生,病原菌为Phomopsis mollissimae。另外报道有板栗黑(圆)斑病,病斑近圆形,有黄色晕圈,其病原为Cylindrosporium castanicola[14–15];病菌Phomopsis castanea亦引起的叶斑病,表现为叶片大面积干枯[16],但该病症状只有文字描述,没有栗园和病树图像记录。以上症状描述与本病害均存在不一致性,并且具有病症,而本病害在栗园、室内湿培以及镜检均未见到病症以及特定真菌结构。虽然通过组织分离法,分离到很多真菌,但室内离体叶片实验和板栗盆栽苗接种实验,只有菌株O. setacea和C. pyrinum,能在成熟叶片上引起局限性的褐变病变,但均与林间焦枯病症状不一致。细菌Xylella fastidiosa.在欧美报道能引起严重的多种寄主叶焦枯病[5],但未见到在栗属植物上的为害报道。该病具有依赖昆虫传播的特点,病斑具有侵染点且具有明显的病斑发生发展特征,这与我们观察到的板栗叶焦枯病病斑发病特点明显不同,板栗叶焦枯病发病初期和期间,病斑一出现即为较大面积的坏死病斑,病斑没有侵染起始点、没有从侵染点发展至病健部的病斑发生发展过程特征。难以培养的病原细菌,通常需要昆虫媒介的传播,但本病未观察到刺吸式昆虫的伴随或为害。病组织常规镜检未观察到菌溢现象。因此,本病的生物致病侵染属性还有待进一步的探究。
然而,随着气候变暖和环境污染增加,树木叶焦枯病症状类病害频现,这类病害更多的是环境中不利于植物生长发育的物理和化学等非生物因素引起的非侵染性焦枯病[17]。肥、水、土壤、气候及耕作制度等环境条件与该病发生关系十分密切,其中尤以干旱、高温日灼、营养不足为主导因素。SO2是我国主要大气污染物之一,其危害从植物气孔周围细胞开始,初期斑点与气孔形状相似,后期针叶全叶枯死,阔叶发展为淡褐色或灰白色坏死斑,叶果早落[17]。而臭氧主要侵染叶片栅栏组织,叶面出现白色或褐色斑点,对针叶树,症状多出现在成熟叶,对阔叶树,症状首先出现在抽生枝条的近顶端叶片[17]。曹淑云[1]发现当板栗叶片硼过量(含量超过400 mg·kg−1)时即发生焦枯症状;当板栗叶片缺钾(含量低于4000 mg·kg−1)时即表现焦枯症状。但在本研究调查过程中,针对焦枯病,栗农尝试过包括微量元素全肥在内的多种增加相关肥料的措施,对改善症状减轻程度表述不一。近些年为追求板栗产量,栗农采用了大肥大水的管理措施,尤其是为便利收果,在栗园频繁使用农药和除草剂。这些不合理的栗园管理措施,可能是板栗叶焦枯病发生的主要诱因。另外,叶焦枯病的发生可能与板栗品种有关,发病品种主要为燕山早丰3113品种。该品种由河北省昌黎果树研究所于1973年,在杨家峪1.7万株板栗实生树中选出推广,其板栗优质性状改善了板栗品种的局面,对产区的板栗增产起到了重要作用,在河北地区广泛种植[18]。近些年,在全球气候变化的大环境下和进一步提升板栗产值的农事管理活动中,品种频现一些适应性和抗病性问题。因此,综合现场调查和研究结果,我们认为河北近年大面积发生的板栗叶焦枯病的病因,需要扩展考虑非侵染因素诱导的问题。该病亟待综合病原菌、土壤水分利用、农药、复合肥、除草剂、气候条件、管理措施等所有相关因素的综合考察和研究探明病因,为及早防控病害带来的经济损失提供科学依据。
-
本研究新发现并验证了O.setacea和C. pyrinum两种病原菌可在中国板栗叶片上致病。O.setacea与板栗蛇孢日规壳O.castaneae同属,后者为山东地区板栗褐缘叶枯病原菌板栗拟茎点霉Phomopsis castaneae-mollissimae协同致病菌[19]。除O.castaneae外,该属仅有O.apiospora和O.asiatica在我国赤杨和栎属上报道过有分布[19-20]。O.setacea主要在欧美国家报道,与该属其他成员相比寄主范围相对更广泛一些[21-22],在中国板栗上未有过报道。在实验室本菌PDA培养基培养7天的菌落特征为近圆形,直径约5 cm,边沿起伏,浅黄绿粉质,具环状同心轮纹,培养观察期间未见产孢(图6a)。分子鉴定中,菌株ITS rDNA序列与O. setacea模式种的序列相似性97%,我们将其暂定为此种。
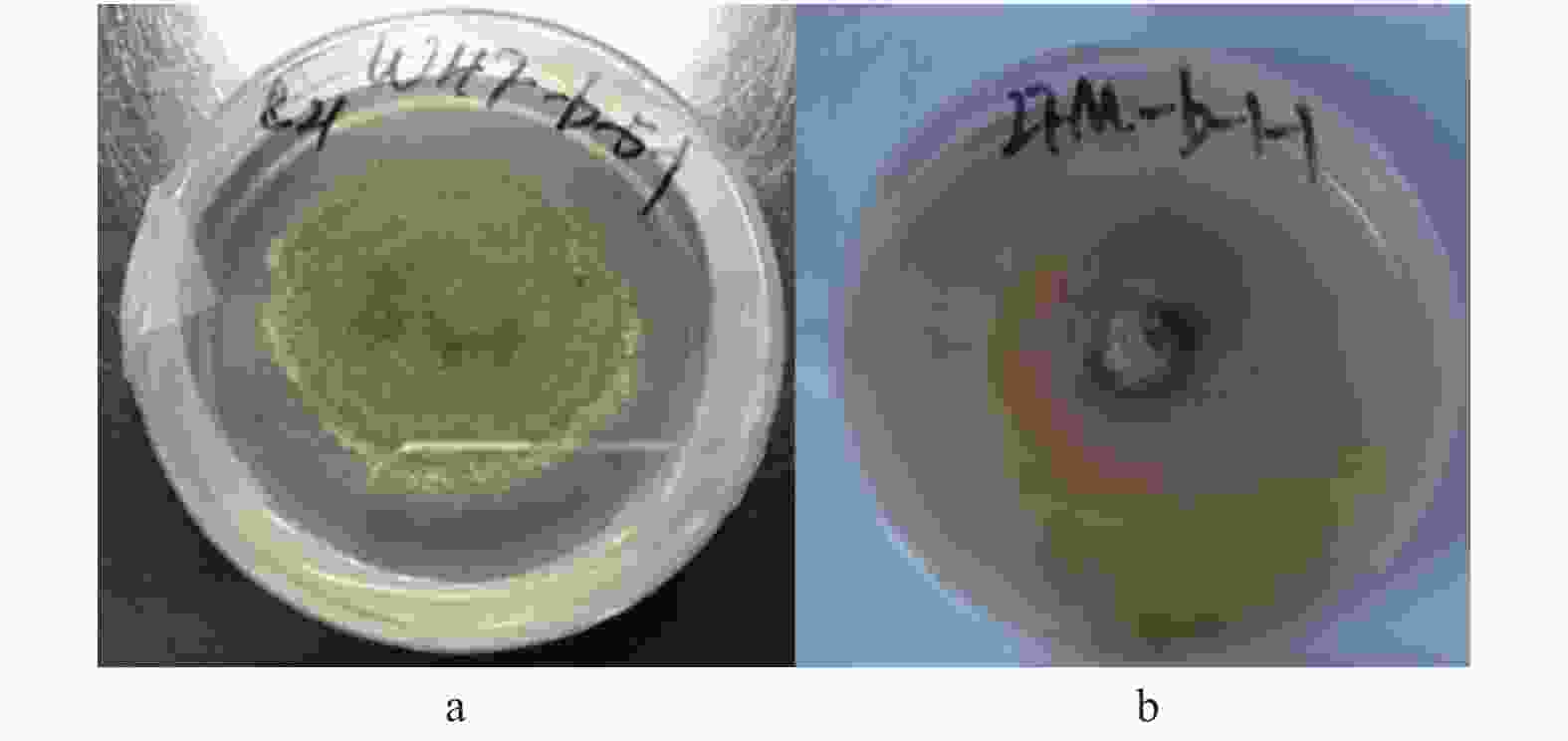
图 6 Ophiognomonia setacea 和Coniothyriumpyrinum 培养物
Figure 6. Culture of Ophiognomonia setacea
and Coniothyrium pyrinum. C. pyrinum主要在苹果叶片形成叶斑,叶斑大小形状颜色各异,斑点严重的叶片变黄早落,一般发生在仲夏雨季[23]。其还可以引起杏树严重的枯死和溃疡病[24]。该菌在板栗上可以引起病害为首次发现。病菌培养性状如图6b。PDA上7天的菌落近圆形,直径约4.5 cm,边缘光滑,菌落棕黄色,产有浅黄色素渗入培养基并弥散于整个培养基中,培养期间未观察到子实体形成。该菌ITS序列与C. pyrinum模式菌株高度同源,覆盖率100%下,相似性达99%。
另外我们分离到的其他一些未表现致病性的菌株,在栗属(Castanea Mill.)其他植物上有相应致病性报道。如分离到的两株Gnomoniopsis smithogilvyi被报道能在欧洲板栗(Castanea sativa Mill.)上引起严重的溃疡症[25]及果腐病[26-27]。在全球气候变化等因素影响下,病原菌寄主范围扩大现象日益普遍,此类病原菌存在成为板栗新发病害的可能性,应予以重视。
-
初步研究结果表明,青龙和迁西板栗果园严重发生的叶焦枯病中,未能明确为上述相关病原菌所引起。其病因有待进一步的综合、深入和细致地开展研究,尤其需要考虑与为追求产量,高投入的不合理水、肥和农药使用等栗园管理措施以及环境因素和品种适应性等相关性的可能。研究中,发现并验证的O. setacea和C. pyrinum两种病原菌可在中国板栗上致病,为板栗可能出现的新病害的发生和认识提供了基础。
板栗叶焦枯病相关病菌分离及病因初探
Primary Study on Causes and Associated Pathogens for Chestnut Leaf Scorch
-
摘要:
目的 近年河北板栗主产区大面积严重发生板栗叶焦枯病,造成板栗减产和栗农经济损失,该病害发病原因亟待明确,尽早进行有效的防控。 方法 通过林间症状调查和病叶组织样本微生物分离、离体叶片及盆栽苗接种致病柯赫法则验证,结合果园管理措施和环境因素分析,对病因进行了探讨。 结果 该病栗园病树率可达10%~50%,单株树病叶率可至90%以上,病叶主要症状为叶缘及叶脉间组织干枯。林间、室内湿培及显微镜切片均未发现病组织存在病症或病原菌组织。组织分离纯化获得相关微生物菌株102株,均为真菌,归属10科12属15种,丰度最高为Alternaria alternata。致病性接种试验表明,其中的Ophiognomonia setacea和Coniothyrium pyrinum能引起接种组织局部褐变发病,但症状表现与林间叶焦枯病有差异。 结论 研究发现Ophiognomonia setacea和Coniothyrium pyrinum两种真菌具有致病性,可在中国板栗引发叶斑病,但症状不同于发生于栗园的板栗叶焦枯病。当前频发性树叶焦枯病类病害常见诱发因素,多与气候变暖、异常天气、环境污染以及生产和病虫害管理措施不当引起的品种生长不适应密切关联。河北大面积发生的板栗叶焦枯病病因有待综合非侵染因子进行深入研究和探明,以期为尽快采取有效防控措施奠定基础。 Abstract:Objective To investigate and analyze leaf scorch of Castanea mollissima occurred in the main chestnut producing areas in Hebei Province. Method Combined the investigation of the symptoms in the forest, the isolation of microbes from the symptomatic leaf tissue, the confirmation of Koch's law in vitro leaves and potted seedlings, with the management measures of orchard and the environmental factors, the causes of disease were analyzed and discussed. Result The disease rate of orchard trees was 20%–50%, the disease rate of individual tree leaves can be up to 100%, the main symptom of individual leaf was dry leaf margin and interveinal tissue. No pathogen was found in the forest symptomatic tissues and microscopic examination. A total of 102 strains were isolated and purified from symptomatic tissues, all of which were fungi, belonging to 10 families, 12 genera and 15 species with Alternaria alternata as the most abundant. The inoculation test showed that Ophiognomonia setacea and Coniothyrium pyrinum could induce the browning of leaf tissue of Chinese chestnut. However, the symptom was significantly different from that of chestnut forest leaf scorch. Conclusion Two pathogens, O. setacea and Coniothyrium Pyrinum, were newly found and verified to be pathogenic to Chinese chestnut leaves, causing chestnut tree leaf spots, which were different with the leaf scorch symptoms in chestnut observed. Currently, leaf scorches in trees are ordinarily connected with environmental and physical factors which influenced tree growth with global warming, extreme weather, pollution, inadequate silviculture and control pests/diseases managements. Therefore, integrating non-infection to investigate causes of the leaf scorch of Castanea mollissima occurred in Hebei Province is strongly proposed in order to effectively prevent and control the disease as soon as possible. -
表 1 Genbank中下载用于比对分析做系统发育树的参考序列
Table 1. Sequences from Genbank for alignment and phylogenetic tree
序列来源
Source菌株号
Strain No.ITS序列号
ITS No.Alternaria alternata isolate Alt-C81 MN044802 Colletotrichum gloeosporioides IMI 356878 from type material NR_160754 Colletotrichum fructicola MFLU 090228 from type material NR_144783 Nigrospora oryzae isolate VARB205 KT966519 Ophiognomonia setacea CBS 128354 MH864895 Alternaria solani CBS 109158 KJ718239 Alternaria arborescens CBS 102605 NR_135927 Phomopsis sp. GJS 83-377 AF102999 Gnomoniopsis smithogilvyi CBS 130190 NR_166040 Coniothyrium pyrinum CBS 110.40 MH856053 Biscogniauxia sp. strain LPS-70 MF379340 Glomerella sp. isolate ERS038 KY413720 Leptostroma sp. strain Ecf-5 MK211262 Sporormiella sp. strain LTL532 MF663586 Dothiorella gregaria isolate hz-S8 FJ517548 Acervus globulosus HKAS 88987 NR_159565 表 2 河北板栗焦枯叶真菌菌株分离结果
Table 2. Fungi isolation from leaf scorch samples
分离菌号
Codes分离物鉴定
Isolates分离物数
Number of
isolates分离频率
Isolated
frequencies /%Bl21 Alternaria alternata 53 51.96 Bl36 Colletotrichum gloeosporioides 21 20.58 Bl30 Colletotrichum fructicola 7 6.86 Bl1 Nigrospora oryzae 3 2.94 Bl47 Ophiognomonia setacea 3 2.94 Bl12 Alternaria solani 2 1.96 Bl54 Alternaria arborescens 2 1.96 Bl62 Phomopsis sp. 2 1.96 Bl58 Gnomoniopsis smithogilvyi 2 1.96 Bl66 Coniothyrium pyrinum 2 1.96 Bl39 Biscogniauxia sp. 1 0.98 Bl5 Glomerella sp. 1 0.98 Bl53 Leptostroma sp. 1 0.98 Bl55 Sporormiella sp. 1 0.98 Bl61 Dothiorella gregaria 1 0.98 -
[1] 曹淑云. 板栗叶片焦枯症的发生与防治[J]. 烟台果树, 2005, (3):54-55. doi: 10.3969/j.issn.1005-9938.2005.01.043 [2] 周道明. 棉花红叶茎枯病及防治措施[J]. 农村科技, 2006,(12):34. [3] 姜淑霞, 仲伟元, 高 锋, 等. 板栗褐缘叶枯病发病规律及防治试验[J]. 中国森林病虫, 2010, 29(5):30-31. doi: 10.3969/j.issn.1671-0886.2010.05.010 [4] 龚 赛, 张秀停, 聂 阳, 等. 板栗褐缘叶枯病协同致病菌Ophiognomonia castaneae的生活史[J]. 菌物学报, 2016, 35(10):1178-1186. [5] Bucci E M. Xylella fastidiosa, a new plant pathogen that threatens global farming: Ecology, molecular biology, search for remedies[J]. Biochemical and Biophysical Research Communications, 2018, 502(2): 173-182. doi: 10.1016/j.bbrc.2018.05.073 [6] Sicard A, Zeilinger R A, Vanhove M, et al. Xylella fastidiosa: Insights into an emerging plant pathogen[J]. Annual Review of Phytopathology, 2018, 56: 181-202. doi: 10.1146/annurev-phyto-080417-045849 [7] 黄武刚, 程丽莉, 周志军, 等. 板栗野生居群遗传多样性研究[J]. 果树学报, 2010, 2(27):227-232. [8] 侯 明. 河北板栗资源开发设想[J]. 河北省科学院学报, 1993, (4):23-27. [9] 周定奎, 吕纪水. 开拓河北板栗市场[J]. 合作经济与科技, 2005, 13:10-11. doi: 10.3969/j.issn.1672-190X.2005.19.003 [10] 郗荣庭, 曲宪忠. 河北经济林[M]. 北京: 中国林业出版社, 2001.01. [11] 姬广海, 吴亚鹏, 张乃明, 等. 芦荟根腐病病原菌的鉴定[J]. 植物病理学报, 2007, 37(2):207-209. doi: 10.3321/j.issn:0412-0914.2007.02.015 [12] Chang S, Puryear J, Cairney J. A simple and efficient method for isolating RNA from pine trees[J]. Plant Molecular Biology Reporter, 1993, 11(2): 113-116. doi: 10.1007/BF02670468 [13] 姜淑霞, 刘传忠, 王庆华, 等. 板栗新病害褐缘叶枯病及病原鉴定[J]. 林业科学, 2011, 47(5):177-180. doi: 10.11707/j.1001-7488.20110127 [14] 陆家云. 植物病原真菌学[M]. 北京: 中国农业出版杜, 2001, 461-462. [15] 谢宝多. 板栗病害[M]. 北京: 中国林业出版杜, 1998, 31-37. [16] 戚佩坤, 姜子德, 向梅梅. 中国真菌志-第三十四卷, 拟茎点霉属[M]. 北京: 科学出版杜, 2007, 2-74. [17] 贺 伟, 叶建仁. 森林病理学[M]. 北京: 中国林业出版社, 2017, 295-313. [18] 于秋香. 介绍几个燕山板栗优良品种[J]. 河北果树, 2016, (6):28-29. [19] Gong S, Zhang X, Jiang S, et al. A new species of Ophiognomonia from Northern China inhabiting the lesions of chestnut leaves infected with Diaporthe eres[J]. Mycological Progress, 2017, 16(1): 83-91. doi: 10.1007/s11557-016-1255-z [20] Walker D M, Castlebury L A, Rossman A Y, et al. Phylogeny and taxonomy of Ophiognomonia (Gnomoniaceae, Diaporthales), including twenty-five new species in this highly diverse genus[J]. Fungal Diversity, 2012, 57(1): 85-147. doi: 10.1007/s13225-012-0200-y [21] Sogonov M V, Castlebury L A, Rossman A Y, et al. Leaf-inhabiting genera of the Gnomoniaceae, Diaporthales[J]. Studies in Mycology, 2008, 62: 1-77. doi: 10.3114/sim.2008.62.01 [22] Stoykov D Y. New records of Ophiognomonia (Gnomoniaceae, Diaporthales) from Bulgaria, Greece and Turkey[J]. Phytologia Balcanica: International Journal of Balkan Flora and Vegetation, 2016, 22(3): 297-301. [23] Ahmed N, Ahmed T. Fruits related problems and their management in Rajouri district of Jammu and Kashmir[J]. Journal of the Humanities and Social Sciences, 2013, 12(2): 65-75. [24] Fazli M A, Razdan V K. A new record of Coniothyrium pyrinum on almond trees[J]. Indian Phytopathology, 1991, 44(3). [25] Pasche S, Calmin G, Auderset G, et al. Gnomoniopsis smithogilvyi causes chestnut canker symptoms inCastanea sativashoots in Switzerland[J]. Fungal Genetics and Biology, 2016, 87: 9-21. doi: 10.1016/j.fgb.2016.01.002 [26] Visentin I, Gentile S, Valentino D, et al. Gnomoniopsis castanea sp. nov. (Gnomoniaceae, Diaporthales) as the causal agent of nut rot in sweet chestnut[J]. Journal of Plant Pathology, 2012: 411-419. [27] Shuttleworth L A, Walker D M, Guest D I. The chestnut pathogen Gnomoniopsis smithogilvyi (Gnomoniaceae, Diaporthales) and its synonyms[J]. Mycotaxon, 2016, 130(4): 929-940. doi: 10.5248/130.929 -





 下载:
下载: