-
大量的监测数据和全球气候变化模型表明降雨时空格局正在发生改变,由此引起的干旱频度、强度以及持续时间的增加将会成为许多地区面临的问题[1],尤其是在亚热带地区[2-3]。干旱的增加将会对森林生态系统的群落多样性、生存以及生产力产生巨大影响[4],进而可能影响局域或全球的碳水平衡。
叶片对环境变化较为敏感且可塑性较大,其性状特征与植物生存策略和利用资源的能力密切相关[5-6]。因此,加强叶片性状的研究有助于更好地了解植物对干旱的响应与适应。目前,国内外学者从叶片形态、解剖以及生理等性状探究了植物对干旱的响应与适应[7-11]。然而,这些研究大多数是短期的控制实验且缺乏对于叶片解剖结构和光合生理同步研究。因此,对于叶片解剖结构和光合生理对于相对长期干旱胁迫的适应以及二者之间关系的了解依然不足。
研究降雨减少对于叶片解剖结构和光合生理的影响有助于揭示树木对于干旱的适应机制[11]。不同树种对于干旱的敏感性和响应时间存在差异,其叶片解剖结构和光合生理对干旱的响应和适应可能存在物种特异性[11]。对不同水分胁迫条件下地中海2种观赏灌木(马缨丹(Lantana camara L.)和女贞(Ligustrum lucidum W.T.))的研究发现:2种灌木叶片厚度对于水分胁迫响应存在明显差异并指出叶片光合能力与水分胁迫的强度有关[8]。Gratani等[12]发现,干旱条件下叶片厚度的增加可以减少蒸腾失水,维持细胞内较高的水势,从而保证正常的光合速率,提高植物的水分利用效率。然而,Binks等[13]和Rowland等[14]对模拟干旱条件下热带雨林树种的研究证实,叶片解剖结构对于干旱的响应与物种的抗旱性有关,且发现由干旱引起的叶片解剖结构改变未对光合性状产生影响[13]。因此,到目前为止,叶片解剖结构和光合生理对降雨减少的响应认知仍然存在很大争议,这使得理解树木生长对于气候变化的响应具有较大的不确定性。
马尾松(Pinus massoniana Lamb.)和红锥(Castanopsis hystrix Miq.)是我国亚热带地区的典型造林树种。马尾松具有适应性强、生长迅速、生产力高等特点[15],红锥材质优良,具有多种用途[16]。红锥和马尾松的叶片习性、木材解剖和水力学性状存在明显差异,导致2树种拥有不同的生活史特征和环境需求[17-18],这为探究具有不同生活史特征的物种对于干旱的响应和适应提供了绝佳的机会。前人研究已经认识到马尾松和红锥人工林生长对于干旱的响应存在差异,穿透雨减少后马尾松径向生长未发生变化而红锥径向生长出现显著下降[19]。然而,马尾松和红锥叶片解剖结构和光合生理如何响应干旱目前尚未可知。为了进一步探究叶片解剖结构、养分和光合生理对于干旱的响应,解析三者之间的关系,本研究在南亚热带选取马尾松和红锥人工纯林设置穿透雨减少实验,测定了2种树木叶片解剖结构、叶片养分(氮和磷)、气体交换参数和叶绿素荧光参数,旨在探索以下科学问题:(1)马尾松和红锥叶片解剖、叶片养分以及光合生理如何响应干旱?减雨处理后树木是否能够产生更耐旱的叶片?(2)叶片解剖和叶片养分是否能够影响叶片的光合生理?
-
研究地点位于广西壮族自治区凭祥市中国林业科学研究院热带林业实验中心伏波实验场(22°10′ N, 106°50′ E),该地区位于南亚热带季风气候区,属湿润-半湿润气候,干湿季分明。年降水量1 241~1 695 mm,降雨主要集中在5—9月,年平均温度21~22 ℃,相对湿度80%~84%,海拔550 m。主要的地貌类型以低山丘陵为主,土壤为红壤,质地为砂质壤土。
马尾松和红锥人工林均是1983年皆伐杉木(Cunninghamia lanceolata (Lamb.) Hook.)林后种植,最初的土壤质地和养分一致,林分密度均为2 500 株·hm−2,之后经过多年间伐,马尾松和红锥人工林密度分别为275 ± 45、334 ± 92株·hm−2,平均胸径分别为31.26 ± 2.15、27.15 ± 1.96 cm,平均树高分别为18.8 ± 1.30 、19.84 ± 2.96 m。马尾松林下草本植物主要有弓果黍(Cyrtococcum patens (L.) A. Camus)、乌毛蕨(Blechnum orientale L.)、荩草(Arthraxon hispidus (Trin.) Makino)和小花露籽草(Ottochloa nodosa var. micrantha (Balansa) Keng f.)等。红锥林下草本主要有金毛狗(Cibotium barometz (L.) J. Sm.)、淡竹叶(Lophatherum gracile Brongn.)、铁芒萁(Dicranopteris linearis (Burm.) Underw.)和扇叶铁线蕨(Adiantum flabellulatum L.)等。
-
2012年9月在马尾松和红锥人工林中分别设置了6块20 m × 20 m样地,样地间距离>20 m,实施了自然条件下降雨(对照)和部分穿透雨剔除(50%,TFE)2种不同的处理,每个处理3个重复,2个树种共12块样地。在穿透雨剔除样地距离地面0.5~1.5 m处安装了由不锈钢架和PEP膜组成的穿透雨剔除装置,铺设的透明塑料薄膜面积为样地面积的50%。截取的穿透雨通过导水槽及样地周围的排水沟排出样地。为降低TFE装置对样地环境的影响,在对照样地同样的高度假架设了有不锈钢架和3 mm孔径白色尼龙网组成的装置。经测试,各样地装置下照度无显著差异。定期对PEP膜、尼龙网以及导水槽上的凋落物清理并原位归还到样地。与此同时,在每个样地都安装HoBo土壤温湿度记录仪(Onset Computer Corp.,美国)对5 cm表层土壤和深层土壤(10、30、50、70、90 cm)的温湿度进行记录,记录间隔为1 h,每个样地各安装了3个记录仪。
-
由于马尾松和红锥树冠较高,难以原位测定,因此,本研究采用高枝剪和人为爬树剪取离体枝的方法对光合生理参数进行测量。为避免枝条栓塞影响测定结果,所采集测定气体交换和叶绿素荧光参数的枝条都接近1 m且均为冠层顶部的阳生枝条,将获取的离体枝条立即插入装有水的水桶中,在水中用剪枝剪在距离切口10 cm处重新修剪。通过这样的处理一定程度上避免离体枝条发生栓塞,确保一段时间内离体枝条上叶片样品的生理活性与树体一致[20-21]。
气体交换参数的测定:2019年8月中旬(湿季)和2020年1月中旬(干季)选择无雨天气于9:00—11:30时段,采用便携式光合仪(LI-6400, Li-Cor, Lincoln Inc., NE, USA)测定了马尾松和红锥当年生枝条上完全伸展的健康叶片的净光合速率(Pn)、气孔导度(Gs)、胞间CO2浓度(Ci)、蒸腾速率(Tr)等参数。设定的光量子通量密度为1 500 μmol·m−2·s−1,CO2浓度设置为环境CO2浓度,环境温度控制在25 ℃。在对照样地和处理样地分别随机选取了3棵树,每棵树选取2~3个叶片,每个叶片测定3次重复。利用下列公式计算水分利用效率(WUE):WUE = Pn/Tr。
叶绿素荧光参数于2019年8月测定。具体方法是先将叶片暗适应30 min用PAM-2500便携式叶绿素荧光仪(Heinz Walz GmbH,Effeltrich,Germany)测定最小初始荧光(Fo)、暗适应下最大荧光(Fm)、光系统Ⅱ(PSⅡ)最大光化学效率(Fv/Fm)、实际光化学效率(ΦPSII)、光化学猝灭系数(qP)、非光化学猝灭系数(NPQ和qN)和表观电子传递速率(ETR)等荧光参数。
-
2020年1月在红锥和马尾松的对照样地和穿透雨减少样地选取与测定气体交换参数的叶片位置相近、发育一致的健康叶片各5片(马尾松针叶各5簇),用FAA固定液保存,红锥剪取叶片中部与叶边缘之间1~2 cm2,马尾松剪取中部5~6 mm片段,采用常规石蜡切片法制片。切片厚度为8~12 μm,用番红-固绿染色,中性树胶封片。对照样地和处理样地分别随机选取了3片叶子的制片置于Nikon(NIS-Elements D)光学显微镜下观察和拍照记录,并用仪器自带分析软件测量了红锥叶片上下表皮厚度、叶肉栅栏组织和海绵组织厚度、叶片厚度和马尾松针叶的上下表皮厚度、叶肉厚度以及针叶厚度。
-
采用指甲油印迹法。每个样地在冠层树木枝条上随机选取3片健康完全开展的阳生叶片,红锥叶片避开主脉用无色指甲油均匀涂抹于叶片中部,马尾松针叶用指甲油涂抹叶片中部腹面,干燥后剥下所形成的印痕干膜,制片后用Nikon光学显微镜观察和拍照,利用自带测量软件获取气孔密度。
-
在测定光合特性指标的枝条上,随机选取一定量的健康成熟无病虫害的叶片,放入信封袋中带回实验室。将叶片清洗干净后在600 w的微波炉中杀青90 s,然后再将其放到65 ℃的烘箱中烘干48 h。用高速粉碎机将烘干的叶片粉碎后过100目筛(0.147 mm)备用。叶片全氮(TN)和全磷(TP)分别采用凯氏定氮法和钼锑抗比色法测定。
-
用SPSS17.0软件对数据进行统计分析,采用独立样本t检验分析处理之间、树种之间以及季节之间各指标的差异性,以p < 0.05为显著性评价标准;采用Pearson相关系数分析叶片解剖性状、叶片养分和气体交换参数之间的相关关系。本研究中所有的数据均采用平均值 ± 标准误(Mean ± S.E.)的形式表示。用Origin 2021软件作图。
-
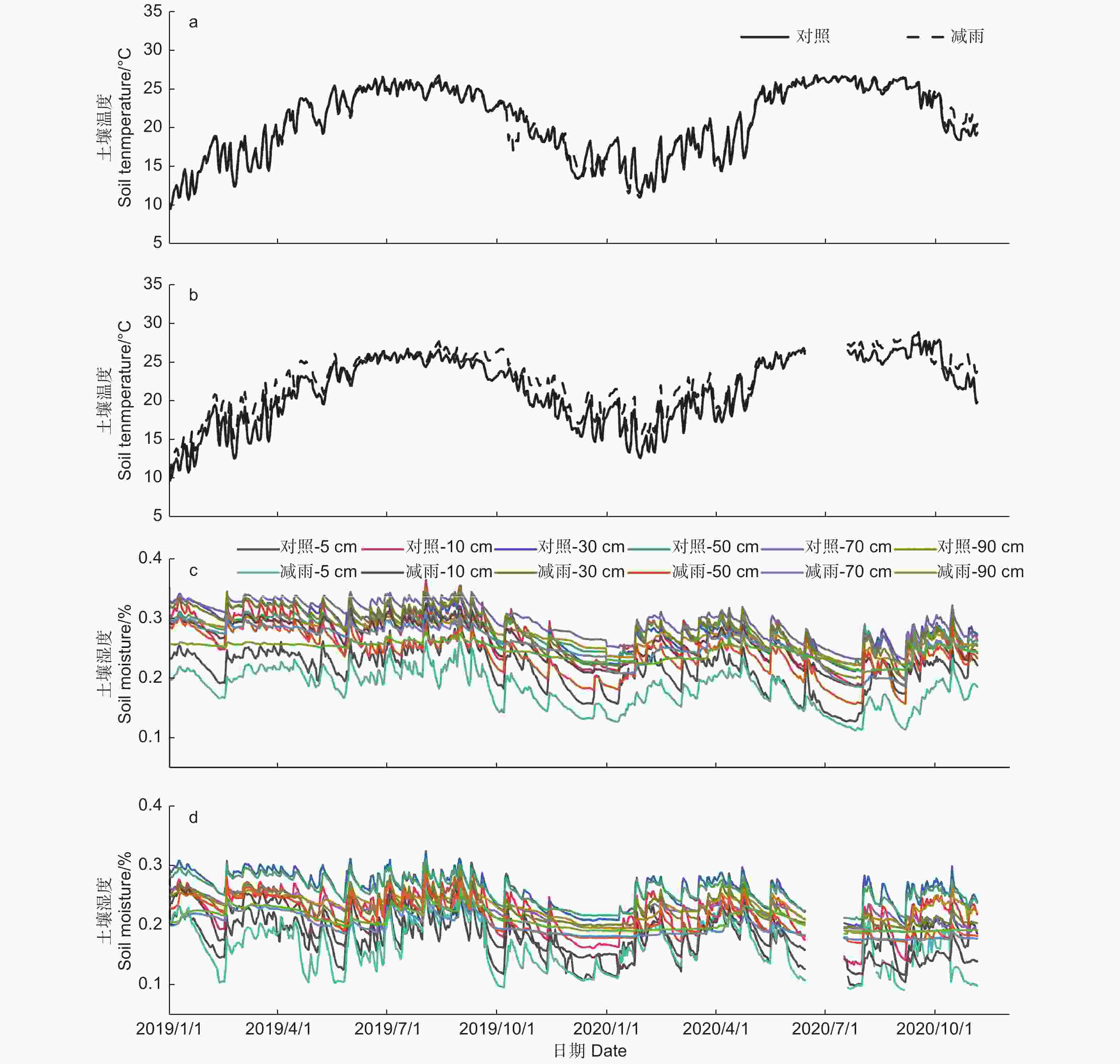
土壤温湿度存在明显的季节差异,湿季(5—9月)的土壤温湿度大于干季(10月—次年4月)的土壤温湿度(p < 0.05)。穿透雨减少处理对马尾松和红锥表层(5 cm)土壤温度无影响(p > 0.05),但降低了2树种的表层(5 cm)和深层(10、30、50、70、90 cm)土壤湿度(p < 0.05;图1),其中,穿透雨减少使50 cm土层的土壤湿度下降最大,马尾松和红锥分别下降了15.96%和16.76%。
-
由图2得出,马尾松和红锥叶片解剖结构明显不同。马尾松的叶片横切面呈半圆形,叶肉不分化栅栏组织和海绵组织,细胞排列不规则但排列紧密,叶肉细胞间存在多个树脂道。红锥叶片叶肉存在明显分化的栅栏组织和海绵组织,细胞排列规则但不紧密,海绵组织发达。穿透雨减少8 a后马尾松针叶的上、下表皮厚度、内皮层厚度、针叶厚度以及气孔密度分别显著增加了28.35%、29.94%、8.46%、8.13%和10.55%,但叶肉厚度未发生显著改变。对于红锥来说,穿透雨减少不仅明显增加了上表皮厚度(24.43%),而且显著的降低了海绵组织的厚度(23.90%)。穿透雨减少使红锥叶片栅栏和海绵组织比值增加明显,但未改变其叶片厚度和气孔密度(表1)。
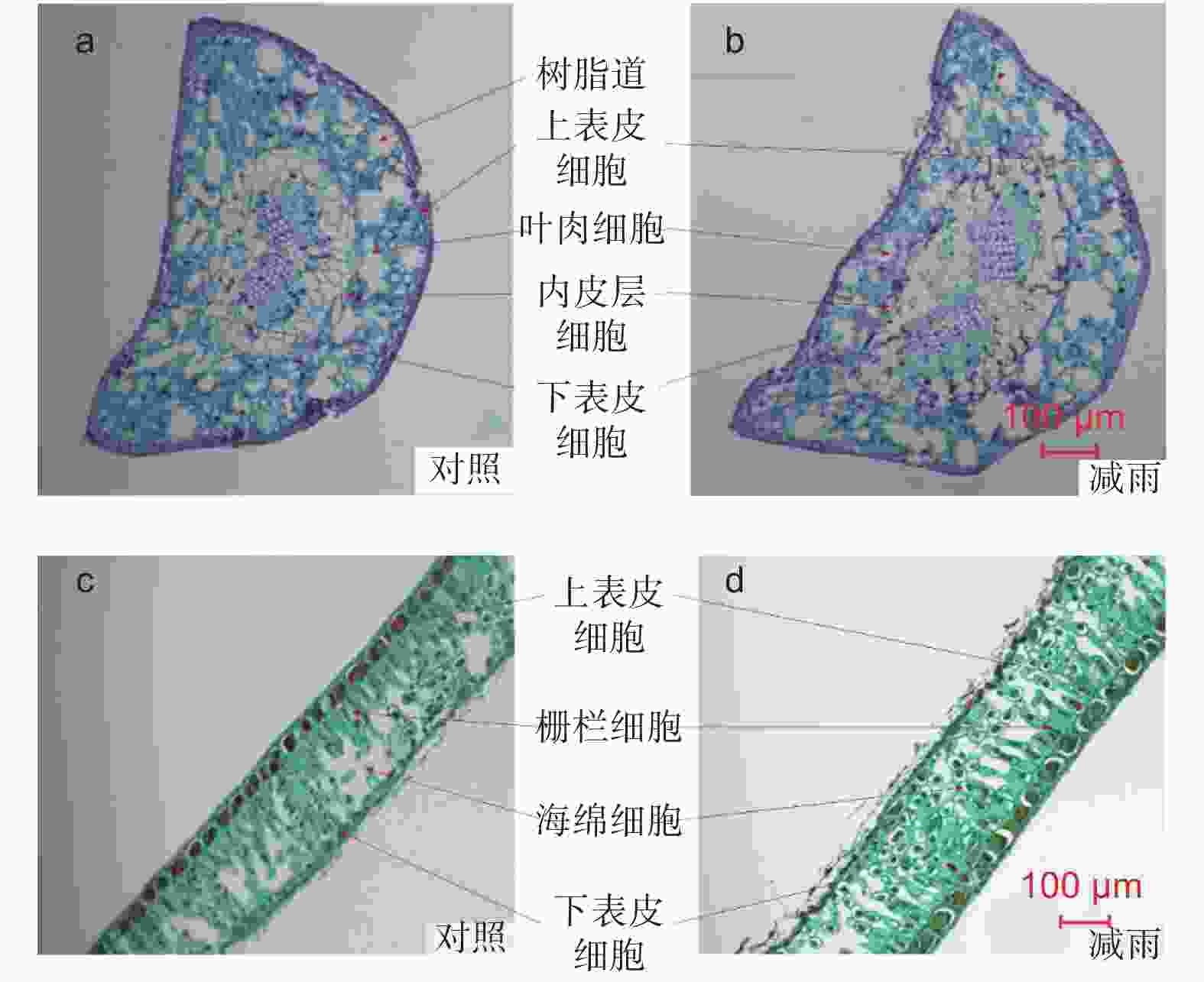
图 2 对照样地和减雨样地马尾松(a, b)和红锥(c, d)叶片解剖
Figure 2. Leaf anatomical structure of Pinus massoniana (a, b) and Castanopsis hystrix (c, d) in the control and the rainfall-reduced plots
表 1 马尾松和红锥叶片解剖结构
Table 1. Leaf anatomical structure of Pinus massoniana and Castanopsis hystrix
树种
Tree species叶片指标
Index of leaf对照
Control处理
Treatment马尾松
Pinus massoniana上表皮厚度 Thickness of upper epidermis/μm 12.77 ± 0.21 b 16.39 ± 0.67 a 下表皮厚度 Thickness of lower epidermis /μm 21.41 ± 0.21 b 27.82 ± 0.75 a 叶肉厚度 Mesophyll thickness/μm 104.87 ± 8.91 a 99.79 ± 12.30 a 内皮层厚度 Endothelium thickness/μm 13.83 ± 0.43 b 15.00 ± 0.31 a 树脂道数 Resin canals number 11.33 ± 0.67 a 11.00 ± 0.65 a 针叶厚度 Needle thickness/μm 507.94 ± 3.71 b 549.22 ± 30.94 a 针叶宽 Needle width/μm 875.49 ± 22.11 a 854.03 ± 14.81 a 气孔密度 Stomata density/(number·mm−2) 119.96 ± 10.48 b 132.61 ± 10.21 a 红锥
Castanopsis hystrix上表皮厚度 Thickness of upper epidermis /μm 17.44 ± 0.37 b 21.7 ± 0.54 a 下表皮厚度 Thickness of lower epidermis /μm 11.86 ± 0.37 a 13.27 ± 0.66 a 栅栏组织 Palisade tissue thickness/μm 53.43 ± 1.78 a 53.64 ± 1.38 a 海绵组织 Spongy tissue thickness/μm 44.26 ± 1.5 a 33.68 ± 1.3 b 栅栏/海绵 Ratio of Palisade tissue and spongy tissue 1.22 ± 0.04 b 1.60± 0.04 a 叶片厚度 Leaf thickness/μm 140.10 ± 2.44 a 136.02 ± 2.02 a 气孔密度 Stomata density /(number·mm−2) 468.71 ± 16.68 a 489.58 ± 21.67 a 注:不同的小写字母代表对照和减雨样地之间存在显著差异(p < 0.05)
Note: Lowercase letters indicate significant differences between the control plots and treatment plots (p < 0.05) -
由表2得出:马尾松和红锥的气体交换参数存在季节差异和种间差异(p < 0.05);湿季马尾松和红锥的净光合速率(Pn)、蒸腾速率(Tr)、气孔导度(Gs)显著高于干季( p < 0.05);与马尾松对比,红锥受到的气孔限制(Ls)更大,水分利用效率(WUE)更高,尤其是在干季。穿透雨减少对马尾松和红锥气体交换参数和叶绿素荧光参数均无影响(p > 0.05;表2、3和图3)。
表 2 对照样地和减雨样地马尾松和红锥气体交换参数
Table 2. Gas exchange parameters of Pinus massoniana and Castanopsis hystrix in control and rainfall-reduced plots
树种
Tree species气体交换参数
Gas exchange parameters湿季 Wet season 干季 Dry season 对照 Control 处理 Treatment 对照 Control 处理 Treatment 马尾松
Pinus massonianaPn / (μmol·m−2·s−1) 4.17 ± 0.18 Aa 3.92 ± 0.20 Aa 2.10 ± 0.19 Ba 1.72 ± 0.03 Ba Gs / (mol·m−2·s−1) 0.063 ± 0.006 Aa 0.058 ± 0.004 Aa 0.025 ± 0.002 Ba 0.023 ± 0.003 Ba Ci / (μmol·m−2·s−1) 295.95 ± 12.71 Aa 289.42 ± 10.22 Aa 305.44 ± 8.03 Aa 308.22 ± 22.37 Aa Tr / (mmol·m−2·s−1) 0.89 ± 0.76 Aa 0.86 ± 0.06 Aa 0.48 ± 0.05 Ba 0.34 ± 0.064 Ba WUE 4.88 ± 0.47 Aa 4.67 ± 0.41 Aa 4.79 ± 0.52 Aa 6.08 ± 1.18 Aa Ls 0.29 ± 0.07 Ba 0.30 ± 0.02 Ba 0.33 ± 0.044 Aa 0.36 ± 0.065 Aa 红锥
Castanopsis hystrixPn / (μmol·m−2·s−1) 4.59 ± 0.38 Aa 4.42 ± 0.39 Aa 3.66 ± 0.51 Ba 3.24 ± 0.32 Ba Gs / (mol·m−2·s−1) 0.07 ± 0.01 Aa 0.06 ± 0.01 Aa 0.02 ± 0.002 Ba 0.017 ± 0.002 Ba Ci / (μmol·m−2·s−1) 289.10 ± 12.87 Aa 273.31 ± 11.09 Aa 253.24 ± 10.46 Aa 262.85 ± 13.63 Aa Tr / (mmol·m−2·s−1) 0.85 ± 0.09 Aa 0.67 ± 0.06 Aa 0.42 ± 0.02 Ba 0.35 ± 0.02 Ba WUE 5.79 ± 0.62 Ba 6.74 ± 0.70 Ba 8.65 ± 1.08 Aa 9.26 ± 0.66 Aa Ls 0.30 ± 0.03 Ba 0.33 ± 0.03 Ba 0.77 ± 0.02 Aa 0.80 ± 0.03 Aa 注:Pn:净光合速率;Gs:气孔导度;Tr:蒸腾速率;WUE:水分利用效率;Ls:气孔限制。不同的小写字母代表对照和减雨样地之间存在显著差异,不同的大写字母表示季节间存在显著差异(p < 0.05)
Notes: Pn: Net photosynthetic rate; Gs: Stomatal conductance; Tr : Transpiration rate; WUE: Water use efficiency; Ls: Stomatal limitation. Lowercase letters indicate significant differences between the control plots and treatment plots. Uppercase letters indicate significant differences among seasons (p < 0.05)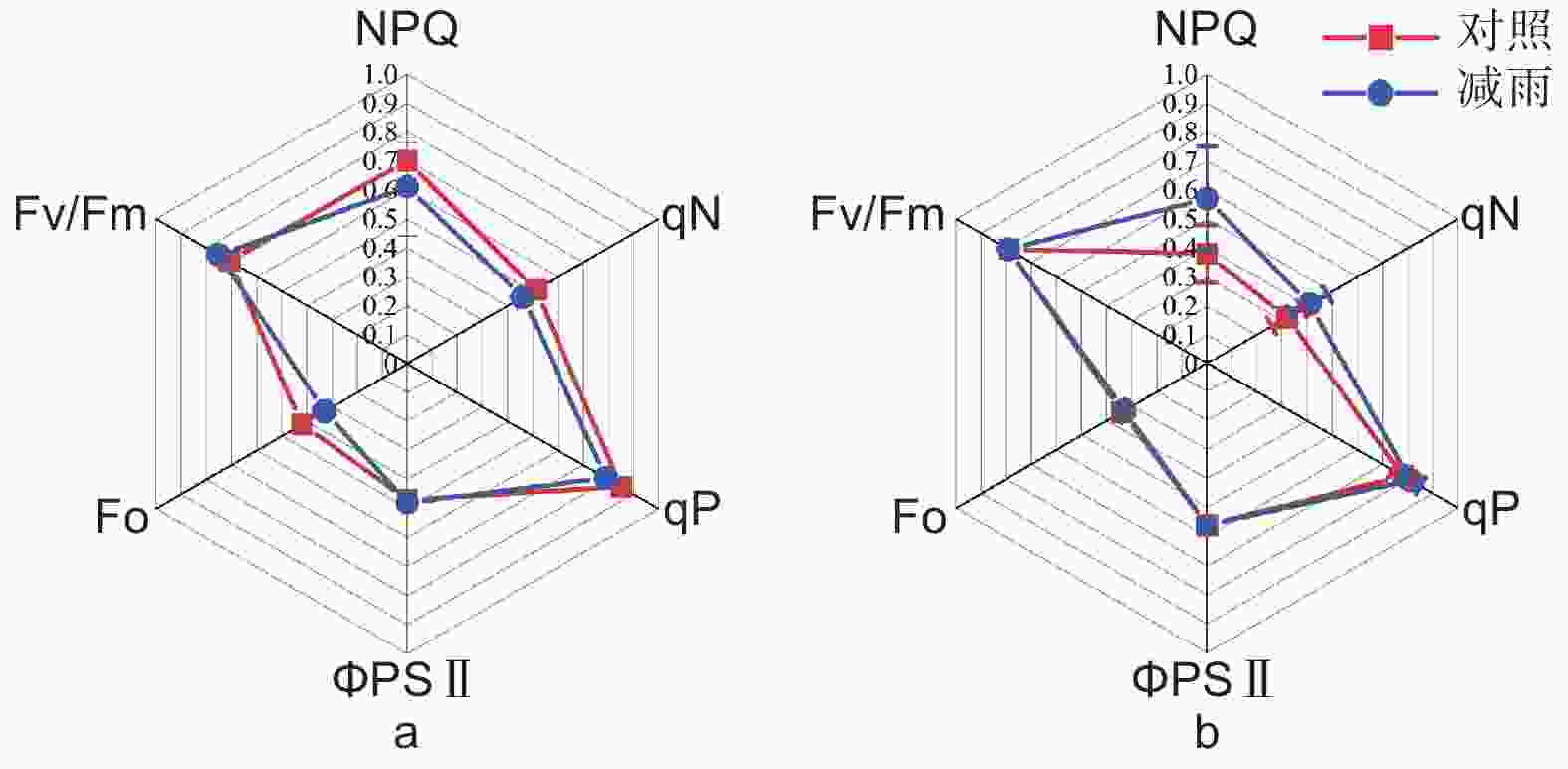
图 3 对照样地和减雨样地马尾松(a)和红锥(b)叶绿素荧光参数
Figure 3. Chlorophyll fluorescence parameters of Pinus massoniana (a) and Castanopsis hystrix (b) in the control and the rainfall-reduced plots
表 3 对照样地和减雨样地马尾松和红锥叶绿素荧光参数
Table 3. Chlorophyll fluorescence parameters of Pinus massoniana and Castanopsis hystrix in control and rainfall-reduced plots
叶绿素荧光参数
Chlorophyll fluorescence parameters马尾松 Pinus massoniana 红锥 Castanopsis hystrix 对照 Control 处理 Treatment 对照 Control 处理 Treatment 最大荧光 Fm 1.46 ± 0.08 a 1.31 ± 0.07 a 1.62 ± 0.13 a 1.58 ± 0.10 a 最大可变荧光 Fv 1.04 ± 0.06 a 0.98 ± 0.06 a 1.28 ± 0.11 a 1.25 ± 0.08 a 表观电子传递速率 ETR 28.91 ± 0.99 a 29.70 ± 1.39 a 34.48 ± 2.03 a 34.52 ± 1.28 a 注:不同的小写字母代表对照和减雨样地之间存在显著差异(p < 0.05)
Note: Lowercase letters indicate significant differences between the control plots and treatment plots (p < 0.05) -
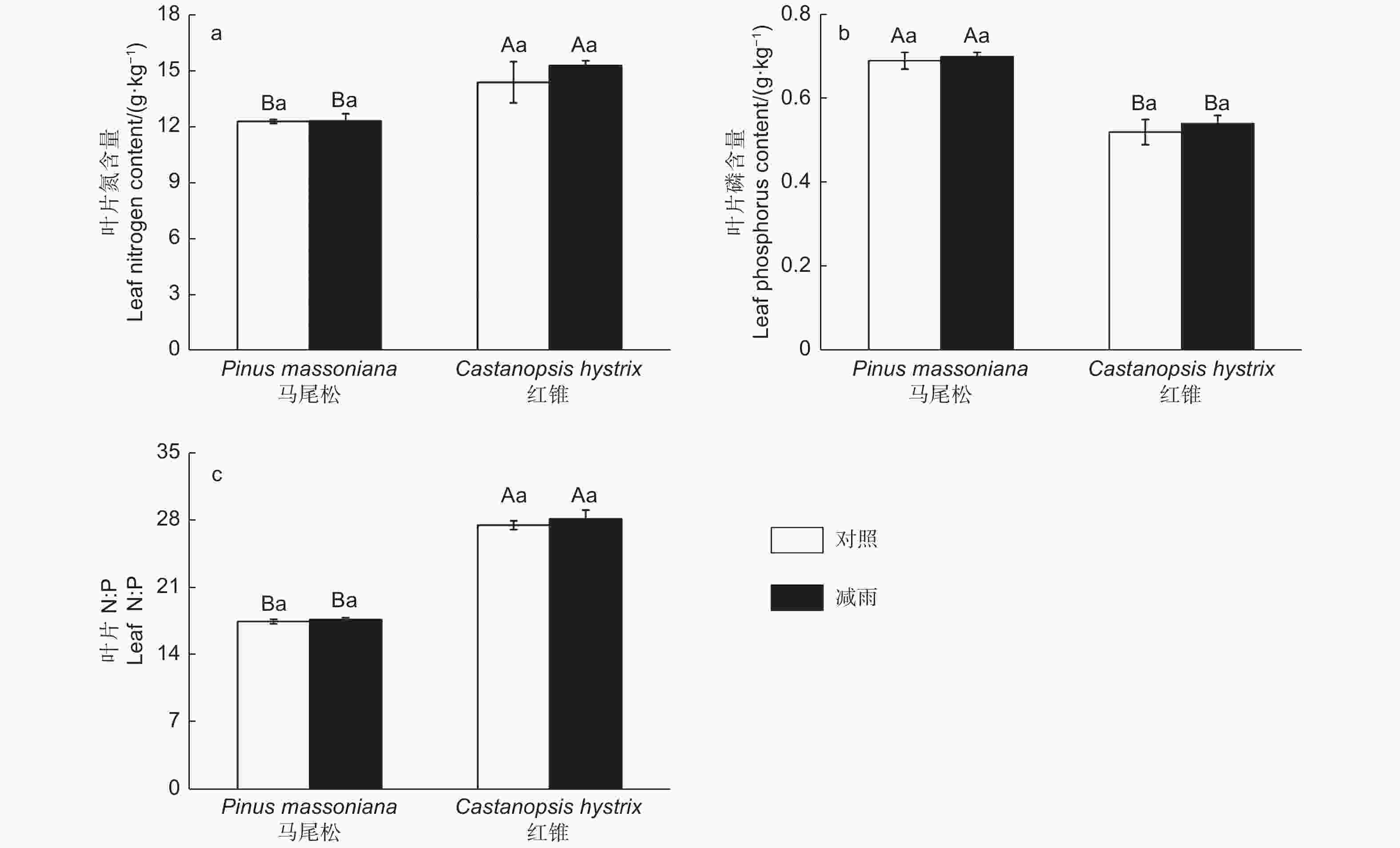
由图4得出:叶片氮、磷含量和氮磷比存在明显的种间差异。马尾松的叶片氮含量以及氮磷比显著低于红锥的叶片氮含量和氮磷比,而马尾松的叶片磷含量显著高于红锥的磷含量。穿透雨减少处理未对马尾松和红锥叶片N、P含量以及N:P产生影响(p > 0.05)。
-
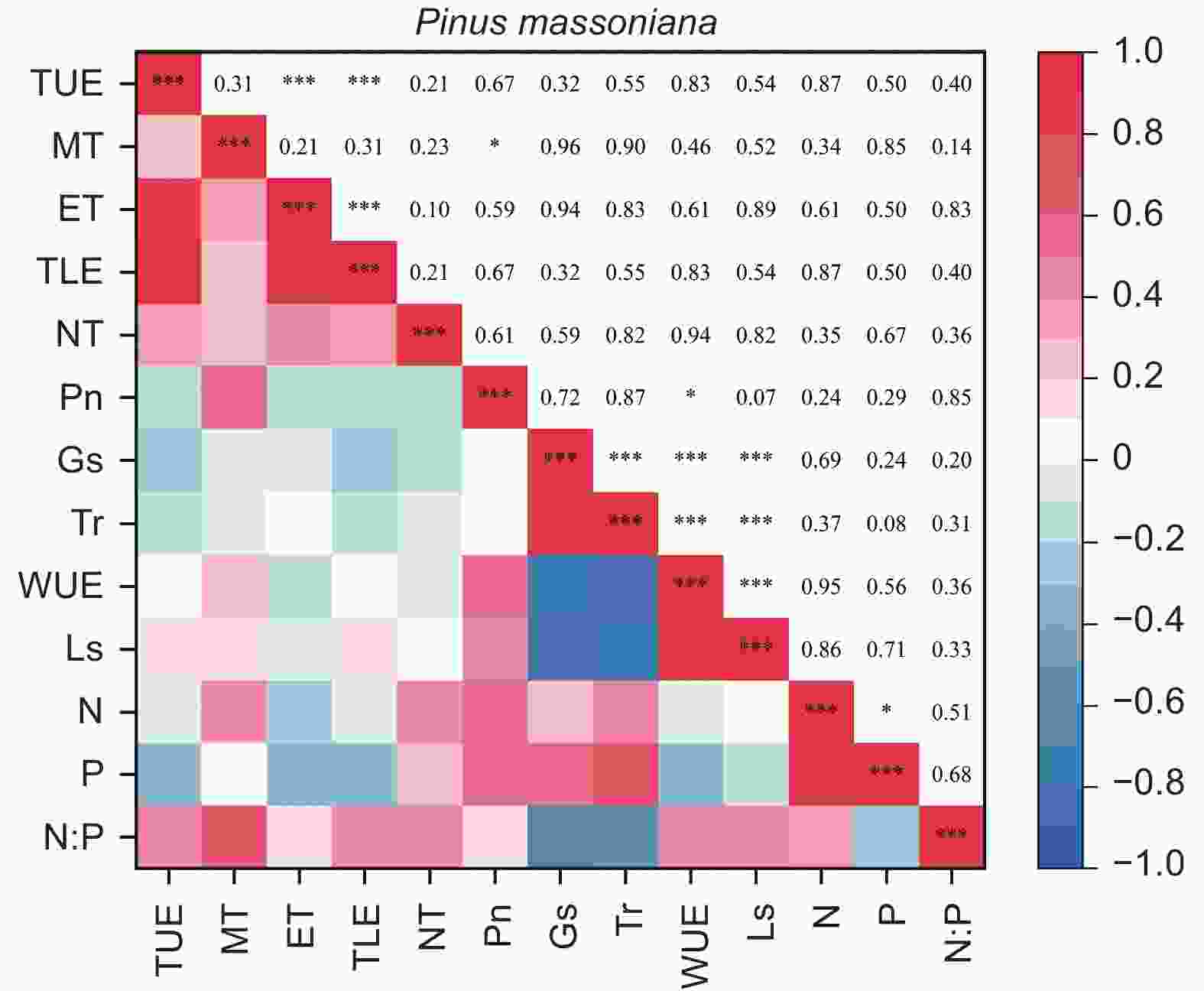
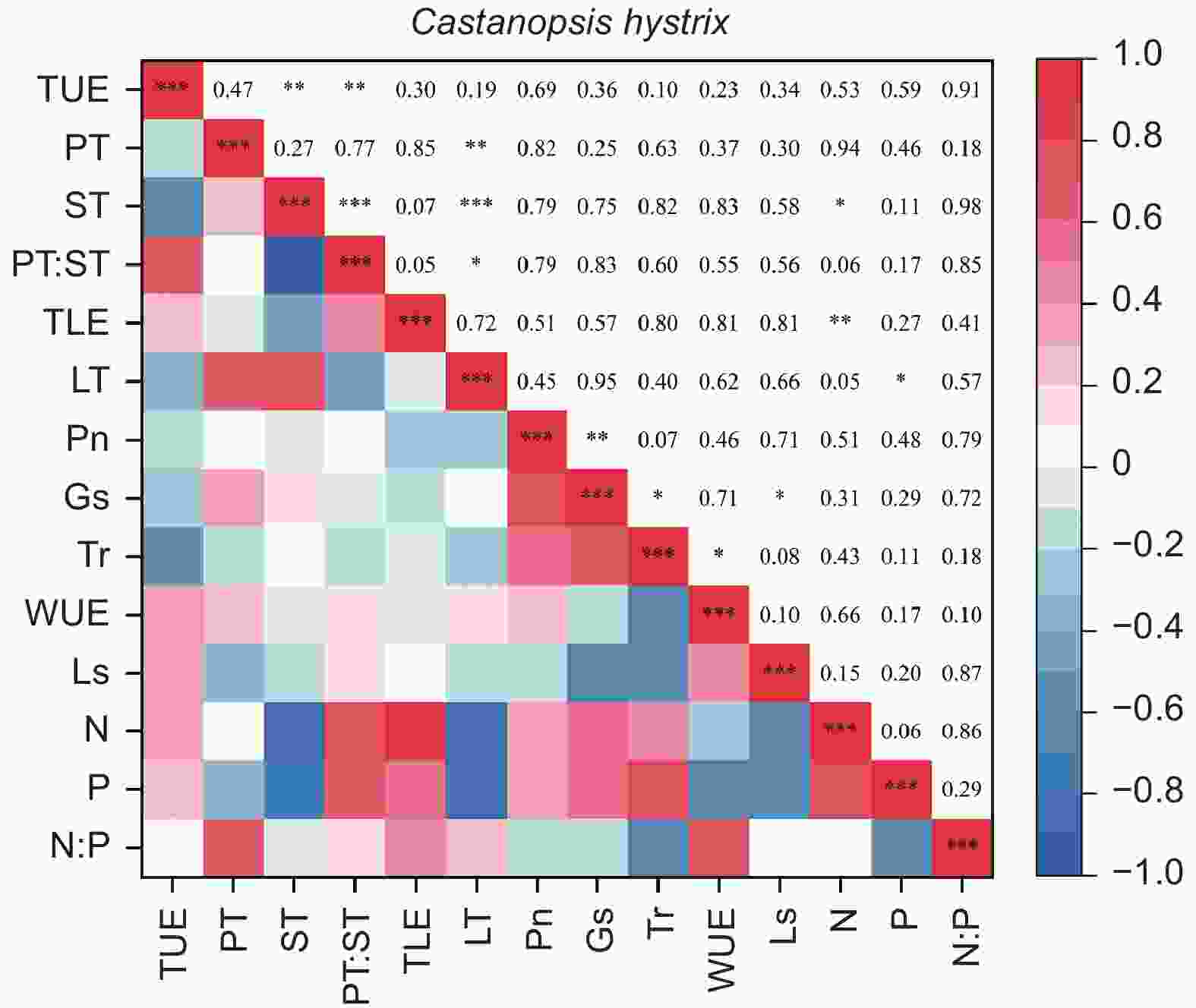
由图5、6得出:马尾松和红锥的叶片解剖结构之间、叶片氮和磷之间以及气体交换参数之间自身存在明显地相关关系(p < 0.05),但两树种的叶片解剖结构、叶片养分与气体交换参数三种叶片功能性状之间未发现相关关系(p > 0.05)。
-
穿透雨减少后马尾松和红锥叶片均表现出一定的干旱适应特征,但物种间差异明显。马尾松针叶的上、下表皮厚度和气孔密度在穿透雨减少后显著增加,而红锥在穿透雨减少后仅叶片的上表皮厚度出现明显增加。较厚的表皮能够使干旱条件下植物叶片维持较高的水势;气孔密度的增加有助于提高植物对于蒸腾控制[22-23],维持正常的生理代谢,从而有助于提高植物的抗旱性。Binks等[13]在对降雨减少条件下的热带雨林树种研究中也发现,降雨减少后叶片下表皮厚度增加明显,并提出下表皮的增加能够提高树种的抗旱性。然而,黄娟等[24]和吴丽君等[25]对干旱胁迫下银中杨和赤皮青冈研究发现,干旱胁迫使2树种叶片变薄,下上表皮厚度均出现显著下降,还认为干旱胁迫造成的叶片水分亏缺导致了叶片细胞生长和分裂受阻,从而导致叶片生长受限。本研究中,马尾松针叶厚度在穿透雨减少后增加明显,而红锥叶片厚度无显著变化。减雨后马尾松更厚的叶片往往能够使其拥有更高的水分储存能力,从而提高了马尾松的干旱适应能力[26]。红锥叶片在减雨后海绵组织厚度出现明显下降,海绵组织的减少促使叶肉细胞之间变得更为紧密,细胞间隙减小,该特征有助于CO2从气孔下室到光合作用位点的传导,抵消因叶肉结构变化和气孔关闭导致的CO2传导率的降低[27],能够维持正常的光合生理。另外,由于海绵组织所占比例减少使得叶片细胞之间更为紧密,提高了叶片的密度,能够减少叶片由于失水造成的物理损伤,有助于提高植物的水分利用效率,增强了植物干旱环境下的生存能力[28]。显然,树木叶片解剖对于干旱响应是复杂的,这可能归因于树种自身属性和干旱强度的差异[29]。为了探究树种对于干旱的响应和适应,加强叶片解剖结构的研究是非常必要。
-
植物在长期的进化过程中为适应环境变化,不仅在叶片形态和解剖结构上表现出可塑性,同时植物的生理也发生着改变。以往的研究中发现,轻微和中度的水分胁迫会通过减少大气到叶绿体羧化位点之间CO2的扩散来限制光合作用[30]。然而,在本研究中发现,穿透雨减少后马尾松和红锥叶片的光合参数(Pn、Gs、Tr、Ls)以及叶绿素荧光参数(Fv/Fm、ΦPSII、ETR)均未发生变化(p > 0.05)。Fv/Fm是表征光合单元结构健康状况的指标,穿透雨减少后未发生显著的改变说明穿透雨减少引起的水分胁迫并未对马尾松和红锥叶片的光合系统造成损伤,或经过8 a的穿透雨减少后2树种已近乎完全地适应了新的干旱环境,从而能够维持2树种正常的光合能力。以往的研究表明,干旱胁迫对光合作用的影响主要受叶片水势控制,轻微的水分胁迫不会影响植物的光合生理,只有当叶片水势下降到一定阈值后,光合生理才会受到影响[31-32]。在亚热带和热带开展的类似研究中也发现,叶片的光合作用速率在降雨减少后依然能够保持稳定,并认为是叶片解剖、水分关系以及光合特性之间紧密的相互协调的结果[13-14,33]。此外,通过对干旱条件下全球C3和C4植物气体交换参数的整合分析,证明了Gs对于干旱敏感性要高于Pn和Tr,并且发现Gs决定着光合作用速率和蒸腾速率[34-35]。以上研究结论可以用于解释本研究中处理间马尾松和红锥的Pn和Tr无差异的原因,可能是Gs在穿透雨减少后未发生改变。尽管马尾松和红锥叶片的光合生理在处理间无显著差异,但季节变化明显。与湿季相比,干季红锥的气孔限制和水分利用效率明显更高,说明红锥更倾向于通过调节气孔(Ls)来提高植物的水分利用效率(WUE),采取的保守型水分利用策略来适应干旱[36-37]。马尾松叶片气孔限制也存在明显季节差异,但水分利用效率(WUE)在湿季和干季之间几乎未发生改变,说明干旱条件下马尾松可能采取的是挥霍型水分利用策略[36-37]。本研究结果证明树种间光合生理对于干旱的差异响应除了与干旱胁迫强度有关外,也可能与物种所采取的水分利用策略有关。
-
结构是叶片功能的基础,叶片结构的变化往往影响着植物的光合、呼吸以及蒸腾等生理过程。季子敬等[38]对兴安落叶松针叶的研究中发现,叶片的最大光合速率和水分利用效率与叶片厚度、内皮层厚度以及传输组织厚度存在显著的正线性关系。在本研究中未发现马尾松和红锥解剖性状与气体交换参数之间存在显著关系。产生这种结果的主要原因可能是叶片生长和光合生理对于干旱的响应敏感性差异,造成了叶片解剖结构和光合生理之间解耦。以往的研究证实植物光合作用受到干旱胁迫影响之前植物的生长就已经停止[39],换言之,干旱条件下植物的光合生理和生长可能不同步[40]。通过对热带森林叶片的解剖和光合生理研究证明了改变的叶片解剖结构未对植物的光合能力产生显著影响[13-14]。对不同土壤条件下,苋菜(Amaranthus sp.)叶片的生长、气孔导度以及蒸腾对土壤干旱的响应研究中同样证实了叶片生长的土壤水分阈值要高于气孔导度和蒸腾的土壤水分阈值[31]。在5种多年生木本植物的日蒸腾速率对于土壤干旱的响应研究中,也证实了只有土壤水分下降到一定值时植物的蒸腾速率才会出现明显下降[41]。最近研究也发现,植物光合能力不能完全通过叶片的解剖性状来解释[13-14],从而推断出叶片解剖性状在调控光合作用过程中作用可能不如叶片的生物化学性状重要[42]。本研究中,叶片的氮含量以及叶片胞间CO2浓度在穿透雨减少后未发生显著改变在一定程度上支持了这一观点。穿透雨减少后,胞间CO2浓度未发生显著改变说明光合作用未受到生物化学限制[43];同样叶片氮含量未发生显著改变也表明了Rubisco含量或活性在处理间可能未发生显著变化[13,44],稳定的Rubisco含量或活性保证了叶片的光合能力稳定。因此,叶片解剖结构和光合生理之间关系是复杂多维的受多种因素影响,为了进一步加深干旱条件下叶片光合生理的调控机制的理解,在今后的研究中需对叶片解剖结构和叶片生理化学性状进行同步监测。
-
马尾松和红锥叶片解剖结构对穿透雨的减少表现出了一定的适应性,但物种间差异显著。穿透雨减少显著地改变了马尾松的针叶解剖结构,而对红锥叶片解剖结构影响有限。马尾松和红锥叶片光合生理在穿透雨减少后未发生改变,但存在明显的季节差异。马尾松和红锥气孔限制(Ls)和水分利用效率(WUE)的季节差异说明了红锥倾向于采用保守型水分利用策略,而马尾松采取挥霍型水分利用策略。马尾松针叶解剖结构在穿透雨减少表现出更明显的旱生叶片特征,这表明马尾松对于干旱胁迫的适应是通过调节针叶的解剖结构形成更具耐旱的针叶,而红锥倾向于调节气孔导度来适应干旱。另外,本研究中,叶片的光合生理与叶片解剖结构之间无相关性,说明了树木的光合生理除了受叶片解剖结构影响外也受其它因素影响(如Rubisco含量或活性等)。
马尾松和红锥叶片解剖和光合对穿透雨减少的响应
The Response of Leaf Anatomical Structure and Photosynthetic Physiology of Pinus massoniana Lamb. and Castanopsis hystrix Miq. to Throughfall Reduction
-
摘要:
目的 探究8 a干旱胁迫条件下树木叶片光合生理特征与叶片解剖结构的响应及二者之间的关系。 方法 在南亚热带选取2种典型人工林(马尾松和红锥)设置林内穿透雨减少的模拟干旱实验,在穿透雨减少8 a后测定叶片气体交换参数、叶绿素荧光参数、叶片解剖特征以及叶片养分(N、P)含量。 结果 (1)穿透雨减少后,马尾松针叶上、下表皮、叶片厚度以及气孔密度显著增加,而红锥仅叶片上表皮厚度显著增加,海绵组织厚度显著降低。(2)马尾松和红锥的气体交换参数(净光合速率Pn、气孔导度Gs、蒸腾速率Tr)、叶绿素荧光参数(光系统Ⅱ最大光化学效率Fv/Fm、实际光化学效率 ΦPSII,表观电子传递速率ETR)以及叶氮、磷含量在穿透雨减少8 a后均未发生显著变化。(3)与马尾松相比,红锥受到的气孔限制(Ls)更大,水分利用效率(WUE)更高,尤其在干季更明显。 结论 马尾松和红锥应对干旱胁迫的策略存在差异,马尾松通过调节叶片解剖结构产生更具耐旱的针叶来应对干旱,红锥则更倾向通过调节气孔(Ls)、提高水分利用效率(WUE)来适应干旱。 Abstract:Objective To explore the response of leaf anatomy and leaf photosynthetic physiology to long-term drought and analyze the relationship between leaf anatomy and leaf photosynthetic physiology under long-term drought conditions. Method We conducted a manipulation drought experiment by reducing the throughfall in the forest and measured the leaf gas exchange, chlorophyll fluorescence, leaf nutrient (N, P) and leaf anatomy of the two typical tree species (Pinus massoniana Lamb. and Castanopsis hystrix Miq.) in subtropical China at the 8th year after the throughfall reduction treatment. Results (1) Significant differences were observed in leaf anatomy between P. massoniana and C. hystrix in response to throughfall reduction; the upper and lower epidermis, leaf thickness and stomatal density of P. massoniana needles increased significantly. However, the thickness of the upper epidermis of the leaves of C. hystrix increased significantly, and the thickness of the spongy tissue decreased significantly. (2) No significant differences were observed in the gas exchange parameters (net photosynthetic rate Pn, stomatal conductance Gs, transpiration rate Tr), chlorophyll fluorescence parameters (photosystem II maximum photochemical efficiency Fv/Fm, actual photochemical efficiency ΦPSII, apparent electron transfer rate ETR) and leaf nutrient contents of both species after 8 years of throughfall reduction. (3) Compared with P. massoniana, the stomatal limitation (Ls) of the C. hystrix was larger and the water use efficiency (WUE) was higher, especially in the dry season. Conclusion There are differences in the strategies of P. massoniana and C. hystrix to cope with drought stress. P. massoniana responds to drought by adjusting leaf anatomy to produce xeromorphic needles, while C. hystrix is more inclined to adjust stomata (Ls) and improve water use efficiency (WUE) to adapt to drought. -
表 1 马尾松和红锥叶片解剖结构
Table 1. Leaf anatomical structure of Pinus massoniana and Castanopsis hystrix
树种
Tree species叶片指标
Index of leaf对照
Control处理
Treatment马尾松
Pinus massoniana上表皮厚度 Thickness of upper epidermis/μm 12.77 ± 0.21 b 16.39 ± 0.67 a 下表皮厚度 Thickness of lower epidermis /μm 21.41 ± 0.21 b 27.82 ± 0.75 a 叶肉厚度 Mesophyll thickness/μm 104.87 ± 8.91 a 99.79 ± 12.30 a 内皮层厚度 Endothelium thickness/μm 13.83 ± 0.43 b 15.00 ± 0.31 a 树脂道数 Resin canals number 11.33 ± 0.67 a 11.00 ± 0.65 a 针叶厚度 Needle thickness/μm 507.94 ± 3.71 b 549.22 ± 30.94 a 针叶宽 Needle width/μm 875.49 ± 22.11 a 854.03 ± 14.81 a 气孔密度 Stomata density/(number·mm−2) 119.96 ± 10.48 b 132.61 ± 10.21 a 红锥
Castanopsis hystrix上表皮厚度 Thickness of upper epidermis /μm 17.44 ± 0.37 b 21.7 ± 0.54 a 下表皮厚度 Thickness of lower epidermis /μm 11.86 ± 0.37 a 13.27 ± 0.66 a 栅栏组织 Palisade tissue thickness/μm 53.43 ± 1.78 a 53.64 ± 1.38 a 海绵组织 Spongy tissue thickness/μm 44.26 ± 1.5 a 33.68 ± 1.3 b 栅栏/海绵 Ratio of Palisade tissue and spongy tissue 1.22 ± 0.04 b 1.60± 0.04 a 叶片厚度 Leaf thickness/μm 140.10 ± 2.44 a 136.02 ± 2.02 a 气孔密度 Stomata density /(number·mm−2) 468.71 ± 16.68 a 489.58 ± 21.67 a 注:不同的小写字母代表对照和减雨样地之间存在显著差异(p < 0.05)
Note: Lowercase letters indicate significant differences between the control plots and treatment plots (p < 0.05)表 2 对照样地和减雨样地马尾松和红锥气体交换参数
Table 2. Gas exchange parameters of Pinus massoniana and Castanopsis hystrix in control and rainfall-reduced plots
树种
Tree species气体交换参数
Gas exchange parameters湿季 Wet season 干季 Dry season 对照 Control 处理 Treatment 对照 Control 处理 Treatment 马尾松
Pinus massonianaPn / (μmol·m−2·s−1) 4.17 ± 0.18 Aa 3.92 ± 0.20 Aa 2.10 ± 0.19 Ba 1.72 ± 0.03 Ba Gs / (mol·m−2·s−1) 0.063 ± 0.006 Aa 0.058 ± 0.004 Aa 0.025 ± 0.002 Ba 0.023 ± 0.003 Ba Ci / (μmol·m−2·s−1) 295.95 ± 12.71 Aa 289.42 ± 10.22 Aa 305.44 ± 8.03 Aa 308.22 ± 22.37 Aa Tr / (mmol·m−2·s−1) 0.89 ± 0.76 Aa 0.86 ± 0.06 Aa 0.48 ± 0.05 Ba 0.34 ± 0.064 Ba WUE 4.88 ± 0.47 Aa 4.67 ± 0.41 Aa 4.79 ± 0.52 Aa 6.08 ± 1.18 Aa Ls 0.29 ± 0.07 Ba 0.30 ± 0.02 Ba 0.33 ± 0.044 Aa 0.36 ± 0.065 Aa 红锥
Castanopsis hystrixPn / (μmol·m−2·s−1) 4.59 ± 0.38 Aa 4.42 ± 0.39 Aa 3.66 ± 0.51 Ba 3.24 ± 0.32 Ba Gs / (mol·m−2·s−1) 0.07 ± 0.01 Aa 0.06 ± 0.01 Aa 0.02 ± 0.002 Ba 0.017 ± 0.002 Ba Ci / (μmol·m−2·s−1) 289.10 ± 12.87 Aa 273.31 ± 11.09 Aa 253.24 ± 10.46 Aa 262.85 ± 13.63 Aa Tr / (mmol·m−2·s−1) 0.85 ± 0.09 Aa 0.67 ± 0.06 Aa 0.42 ± 0.02 Ba 0.35 ± 0.02 Ba WUE 5.79 ± 0.62 Ba 6.74 ± 0.70 Ba 8.65 ± 1.08 Aa 9.26 ± 0.66 Aa Ls 0.30 ± 0.03 Ba 0.33 ± 0.03 Ba 0.77 ± 0.02 Aa 0.80 ± 0.03 Aa 注:Pn:净光合速率;Gs:气孔导度;Tr:蒸腾速率;WUE:水分利用效率;Ls:气孔限制。不同的小写字母代表对照和减雨样地之间存在显著差异,不同的大写字母表示季节间存在显著差异(p < 0.05)
Notes: Pn: Net photosynthetic rate; Gs: Stomatal conductance; Tr : Transpiration rate; WUE: Water use efficiency; Ls: Stomatal limitation. Lowercase letters indicate significant differences between the control plots and treatment plots. Uppercase letters indicate significant differences among seasons (p < 0.05)表 3 对照样地和减雨样地马尾松和红锥叶绿素荧光参数
Table 3. Chlorophyll fluorescence parameters of Pinus massoniana and Castanopsis hystrix in control and rainfall-reduced plots
叶绿素荧光参数
Chlorophyll fluorescence parameters马尾松 Pinus massoniana 红锥 Castanopsis hystrix 对照 Control 处理 Treatment 对照 Control 处理 Treatment 最大荧光 Fm 1.46 ± 0.08 a 1.31 ± 0.07 a 1.62 ± 0.13 a 1.58 ± 0.10 a 最大可变荧光 Fv 1.04 ± 0.06 a 0.98 ± 0.06 a 1.28 ± 0.11 a 1.25 ± 0.08 a 表观电子传递速率 ETR 28.91 ± 0.99 a 29.70 ± 1.39 a 34.48 ± 2.03 a 34.52 ± 1.28 a 注:不同的小写字母代表对照和减雨样地之间存在显著差异(p < 0.05)
Note: Lowercase letters indicate significant differences between the control plots and treatment plots (p < 0.05) -
[1] Dai A G. Hydroclimatic trends during 1950–2018 over global land[J]. Climate Dynamics, 2021, 56(9): 4027-4049. [2] Zhou G Y, Wei X H, Wu Y P, et al. Quantifying the hydrological responses to climate change in an intact forested small watershed in Southern China[J]. Global Change Biology, 2011, 17(12): 3736-3746. doi: 10.1111/j.1365-2486.2011.02499.x [3] Zhou G Y, Peng C H, Li Y L, et al. A climate change-induced threat to the ecological resilience of a subtropical monsoon evergreen broad-leaved forest in Southern China[J]. Global Change Biology, 2013, 19(4): 1197-1210. doi: 10.1111/gcb.12128 [4] Allen C D, Macalady A K, Chenchouni H, et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests[J]. Forest ecology and management, 2010, 259(4): 660-684. doi: 10.1016/j.foreco.2009.09.001 [5] Poorter L, Bongers F. Leaf traits are good predictors of plant performance across 53 rain forest species[J]. Ecology, 2006, 87(7): 1733-1743. doi: 10.1890/0012-9658(2006)87[1733:LTAGPO]2.0.CO;2 [6] De Carcer P S, Signarbieux C, Schlaepfer R, et al. Responses of antinomic foliar traits to experimental climate forcing in beech and spruce saplings[J]. Environmental and Experimental Botany, 2017, 140: 128-140. doi: 10.1016/j.envexpbot.2017.05.013 [7] 李 周, 赵雅洁, 宋海燕, 等. 不同水分处理下喀斯特土层厚度异质性对两种草本叶片解剖结构和光合特性的影响[J]. 生态学报, 2018, 38(2):721-732. [8] Toscano S, Ferrante A, Tribulato A, et al. Leaf physiological and anatomical responses of Lantana and Ligustrum species under different water availability[J]. Plant Physiology and Biochemistry, 2018, 127: 380-392. doi: 10.1016/j.plaphy.2018.04.008 [9] 朱教君, 康宏樟, 李智辉, 等. 水分胁迫对不同年龄沙地樟子松幼苗存活与光合特性影响[J]. 生态学报, 2005, 25(10):2527-2533. doi: 10.3321/j.issn:1000-0933.2005.10.010 [10] Bacelar E A, Santos D L, Moutinho-Pereira J M, et al. Immediate responses and adaptative strategies of three olive cultivars under contrasting water availability regimes: changes on structure and chemical composition of foliage and oxidative damage[J]. Plant Science, 2006, 170(3): 596-605. doi: 10.1016/j.plantsci.2005.10.014 [11] Toscano S, Ferrante A, Romano D. Response of Mediterranean ornamental plants to drought stress[J]. Horticulturae, 2019, 5(1): 6. doi: 10.3390/horticulturae5010006 [12] Gratani L, Catoni R, Varone L. Morphological, anatomical and physiological leaf traits of Q. ilex, P. latifolia, P. lentiscus, and M. communis and their response to Mediterranean climate stress factors[J]. Botanical Studies, 2013, 54(1): 1-12. doi: 10.1186/1999-3110-54-1 [13] Binks O, Meir P, Rowland L, et al. Limited acclimation in leaf anatomy to experimental drought in tropical rainforest trees[J]. Tree Physiology, 2016, 36(12): 1550-1561. doi: 10.1093/treephys/tpw078 [14] Rowland L, Lobo‐do‐Vale R L, Christoffersen B O, et al. After more than a decade of soil moisture deficit, tropical rainforest trees maintain photosynthetic capacity, despite increased leaf respiration[J]. Global Change Biology, 2015, 21(12): 4662-4672. doi: 10.1111/gcb.13035 [15] 杨予静, 刘世荣, 陈 琳, 等. 马尾松人工林土壤和团聚体有机碳矿化对穿透雨减少的响应[J]. 应用生态学报, 2018, 29(6):1779-1786. [16] 王仁杰, 蒋 燚, 王 勇, 等. 南亚热带不同红锥混交林土壤碳库稳定性与碳库管理指数变化[J]. 林业科学研究, 2021, 34(2):24-31. [17] Carnicer J, Barbeta A, Sperlich D, et al. Contrasting trait syndromes in angiosperms and conifers are associated with different responses of tree growth to temperature on a large scale[J]. Frontiers in Plant Science, 2013, 4: 409. [18] Zhao X, Zhao P, Zhu L, et al. Exploring the influence of biological traits and environmental drivers on water use variations across contrasting forests[J]. Forests, 2021, 12(2): 161. doi: 10.3390/f12020161 [19] 陈 琳, 刘世荣, 温远光, 等. 南亚热带红锥和马尾松人工林生长对穿透雨减少的响应[J]. 应用生态学报, 2018, 29(7):2330-2338. [20] 唐 艳, 王传宽. 东北主要树种光合作用可行的离体测定方法[J]. 植物生态学报, 2011, 35(4):452-462. [21] 陈志成, 陆海波, 刘世荣, 等. 锐齿栎水力结构和生长对降雨减少的响应[J]. 生态学报, 2018, 38(7):2405-2413. [22] Bosabalidis A M, Kofidis G. Comparative effects of drought stress on leaf anatomy of two olive cultivars[J]. Plant science, 2002, 163(2): 375-379. doi: 10.1016/S0168-9452(02)00135-8 [23] 胡潇予, 于海燕, 崔艺凡, 等. 不同种源文冠果叶片气孔分布特征对水分胁迫的响应[J]. 林业科学研究, 2019, 32(1):169-174. [24] 黄 绢, 陈 存, 张伟溪, 等. 干旱胁迫对转JERF36银中杨苗木叶片解剖结构及光合特性的影响[J]. 林业科学, 2017, 53(5):8-15. doi: 10.11707/j.1001-7488.20170502 [25] 吴丽君, 李志辉, 杨模华, 等. 赤皮青冈幼苗叶片解剖结构对干旱胁迫的响应[J]. 应用生态学报, 2015, 26(12):3619-3626. [26] Velikova V, Arena C, Izzo L G, et al. Functional and structural leaf plasticity determine photosynthetic performances during drought stress and recovery in two Platanus orientalis populations from contrasting habitats[J]. International journal of molecular sciences, 2020, 21(11): 3912. doi: 10.3390/ijms21113912 [27] 李芳兰, 包维楷. 植物叶片形态解剖结构对环境变化的响应与适应[J]. 植物学通报, 2005, 22(S1):118-127. [28] Guerfel M, Baccouri O, Boujnah D, et al. Impacts of water stress on gas exchange, water relations, chlorophyll content and leaf structure in the two main Tunisian olive (Olea europaea L. ) cultivars[J]. Scientia Horticulturae, 2009, 119(3): 257-263. doi: 10.1016/j.scienta.2008.08.006 [29] Wu H, Fan Y, Yu F, et al. Leaf anatomical plasticity of Phyllostachys glauca McClure in limestone mountains was associated with both soil water and soil nutrients[J]. Forests, 2022, 13(4): 493. doi: 10.3390/f13040493 [30] Pinheiro C, Chaves M M. Photosynthesis and drought: can we make metabolic connections from available data?[J]. Journal of experimental botany, 2011, 62(3): 869-882. doi: 10.1093/jxb/erq340 [31] Sadras V O, Milroy S P. Soil-water thresholds for the responses of leaf expansion and gas exchange: A review[J]. Field Crops Research, 1996, 47(2-3): 253-266. doi: 10.1016/0378-4290(96)00014-7 [32] Liu F, Stutzel H. Leaf expansion, stomatal conductance, and transpiration of vegetable amaranth (Amaranthus sp. ) in response to soil drying[J]. Journal of the American Society for Horticultural Science, 2002, 127(5): 878-883. doi: 10.21273/JASHS.127.5.878 [33] Zhang P, Zhou X, Fu Y, et al. Differential effects of drought on nonstructural carbohydrate storage in seedlings and mature trees of four species in a subtropical forest[J]. Forest Ecology and Management, 2020, 469: 118159. doi: 10.1016/j.foreco.2020.118159 [34] Yan W, Zhong Y, Shang G. A meta-analysis of leaf gas exchange and water status responses to drought[J]. Scientific Reports, 2016, 6(1): 1-9. doi: 10.1038/s41598-016-0001-8 [35] Zhang J, Jiang H, Song X, et al. The responses of plant leaf CO2/H2O exchange and water use efficiency to drought: A meta-analysis[J]. Sustainability, 2018, 10(2): 551. doi: 10.3390/su10020551 [36] Passioura J B. Water in the soil-plant-atmosphere continuum[M]//Physiological Plant Ecology II. Berlin: Springer, Heidelberg, 1982: 5-33. [37] 曹生奎, 冯 起, 司建华, 等. 植物叶片水分利用效率研究综述[J]. 生态学报, 2009, 29(7):3882-3892. doi: 10.3321/j.issn:1000-0933.2009.07.051 [38] 季子敬, 全先奎, 王传宽. 兴安落叶松针叶解剖结构变化及其光合能力对气候变化的适应性[J]. 生态学报, 2013, 33(21):6967-6974. [39] Woodruff D R, Meinzer F C. Water stress, shoot growth and storage of non-structural carbohydrates along a tree height gradient in a tall conifer[J]. Plant, Cell & Environment, 2011, 34(11): 1920-1930. [40] Hernandez‐Santana V, Perez‐Arcoiza A, Gomez‐Jimenez M C, et al. Disentangling the link between leaf photosynthesis and turgor in fruit growth[J]. The Plant Journal, 2021, 107(6): 1788-1801. doi: 10.1111/tpj.15418 [41] Sinclair T R, Holbrook N M, Zwieniecki M A. Daily transpiration rates of woody species on drying soil[J]. Tree Physiology, 2005, 25(11): 1469-1472. doi: 10.1093/treephys/25.11.1469 [42] Xiong D, Flexas J. Leaf anatomical characteristics are less important than leaf biochemical properties in determining photosynthesis responses to nitrogen top-dressing[J]. Journal of Experimental Botany, 2021, 72(15): 5709-5720. doi: 10.1093/jxb/erab230 [43] 庞 杰, 张凤兰, 郝丽珍, 等. 沙芥幼苗叶片解剖结构和光合作用对干旱胁迫的响应[J]. 生态环境学报, 2013, 22(4):575-581. doi: 10.3969/j.issn.1674-5906.2013.04.005 [44] 唐星林, 曹永慧, 顾连宏, 等. 基于FvCB模型的叶片光合生理对环境因子的响应研究进展[J]. 生态学报, 2017, 37(19):6633-6645. -




 下载:
下载: