-
“觅食”是生物生存与繁衍的基础,是生物间食物链的关键一环,也是生物学及进化生态学的核心内容[1-2]。而植物挥发物(VOCs)在寄主植物-植食性昆虫-天敌三级营养链连接中发挥着至关重要的作用,是植食性昆虫及天敌昆虫搜寻和定位寄主的重要信号物质[3-6]。据统计,在植食性昆虫与寄主互作研究中,超98%的报道是以地面昆虫(多以成虫为主)为对象,对于地下昆虫的关注较少[7-8]。相对于地面昆虫而言,在黑暗(视觉刺激缺失)、复杂的土壤环境中昆虫如何定位到寄主植物知之不多[9-11]。自Doane等[12]首次证实马铃薯叩甲(Ctenicera destructor (Brown))幼虫依靠植物根系释放的CO2定位寄主以来,胡萝卜根实蝇(Psila rosae F.)、玉米根萤叶甲(Diabrotica virgifera virgifera Leconte)及大栗鳃金龟(Melolontha hippocastani Fabricius)幼虫等多种地下昆虫被证实对CO2有显著的正趋向性[9,13],CO2一度被认为是地下植食性昆虫定位寄主的唯一信号物质[14-15]。
随着研究的深入,发现相比于CO2,根部释放的挥发物对土壤植食性昆虫定位寄主更有效[11,16-17],如大麦(Hordeum vulgare L.)根系释放的hexanal、(E)-hex-2-enal及(E)-non-2-enal等4种挥发物能显著激发金针虫(鞘翅目(Coleoptera)叩甲科(Elateridae)昆虫幼虫的统称)的定向移动[11]。植物根系挥发物在地下植食性昆虫寄主搜寻过程中发挥着重要作用,但研究报道并不多。筛胸梳爪叩甲(Melanotus cribricollis (Faldermann))是我国南方竹林金针虫的优势种[18],主要蛀食雷竹(Phyllostachys violascens (Carriere) Riviere & C. Riviere)、毛竹(Ph. edulis (Carriere) J. Houzeau)及乌哺鸡竹(Ph. vivaxs McClure)等多种笋用竹种,造成鲜笋腐烂和退笋,部分林分鲜笋受害率超70%,留养的种笋受害率高达100%,严重影响竹笋的产量和质量,威胁竹笋产业的健康发展[19]。前期研究表明,与普通小麦(Triticum aestivum L.)、玉米(Zea mays L.)等禾本科(Poaceae)作物相比,筛胸梳爪叩甲幼虫偏好取食雷竹笋,竹笋特有的挥发物可能发挥了重要作用[20],但竹笋挥发物的组成及与CO2的引诱活性差异尚未明确。本研究监测比较雷竹笋挥发物和CO2释放剂对筛胸梳爪叩甲幼虫的引诱作用,并鉴定了竹笋挥发物组分,旨在探索竹林金针虫的寄主定位及识别机制,为竹林金针虫的高效监测及防控提供科学依据。
-
2020年3—4月,在浙江省德清县雷竹种植区(120°04′12″ E,30°32′08″ N)采集筛胸梳爪叩甲幼虫,带回至中国林科院亚热带林业研究所恒温养虫室内饲养(养虫室温度20 ± 1 ℃,相对湿度60% ± 10%)。饲养时,将采集的金针虫用无菌水冲洗数次后,依据个体大小分装于养虫箱中(42 cm × 28 cm × 22 cm),单箱饲养50头,养虫箱中的无菌土湿度控制在10%±1%,于黑暗条件下饲养,以鲜笋喂食(定期更换)。试验时选取个体大小相近、龄期一致、健康活跃的幼虫个体供试[21]。
本研究所选用的竹笋为雷竹笋,均采自浙江省杭州市富阳区永昌镇雷竹种植区。试验当天,在雷竹林挖掘新鲜健康竹笋(笋尖出土<2 cm)1000 g,保鲜带回实验室供试。CO2释放剂颗粒由加拿大农业与食品研究中心Todd Kabaluk教授赠送,主要成分为酵母(Saccharomyces cerevisiae Reess ex Hansen)、小麦淀粉和藻酸钙[22]。
-
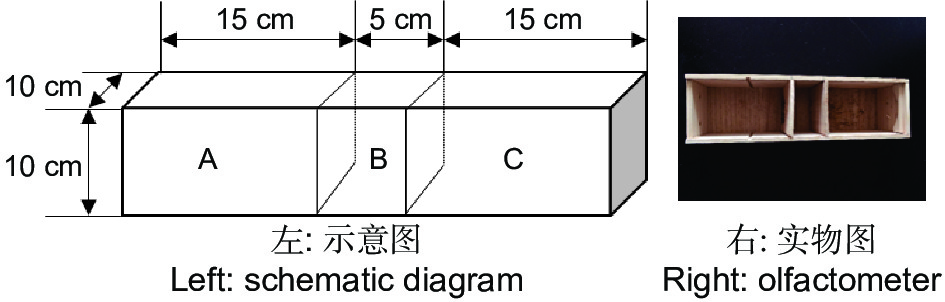
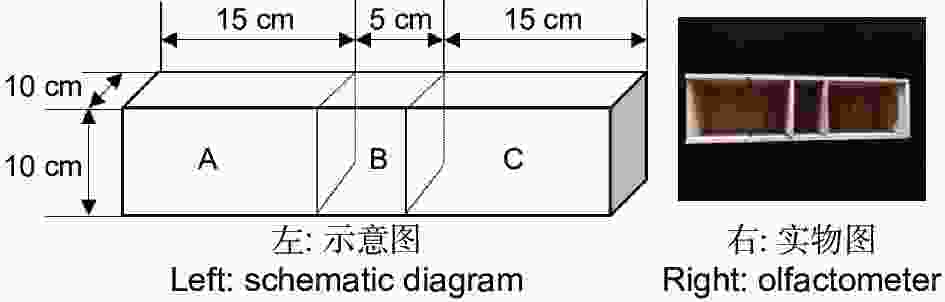
双向选择仪用实木材料(杉木)制成,由中心虫室B(10 cm × 5 cm × 10 cm)和左右两侧2个大小相同的选择室A和C(15 cm × 10 cm × 10 cm)所构成(图1)。中心虫室两侧隔板(10 cm × 10 cm × 0.1 cm)能手动拔除,使虫室和选择室连通,金针虫可自由移动。实验结束时再插入隔板,使中心虫室和选择室分离,便于统计不同选择端的金针虫数量。中心虫室用于放置供试金针虫,左右两侧选择室用于放置竹笋或CO2释放剂颗粒。筛胸梳爪叩甲幼虫在双向选择仪中可自由移动,空白试验(两侧均为无菌土)及同食物诱饵(等量竹笋)预实验测定结果表明,自制双向选择仪对金针虫的选择行为无显著影响(GP=0.06,P>0.05;GH=17.71,P=0.54,G检验,其中GP指每个处理组合中所有重复选择虫数总量的G值;GH指重复之间异质性G值,下同)。
-
将1.75 kg含水量为10%的无菌土加入到双向选择仪内,水平震荡木盒,使盒内土面平整,各部分土壤质地均匀一致,插入分隔板,备用。将新鲜雷竹笋(林地土壤以下的部分)的中间部分切成若干段,分别称取10、20和40 g,作为3个处理,随机埋入双向选择仪的任意一侧选择室的中心,另一侧选择室作空白对照(不放置竹笋)。同时,在中心虫室的无菌土中放入10头金针虫(饥饿24 h,下同),并取出分隔板。24 h后,插入分隔板。首先取出中心虫室的无菌土,在托盘内仔细分离统计其内金针虫的数量(标记为“未作出选择”),然后依次统计两侧选择室内金针虫的数量。
上述试验均在黑暗条件下进行(室内温度25±1 ℃,相对湿度80%±10%),每个处理共计20个重复。
-
取CO2释放剂颗粒10粒(2 g)、20粒(4 g)和40粒(8 g),随机埋入双向选择仪的一侧选择室的中心,另一侧作为对照。同时,将10头金针虫在中心虫室的无菌土内释放,并取出隔板。24 h后统计各区域金针虫的数量。
上述试验均在黑暗条件下进行(室内温度25±1 ℃,相对湿度80%±10%),每个处理共计20个重复。
-
取30 g雷竹笋和20粒CO2释放剂颗粒,分别埋入双向选择仪的左右两侧选择室的中心。同时,在中心虫室内释放10头金针虫。设置24、48 h两个处理,于24、48 h后统计各区域金针虫数量。
上述试验均在黑暗条件下进行(室内温度25±1 ℃,相对湿度80%±10%),每个处理共计20个重复。
-
利用固相微萃法提取挥发物。称取雷竹笋50 g,放入洁净的150 mL锥形瓶内(锥形瓶使用前用95%酒精清洗,并置于烘箱中120 ℃烘120 min),用Parafilm封口,静置于实验台上,平衡30 min。将活化的SPME萃取装置(美国Supelco公司生产,100 μm PDMS萃取头)插入锥形瓶内,推出萃取头,使其固定在竹笋上方5 cm处,萃取时间为60 min。萃取结束后,立即将萃取头插入GC-MS进样口(Agilent HP 6890GC/5975B)进行解析。实验于暗室中进行(温度25±1 ℃,相对湿度60%~70%),每组处理3个重复。
-
色谱柱:HP-5MS毛细管柱(极性,柱长30 m,内径0.32 mm,膜厚25 μm);不分流进样;溶剂延迟3 min;进样口温度250 ℃;柱温升温程序为:起始温度40 ℃,维持2 min,14 ℃·min−1升至180 ℃,再以25 ℃·min−1升至260 ℃,维持7 min。以99.999%的高纯氦气作为载气,流量1.0 mL·min−1[17]。质谱条件:电离方式EI,电离电压70 eV,离子源温度230 ℃,电子倍增器电压1340 V,扫描质量范围50~550 aum。
-
根据气-质联用仪的分析结果,比对谱库(NIST 2015)中标准化合物的质谱峰,对不同组分进行初步的物质鉴定;然后购买标准品,用二氯甲烷稀释成一定浓度的溶液,按照与样品相同的升温程序和质谱条件,在气-质联用仪中进样1 μL,将标准品的保留时间、质谱图和结构与样品中相应峰的保留时间、质谱图及结构进行比对,从而确定其挥发物组分。挥发物各组分的定量分析,应用峰面积百分比法(某物质峰面积/总物质峰面积 × 100%)计算各成分的相对含量。
-
选择性试验中,对于金针虫在不同组合和处理间(包括空白对照)的选择偏好均采用重复适合性(Repeated Goodness-of-fit)G检验。不同质量竹笋间以及不同浓度CO2间的诱虫差异分析均采用R × C列联表卡方检验(Pearson卡方检验)。利用DPS数据处理系统V19.05(杭州睿丰信息技术有限公司)进行数据分析,采用Excel 2019、PowerPoint 2019作图。
-
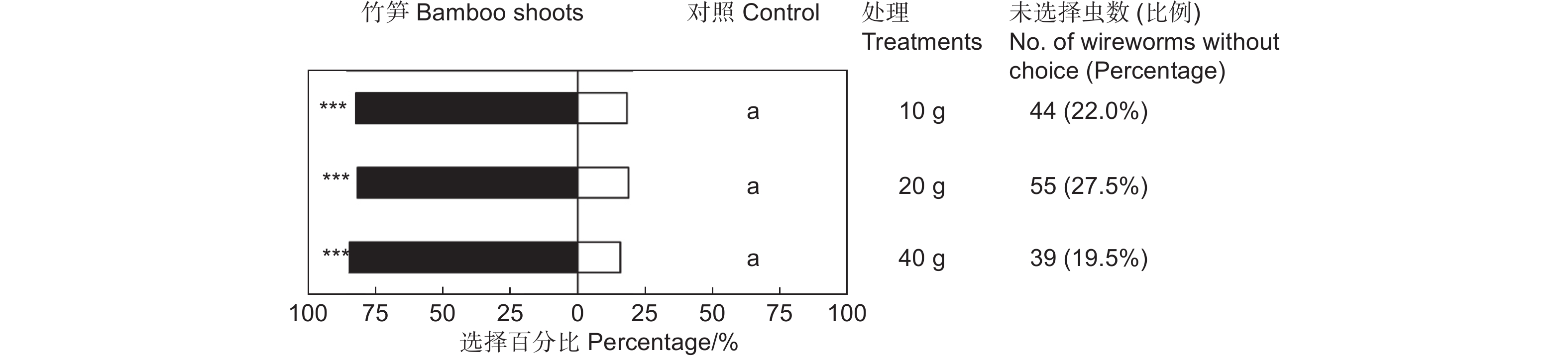
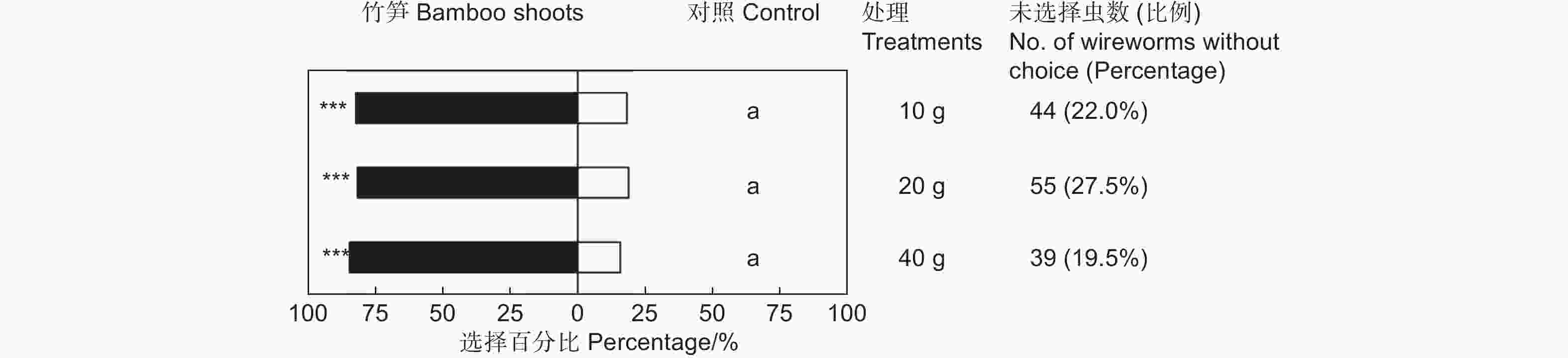
鲜笋诱集试验表明,雷竹鲜笋对筛胸梳爪叩甲幼虫表现出显著的吸引作用(表1),但不同质量的竹笋间差异不显著(χ2=0.576,df=2,P>0.05;图2)。释放金针虫24 h后,大多数金针虫选择在埋有竹笋的一端,聚集在竹笋周围或钻入竹笋内蛀食。供试的200头金针虫,10 g竹笋端有128头,占供试金针虫的64.00%(GP=69.43,P<0.001;GH=21.10,P=0.331;表1);20 g竹笋端有118头,占供试金针虫的59.00%(GP=61.62,P<0.001;GH=23.64,P=0.210;表1);40 g竹笋端有136头,占供试金针虫的68.00%(GP=84.17,P<0.001;GH=8.74,P=0.977;表1)。释放金针虫24 h后,不同处理均有20%左右的金针虫仍停留在中心虫室,未作出选择的金针虫在不同质量鲜笋处理中无显著差异(图2)。
处理
Treatments重复
Replicates作出选择的金针虫数量(百分比)
Number of wireworms at different sides(Percentage)G检验
G test处理端
Treated side对照端
Control sideGP GH 10 g 20 128(82.05%) 28(17.95%) 69.43*** 21.10NS 20 g 20 118(81.38%) 27(18.62%) 61.62*** 23.64NS 40 g 20 136(84.47%) 25(15.53%) 84.17*** 8.74NS 注:仅对作出选择的金针虫个体数进行G检验。***表示P<0.001,NS表示差异不显著(P>0.05)。下同。
Note:G test was performed only on the number of wireworms at the treated and control sides. *** means P<0.001,NS means that the difference is not significant(P>0.05). The same followed.Table 1. Choice tests of M. cribricollis larvae towards different weight bamboo shoots in a dual-choice olfactometer
-
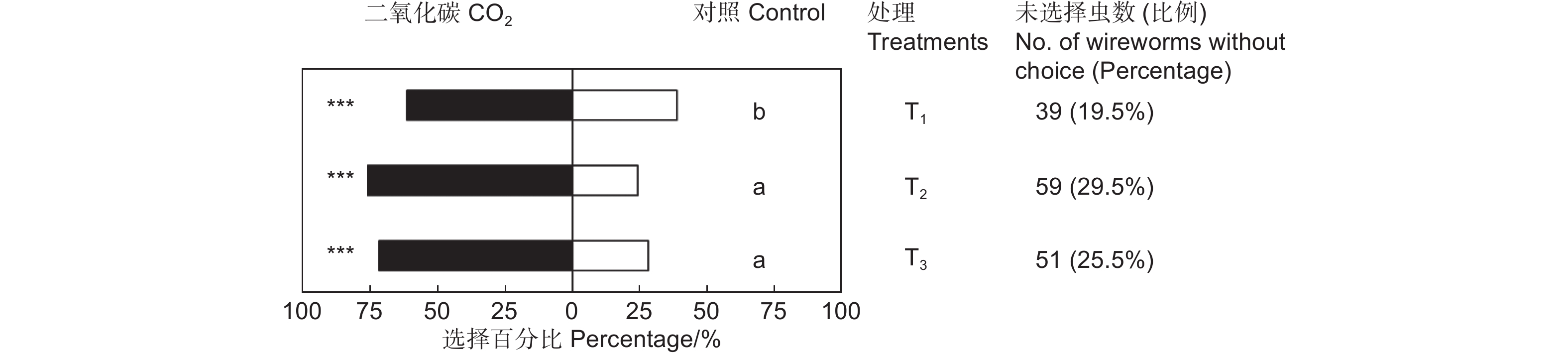
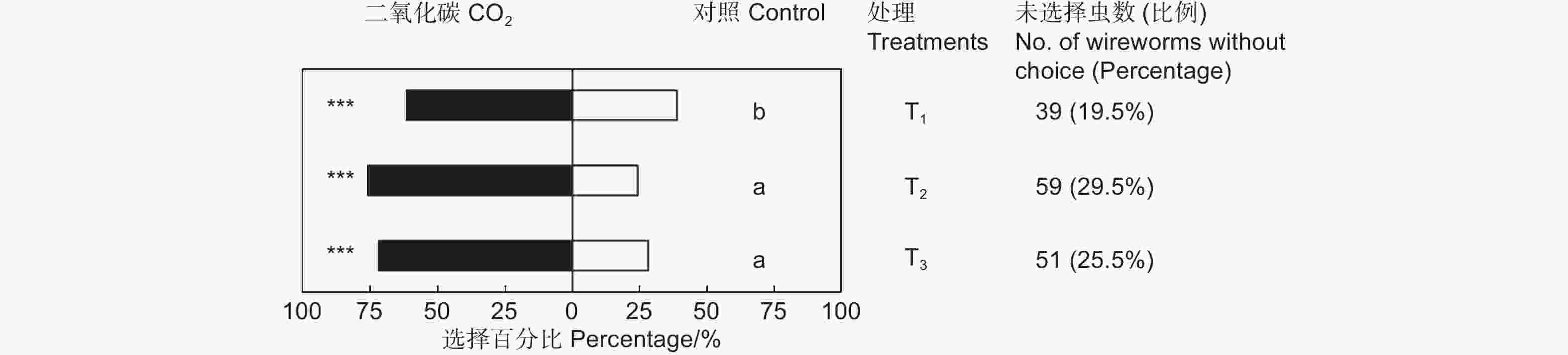
CO2对筛胸梳爪叩甲幼虫有显著的吸引作用,且不同浓度的CO2对金针虫引诱力差异显著(χ2=7.945,df=2,P<0.05;表2,图3)。放入10粒CO2释放剂颗粒时,供试的200头金针虫中161头作出了选择,CO2端有99头,占供试金针虫的49.50%(GP=8.58,P<0.01;GH=9.21,P=0.970);放入20粒CO2释放剂颗粒时,作出选择的有141头,CO2端有107头,占供试金针虫的53.50%(GP=36.70,P<0.001;GH=22.22,P=0.274);放入40粒CO2释放剂颗粒时,作出选择的有149头,CO2端有107头,占供试金针虫的53.50%(GP=29.33,P<0.001;GH=11.82,P=0.893)。20粒CO2释放颗粒剂对金针虫的引诱力最强,与10粒CO2释放颗粒剂相比有显著差异,但与40粒CO2释放颗粒剂间差异不显著(图3)。
处理
Treatments重复
Replicates作出选择的金针虫数量(百分比)
Number of wireworms at different sides(Percentage)G检验
G-test处理端
Treated side对照端
Control sideGP GH T1 20 99(61.49%) 62(38.50%) 8.58** 9.21NS T2 20 107(75.89%) 34(24.11%) 36.70*** 22.22NS T3 20 107(71.81%) 42(28.19%) 29.33*** 11.82NS 注:T1、T2、T3分别表示10、20和40粒CO2释放剂颗粒,下同。**表示P<0.01。
Note: T1, T2 and T3 presents 10, 20 and 40 granules of CO2 resource, respectively. The same followed. ** means P<0.01.Table 2. Choice tests of M. cribricollis larvae towards CO2 of different concentrations in a dual-choice olfactometer
-
在竹笋和CO2释放剂颗粒之间,筛胸梳爪叩甲幼虫显著偏好竹笋,竹笋的吸引力强于CO2,但不同的时间处理(24 h和48 h)间差异不显著(χ2=0.314,df=1,P>0.05)。供试的200头金针虫中,24 h后竹笋端有139头(占供试金针虫的69.5%),CO2端有34头(GP=68.37,P<0.001;GH=15.40,P=0.697;表3),而48 h后,竹笋端有142头(占供试金针虫的71.0%),CO2端有35头(GP =47.69,P<0.001;GH=15.40,P=0.749;表3)。
处理
Treatments重复
Replicates作出选择的金针虫数量(百分比)
Number of wireworms at different sides(Percentage)G检验
G-test竹笋
Bamboo shoots二氧化碳
CO2GP GH 24 h 20 139(80.35%) 34(19.65%) 68.37 *** 15.40 NS 48 h 20 142(80.23%) 35(19.77%) 47.69 *** 14.58 NS Table 3. Choice tests of M. cribricollis larvae between bamboo shoots and CO2 resource in a dual-choice olfactometer
-
固相微萃取法收集、鉴定雷竹笋挥发物17种,其中萜烯类化合物7种(均是倍半萜),羧酸类化合物2种,醇类、酯类及酚类化合物各1种,还有1种烷烃和1种烯烃类化合物(表4)。17种化合物中,相对含量最高的为烯烃类化合物,相对含量占比达66.62%,其次是倍半萜(27.98%),而7种倍半萜中以α-葎草烯含量最高,占16.49%(表4)。
物质峰编号
No. of
peak保留时间
Retention
time/min保留指数
Retention
indices/iu分子式
Molecular
formula化合物名称
Compounds相对含量±标准误
Relative content ± SE/%1 9.12 2705 C20H40O 1-(乙烯氧基)-十八烷 1-(ethenyloxy)-octadecane 0.68±0.21 2 10.11 1180 C9H10O 4-(1-甲基乙烯基)-苯酚 4-(1-methylethenyl)-phenol 0.19±0.03 3 10.65 1471 C11H22O2 十一烷酸 undecanoic acid 0.20±0.05 4 10.96 1620 C16H32 (Z)-7-十六碳烯 (Z)-7-hexadecene 2.58±0.42 5 11.04 1413 C14H30 tetradecane 十四烷 0.30±0.06 6 11.28 1430 C15H24 α-香柠檬烯 α-bergamotene 0.88±0.19 7 11.39 1494 C15H24 β-石竹烯 β-caryophyllene 3.07±1.87 8 11.60 1440 C15H24 (Z)-β-farnesene (Z)-β-法呢烯 0.42±0.11 9 11.64 1446 C15H24 β-倍半水芹烯 β-sesquiphellandrene 1.16±0.08 10 11.71 1579 C15H24 α-葎草烯 α-humulene 16.49±3.25 11 11.88 1304 C13H26 1-十三碳烯 1-tridecene 64.04±8.23 12 12.11 1500 C15H24 β-双油烯 β-bisabolene 2.84±0.98 13 12.23 1435 C15H24 γ-杜松烯 γ-cadinene 3.12±1.01 14 12.84 1790 C16H34O 2-己基-1-癸醇 2-hexyl-1-decanol 0.41±0.03 15 12.91 1910 C19H40 十九烷 nonadecane 0.84±0.92 16 17.43 1908 C16H22O4 邻苯二甲酸二异丁酯 diisobutyl phthalate 1.96±0.97 17 18.56 1968 C16H32O2 十六烷酸 hexadecanoic acid 0.82±0.31 Table 4. Relative content (%) and volatile components of Ph. violascens shoot collected by HS-SPME
-
植物挥发物在植食性昆虫搜寻和定位寄主过程中发挥着关键作用,尤其在黑暗、环境更为复杂的地下植食性昆虫的觅食过程中更为重要[13,15,23]。大量研究表明,CO2是地下植食性昆虫寄主定位的重要信号物质[23-24],如0.5%的CO2对玉米根萤叶甲幼虫有很强的引诱作用[25],且CO2浓度能调节其远距离寄主搜寻和定位行为[23]。本研究利用CO2释放剂证实了筛胸梳爪叩甲幼虫(竹林金针虫)对土壤中的CO2表现出显著的吸引作用,同时CO2浓度对竹林金针虫的定向移动有显著影响,与其它金针虫的相关研究结果吻合[12,17]。CO2浓度与金针虫行为密切相关,过低或过高均影响金针虫的定向选择,CO2浓度过低金针虫难以探测,过高的CO2浓度对金针虫表现出驱避或毒性作用[25-26],研究表明,4 g CO2释放剂颗粒的引诱效果最佳,增加CO2释放剂的量对竹林金针虫的趋向行为影响不显著。
尽管CO2对金针虫、蛴螬等地下植食性昆虫有吸引作用,但土壤环境中CO2普遍存在且浓度多变,地下植食性昆虫仍能快速找到寄主,有学者认为,地下植食性昆虫在缺乏视觉辅助的情况下,拥有更为精巧的化学通讯和寄主识别机制。植物在生长过程中,根系会释放大量的CO2和自身代谢产物(初生及次生代谢物),尤其是次生代谢物对地下昆虫的寄主搜索和选择行为有重要影响[11,15,27]。本研究中,在竹笋和CO2释放剂之间,筛胸梳爪叩甲幼虫显著偏好竹笋,这也表明竹笋的挥发物在金针虫寄主搜寻和定位过程中发挥重要作用。
目前对慈竹(Bambusa emeiensis L. C. Chia & H. L. Fung)、麻竹(Dendrocalamus latiflorus Munro)及绿竹(B. oldhamii Munro)等丛生竹笋[28-29],毛竹[30-31]和苦竹(Pleioblastus amarus (Keng) Keng f.)等散生竹笋的挥发物成分研究有过报道。竹笋挥发物组分因竹种、提取方法及提取部位的不同,挥发物组分差异显著,呈现出一定的风味特征。本研究采用固相微萃取气质联用法收集鉴定了雷竹笋(地下部分)挥发物组分17种,其中以烯烃和萜烯类化合物为主。萜烯类化合物是最为常见的植物挥发性组分,在植食性昆虫产卵及搜寻寄主过程中常发挥重要的作用[32-33]。如β-caryophyllene是玉米根萤叶甲[34]、α-葎草烯是甘薯蚁象(Cylas formicarius Fabr.)[35]、α-pinene及β-pinene是鳃金龟(M. melolontha L.)[36]等地下植食性昆虫幼虫定位寄主的关键信号物质。Barsics等研究表明,大麦根释放的hexanal, (E)-hex-2-enal及(E)-non-2-enal等4种醛类挥发物能吸引金针虫的定向移动[11]。本研究发现的α-葎草烯等7种相对含量较高的倍半萜,是否为筛胸梳爪叩甲幼虫定位竹笋的关键物质,将在后续研究中进一步测定。
大多地下植食性昆虫是世界性重大害虫,危害周期长、隐蔽性强,种群动态监测及防治一直是植物保护工作的重点和难点[37]。近年来,金针虫在浙江、福建及江西等核心竹笋产区爆发成灾,造成重大经济损失,如何绿色、经济、有效地控制竹林金针虫是竹笋产业健康发展中亟待解决的问题。本研究证实了竹笋挥发物及CO2对竹林金针虫有显著的吸引作用,并鉴定了竹笋挥发物组分,为对竹林金针虫具有引诱活性的信号物质的筛选及基于引诱剂的金针虫AK技术(Attract and kill strategy)研发提供了重要依据。
-
研究表明,筛胸梳爪叩甲幼虫对新鲜雷竹笋和CO2释放剂颗粒均有显著趋性,在竹笋和CO2释放剂之间,筛胸梳爪叩甲幼虫显著偏好新鲜竹笋,相比CO2释放剂,竹笋萌发时所释放的挥发物对竹林金针虫搜寻寄主更为有效。本研究分离鉴定了17种可能对筛胸梳爪叩甲幼虫具有吸引作用的萜烯类化合物,为研发高效的金针虫引诱剂,探索竹林金针虫的新型防控技术奠定了基础。
Larval Behavioural Responses of Melanotus cribricollis (Coleoptera: Elateridae) to the Volatiles Emitted from Bamboo Shoot and Carbon Dioxide
- Received Date: 2022-03-07
- Accepted Date: 2022-04-19
- Available Online: 2022-12-20
Abstract:




 DownLoad:
DownLoad: